 |
市场调查报告书
商品编码
1797739
前列腺癌生物标记市场机会、成长动力、产业趋势分析及 2025 - 2034 年预测Prostate Cancer Biomarkers Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2025 - 2034 |
||||||
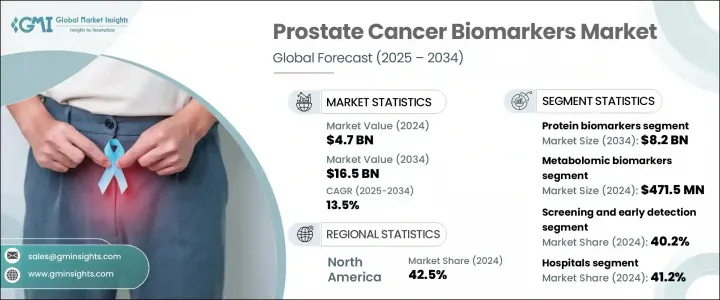
2024 年全球前列腺癌生物标记市场价值为 47 亿美元,预计到 2034 年将以 13.5% 的复合年增长率增长至 165 亿美元。市场成长主要得益于前列腺癌病例数量的增加(尤其是在老化男性群体中),以及先进诊断技术的进步。人们对早期筛检的认识不断提高,加上政府资助的医疗保健计划的增加,推动了初级保健机构对前列腺癌生物标记的采用。人工智慧驱动的评估、液体活检和基因组分析平台等下一代诊断技术的整合,正在增强早期检测能力,同时支援个人化治疗决策。新兴经济体也在增加对卫生基础设施的投资,这正在扩大精准检测方法的覆盖范围。这些生物标记驱动的策略正在透过提供更高的准确性、更低的侵入性和更快的诊断工作流程来重塑癌症治疗。
OPKO Health、贝克曼库尔特、Bio-Rad Laboratories 和 Bio-Techne 等行业主要参与者正引领前列腺癌生物标记市场的创新。这些公司不仅透过尖端诊断平台提高检测的准确性和速度,更致力于塑造个人化癌症治疗的未来。透过开发非侵入性、高灵敏度的生物标记检测方法,并将其与基于人工智慧的诊断系统相结合,它们能够更早地识别疾病,更好地监测病情进展,并制定更有效的治疗方案。
| 市场范围 | |
|---|---|
| 起始年份 | 2024 |
| 预测年份 | 2025-2034 |
| 起始值 | 47亿美元 |
| 预测值 | 165亿美元 |
| 复合年增长率 | 13.5% |
蛋白质生物标记细分市场在2024年创收24亿美元,预计2034年将达到82亿美元,复合年增长率为13.1%。其主导地位源自于广泛的临床认可及其在疾病检测和进展追踪中的常规应用。此类生物标记易于透过标准血液检测进行测量,由于其可靠性、成本效益以及与前列腺癌风险的强相关性,仍然是一线诊断工具。正在进行的研究正在不断改进这些生物标记技术,使其在保持准确性的同时减少不必要的侵入性诊断。新的基于生物标记的评分工具也在改善风险分层和患者选择,以便进行进一步检测,从而优化治疗途径。
在应用方面,筛检和早期检测领域在2024年占据了最高的市场份额,达到40.2%。这归因于公共卫生系统和个别临床实践对早期诊断的日益重视。如今,筛检平台专为高容量工作流程而设计,利用血液或尿液生物标记检测,以最少的处理流程提供快速可靠的结果。这些解决方案非常适合常规健康检查,并且在快速週转和易用性至关重要的门诊环境中迅速普及。非侵入性检测的便利性和可近性使其成为全球前列腺癌预防策略中不可或缺的一部分。
2024年,北美前列腺癌生物标记市场占据42.5%的市场。该地区受益于先进的医疗基础设施、较高的发病率以及加快的监管审批程序。在强劲的医疗支出和不断加强的公众意识宣传活动的推动下,美国和加拿大在采用尖端诊断平台方面继续保持领先地位。持续的研究投入、完善的诊断生态系统以及主要市场参与者的存在,进一步支持了该地区的创新。随着对可靠、快速和微创癌症诊断的需求不断增长,预计这一领先地位将持续下去。
Myriad Genetics、QIAGEN、西门子、Veracyte、Genomic Health、罗氏製药、MDxHealth、赛默飞世尔科技和默克集团等公司是重塑竞争格局的关键参与者。这些公司透过研发投资扩大检测组合,并加强与肿瘤实验室、学术机构和医院的合作。这些公司正在利用先进的基因组学和蛋白质组学技术来提高其生物标记平台的灵敏度和特异性。他们也进行策略合作,以促进全球分销、扩大生产规模并获得监管部门的批准,从而加快产品上市速度。许多公司专注于伴随诊断的开发,这与精准医疗直接相关,有助于增强治疗方案的一致性和患者预后。
目录
第一章:方法论与范围
第二章:执行摘要
第三章:行业洞察
- 产业生态系统分析
- 产业衝击力
- 成长动力
- 摄护腺癌发生率上升
- 基于生物标记的诊断技术的进展
- 公营和私营部门增加研发投资
- 透过生物标记检测实现个人化医疗的日益普及
- 产业陷阱与挑战
- 进阶诊断测试成本高昂
- 农村和欠发达地区的取得途径和认知度有限
- 市场机会
- 非侵入性生物标记平台投资不断增加
- 越来越多地使用组合生物标记组进行早期诊断
- 成长动力
- 成长潜力分析
- 监管格局
- 技术进步
- 当前的技术趋势
- 新兴技术
- 未来市场趋势
- 差距分析
- 波特的分析
- PESTEL分析
第四章:竞争格局
- 介绍
- 公司市占率分析
- 公司矩阵分析
- 主要市场参与者的竞争分析
- 竞争定位矩阵
- 关键进展
- 併购
- 伙伴关係和合作
- 新产品发布
- 扩张计划
第五章:市场估计与预测:按类型,2021 - 2034 年
- 主要趋势
- 基因生物标记
- 蛋白质生物标记
- 基于细胞的生物标记
- 代谢组学生物标誌物
第六章:市场估计与预测:按应用,2021 - 2034 年
- 主要趋势
- 筛检和早期检测
- 诊断和风险分层
- 预后和治疗监测
- 伴随诊断
第七章:市场估计与预测:依最终用途,2021 - 2034 年
- 主要趋势
- 医院
- 诊断实验室
- 癌症研究机构
- 生物製药公司
第八章:市场估计与预测:按地区,2021 - 2034 年
- 主要趋势
- 北美洲
- 我们
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 西班牙
- 义大利
- 荷兰
- 亚太地区
- 中国
- 印度
- 日本
- 澳洲
- 韩国
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中东和非洲
- 沙乌地阿拉伯
- 南非
- 阿联酋
第九章:公司简介
- Beckman Coulter
- Bio-Rad Laboratories
- Bio-Techne
- F. Hoffmann-La Roche
- Genomic health
- MDxHealth
- Merck KGaA
- Myriad Genetics
- OPKO Health
- QIAGEN
- Siemens
- Thermo Fisher Scientific
- Veracyte
The Global Prostate Cancer Biomarkers Market was valued at USD 4.7 billion in 2024 and is estimated to grow at a CAGR of 13.5% to reach USD 16.5 billion by 2034. The market growth is propelled by the rising number of prostate cancer cases, particularly in aging male populations, and the evolution of advanced diagnostics. Growing awareness of early screening, along with a rise in government-sponsored healthcare programs, is fueling adoption across primary care settings. The integration of next-generation diagnostics, such as artificial intelligence-driven assessments, liquid biopsies, and genomic analysis platforms, is enhancing early-stage detection while supporting personalized therapeutic decisions. Emerging economies are also increasing investments in health infrastructure, which is broadening access to precision-based testing methods. These biomarker-driven strategies are reshaping cancer care by offering better accuracy, reduced invasiveness, and faster diagnostic workflows.

Major industry contributors such as OPKO Health, Beckman Coulter, Bio-Rad Laboratories, and Bio-Techne are at the forefront of driving innovation in the prostate cancer biomarkers market. These companies are not only enhancing the accuracy and speed of detection through cutting-edge diagnostic platforms but also shaping the future of personalized cancer care. By developing non-invasive, highly sensitive biomarker assays and integrating them with AI-based diagnostic systems, they enable earlier identification of disease, better monitoring of progression, and more effective treatment planning.
| Market Scope | |
|---|---|
| Start Year | 2024 |
| Forecast Year | 2025-2034 |
| Start Value | $4.7 Billion |
| Forecast Value | $16.5 Billion |
| CAGR | 13.5% |
The protein biomarkers segment generated USD 2.4 billion in 2024 and is forecasted to reach USD 8.2 billion by 2034, growing at a CAGR of 13.1%. Their dominance is anchored in widespread clinical acceptance and their routine use for disease detection and progression tracking. Biomarkers in this category, which are easily measured through standard blood assays, remain the first-line diagnostic tools due to their proven reliability, cost-effectiveness, and strong correlation with prostate cancer risk. Ongoing research continues to refine these biomarker technologies, allowing them to reduce unnecessary invasive diagnostics while maintaining accuracy. New biomarker-based scoring tools are also improving risk stratification and patient selection for further testing, optimizing treatment pathways.
In terms of application, the screening and early detection segment held the highest share of the market in 2024, accounting for 40.2%. This is attributed to the growing emphasis on early diagnosis in both public health systems and individual clinical practices. Screening platforms are now designed for high-volume workflows and utilize blood or urine-based biomarker assays to deliver fast, reliable results with minimal processing. These solutions are highly suitable for routine health checks and are expanding rapidly in outpatient environments where fast turnaround and ease of use are critical. The convenience and accessibility of non-invasive tests are making them integral to prostate cancer prevention strategies globally.
North America Prostate Cancer Biomarkers Market held 42.5% share in 2024. The region benefits from advanced medical infrastructure, high rates of disease incidence, and accelerated pathways for regulatory approvals. The US and Canada continue to lead in adopting cutting-edge diagnostic platforms, driven by strong healthcare expenditure and rising public awareness campaigns. Continuous investment in research, a well-developed diagnostics ecosystem, and the presence of major market participants further support innovation in this region. This leadership position is expected to persist as demand grows for reliable, fast, and minimally invasive cancer diagnostics.
Companies such as Myriad Genetics, QIAGEN, Siemens, Veracyte, Genomic Health, F. Hoffmann-La Roche, MDxHealth, Thermo Fisher Scientific, and Merck KGaA are among the key players reshaping the competitive landscape. These include expanding their test portfolios through R&D investments and strengthening partnerships with oncology labs, academic institutions, and hospitals. Firms are leveraging advanced genomic and proteomic technologies to improve the sensitivity and specificity of their biomarker platforms. They're also entering strategic collaborations for global distribution, scaling up manufacturing, and securing regulatory approvals to speed up time-to-market. Many companies are focusing on companion diagnostics development, which ties directly into precision medicine, enhancing treatment alignment and patient outcomes.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research design
- 1.2.1 Research approach
- 1.2.2 Data collection methods
- 1.3 Data mining sources
- 1.3.1 Global
- 1.3.2 Regional/Country
- 1.4 Base estimates and calculations
- 1.4.1 Base year calculation
- 1.4.2 Key trends for market estimation
- 1.5 Primary research and validation
- 1.5.1 Primary sources
- 1.6 Forecast model
- 1.7 Research assumptions and limitations
Chapter 2 Executive Summary
- 2.1 Industry 3600 synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Type trends
- 2.2.3 Application trends
- 2.2.4 End use trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising incidence of prostate cancer
- 3.2.1.2 Advancements in biomarker-based diagnostic technologies
- 3.2.1.3 Increased R and D investments from the public and private sectors
- 3.2.1.4 Growing adoption of personalized medicine through biomarker testing
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High cost of advanced diagnostic tests
- 3.2.2.2 Limited access and awareness in rural and underdeveloped regions
- 3.2.3 Market opportunities
- 3.2.3.1 Rising investments in non-invasive biomarker platforms
- 3.2.3.2 Increasing use of combination biomarker panels for early diagnosis
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.5 Technological advancements
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Future market trends
- 3.7 Gap analysis
- 3.8 Porter's analysis
- 3.9 PESTEL analysis
Chapter 4 Competitive Landscape, 2024
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Mergers and acquisitions
- 4.6.2 Partnerships and collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Type, 2021 - 2034 ($ Mn)
- 5.1 Key trends
- 5.2 Genetic Biomarkers
- 5.3 Protein Biomarkers
- 5.4 Cell-based Biomarkers
- 5.5 Metabolomic Biomarkers
Chapter 6 Market Estimates and Forecast, By Application, 2021 - 2034 ($ Mn)
- 6.1 Key trends
- 6.2 Screening and early detection
- 6.3 Diagnosis and risk stratification
- 6.4 Prognosis and treatment monitoring
- 6.5 Companion diagnostics
Chapter 7 Market Estimates and Forecast, By End Use, 2021 - 2034 ($ Mn)
- 7.1 Key trends
- 7.2 Hospitals
- 7.3 Diagnostic laboratories
- 7.4 Cancer research institutes
- 7.5 Biopharmaceutical companies
Chapter 8 Market Estimates and Forecast, By Region, 2021 - 2034 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 India
- 8.4.3 Japan
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 Middle East and Africa
- 8.6.1 Saudi Arabia
- 8.6.2 South Africa
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 Beckman Coulter
- 9.2 Bio-Rad Laboratories
- 9.3 Bio-Techne
- 9.4 F. Hoffmann-La Roche
- 9.5 Genomic health
- 9.6 MDxHealth
- 9.7 Merck KGaA
- 9.8 Myriad Genetics
- 9.9 OPKO Health
- 9.10 QIAGEN
- 9.11 Siemens
- 9.12 Thermo Fisher Scientific
- 9.13 Veracyte





