 |
市场调查报告书
商品编码
1936611
创伤性脑损伤评估市场机会、成长要素、产业趋势分析及预测(2026年至2035年)Traumatic Brain Injuries Assessment Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035 |
||||||
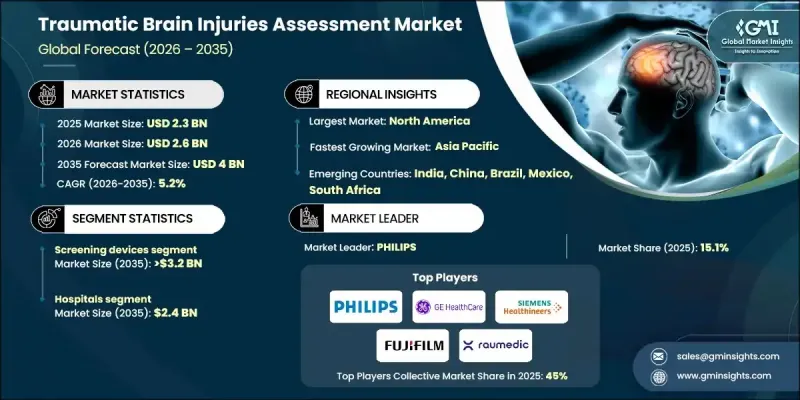
全球创伤性脑损伤评估市场预计到 2025 年将达到 23 亿美元,预计到 2035 年将达到 40 亿美元,年复合成长率为 5.2%。
市场扩张的驱动因素包括头部创伤病例的增加、运动和军事相关脑外伤病例的增多,以及神经影像和诊断技术的不断进步。脑外伤评估涉及全面的临床、认知和神经系统评估,以确定脑损伤的严重程度、功能影响和整体预后。神经系统测试、行为评估、影像技术和生物标记分析被整合在一起,以促进快速诊断、治疗方案製定和预后判断。此外,CT、MRI、EEG和基于生物标记的诊断技术的创新使得能够更快、更准确地检测出以前往往被忽视的轻度和中度脑外伤,从而进一步推动了市场成长。人工智慧驱动的分析、客观的诊断工具和基于血液的筛检解决方案的日益普及也在推动产业转型。
| 市场覆盖范围 | |
|---|---|
| 开始年份 | 2025 |
| 预测期 | 2026-2035 |
| 起始值 | 23亿美元 |
| 预测金额 | 40亿美元 |
| 复合年增长率 | 5.2% |
由于其快速、经济高效且非侵入性的评估能力,预计到2025年,筛检设备市场份额将达到80.1%。这些设备对于在急诊和临床环境中早期发现和优先处理疑似脑损伤至关重要,因此得到了广泛应用。
2025年,医院领域占据57.9%的市场份额,预计2026年至2035年间将达到24亿美元。医院是头部创伤患者的主要治疗中心,大量患者涌入使得先进的评估工具必不可少。道路交通事故和老龄化相关损伤的增加是导致住院人数上升的主要因素,进一步凸显了对高效脑外伤评估解决方案的需求。
预计到2025年,美国创伤性脑损伤评估市场规模将达到8.816亿美元。北美市场需求主要受意外事故、跌倒及其他原因导致的头部损伤所驱动,这使得急救医疗服务提供者对快速且准确的诊断工具提出了更高的要求。医院和急诊对先进神经诊断解决方案的依赖,也持续推动美国市场的成长。
目录
第一章调查方法和范围
第二章执行摘要
第三章业界考察
- 生态系分析
- 产业影响因素
- 司机
- 创伤性脑损伤发生率不断上升
- 创伤性脑损伤评估的技术进步
- 对创伤性脑损伤 (TBI) 评估的非侵入性设备的需求日益增长
- 提高对创伤性脑损伤的认识
- 产业潜在风险与挑战
- 熟练专业人员短缺
- 评估设备高成本
- 市场机会
- 携带式和照护现场评估工具的开发
- 新兴市场蕴藏着巨大的未开发需求
- 司机
- 成长潜力分析
- 监管环境
- 北美洲
- 欧洲
- 亚太地区
- 科技趋势
- 当前技术趋势
- 新兴技术
- 消费者洞察
- 2024年定价分析
- 未来市场趋势
- 价值链分析
- 波特五力分析
- PESTEL 分析
- 差距分析
第四章 竞争情势
- 介绍
- 企业矩阵分析
- 公司市占率分析
- 世界
- 北美洲
- 欧洲
- 亚太地区
- 竞争定位矩阵
- 主要市场公司的竞争分析
- 重大进展
- 併购
- 伙伴关係与合作
- 新产品发布
- 扩张计划
第五章 2022-2035年按产品分類的市场估算与预测
- 筛检设备
- 电脑断层扫描(CT)
- 磁振造影(MRI)
- 其他筛检设备
- 监控设备
- 颅内压(ICP)监测
- 脑组织氧分压(pBrO2)
6. 依最终用途分類的市场估计与预测,2022-2035 年
- 医院
- 诊断中心
- 其他用途
第七章 2022-2035年各地区市场估算与预测
- 北美洲
- 我们
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 西班牙
- 义大利
- 荷兰
- 亚太地区
- 中国
- 日本
- 印度
- 澳洲
- 韩国
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中东和非洲
- 南非
- 沙乌地阿拉伯
- 阿拉伯聯合大公国
第八章 公司简介
- BIOMERIEUX
- BrainScope
- COMPUMEDICS
- FUJIFILM
- GE Healthcare
- InfraScan
- INTEGRA
- Natus Medical
- NIHON KOHDEN
- Oculogica
- PHILIPS
- RAUMEDIC
- SIEMENS Healthineers
- ZOLL Medical
The Global Traumatic Brain Injuries Assessment Market was valued at USD 2.3 billion in 2025 and is estimated to grow at a CAGR of 5.2% to reach USD 4 billion by 2035.

Market expansion is driven by the increasing prevalence of head injuries, the rising number of sports-related and military-associated TBIs, and ongoing advancements in neuroimaging and diagnostic technologies. Traumatic brain injury assessment involves a comprehensive clinical, cognitive, and neurological evaluation to determine the severity, functional impact, and overall outcome of brain injuries. It incorporates neurological tests, behavioral assessments, imaging techniques, and biomarker analysis to facilitate prompt diagnosis, treatment planning, and prognosis. The market is further fueled by innovations in CT, MRI, EEG, and biomarker-based diagnostics, enabling faster and more accurate detection of mild and moderate TBIs, which were often overlooked previously. Growing adoption of AI-driven analytics, objective diagnostic tools, and blood-based screening solutions is also reshaping the industry.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $2.3 Billion |
| Forecast Value | $4 Billion |
| CAGR | 5.2% |
The screening devices segment held an 80.1% share in 2025, due to their rapid, cost-effective, and non-invasive assessment capabilities. These devices are essential for early detection and prioritization of suspected brain injuries in emergency and point-of-care settings, driving their widespread adoption.
The hospitals segment held a 57.9% share in 2025, expected to reach USD 2.4 billion during 2026-2035. Hospitals remain the primary care centers for head injury patients, with high patient inflow demanding advanced assessment tools. Rising road traffic incidents and age-related injuries are key factors contributing to increased hospital admissions, reinforcing the need for efficient TBI evaluation solutions.
U.S. Traumatic Brain Injuries Assessment Market was valued at USD 881.6 million in 2025. North America's demand is largely fueled by head injuries stemming from accidents, falls, and other causes, creating significant pressure on emergency care providers to adopt rapid and accurate diagnostic tools. The reliance on advanced neurodiagnostic solutions in hospitals and emergency units continues to drive the U.S. market forward.
Key players in the Global Traumatic Brain Injury Assessment Market include Philips, BrainScope, INTEGRA, RAUMEDIC, Fujifilm, Natus Medical, GE Healthcare, BIOMERIEUX, Oculogica, NIHON KOHDEN, InfraScan, SIEMENS Healthineers, COMPUMEDICS, and ZOLL Medical. To strengthen their presence, companies are pursuing strategies such as launching advanced diagnostic tools with faster turnaround times, integrating AI and machine learning into imaging platforms, and developing non-invasive biomarker-based tests. Many firms are expanding geographically to access high-growth regions and forming partnerships with hospitals, research institutions, and emergency care providers. Investment in marketing campaigns, training programs for healthcare professionals, and after-sales support ensures broader adoption and improved clinical outcomes.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definitions
- 1.2 Research design
- 1.2.1 Research approach
- 1.2.2 Data collection methods
- 1.3 Data mining sources
- 1.3.1 Global
- 1.3.2 Regional/country
- 1.4 Base estimates and calculations
- 1.4.1 Base year calculation
- 1.4.2 Key trends for market estimation
- 1.5 Primary research and validation
- 1.5.1 Primary sources
- 1.6 Forecast model
- 1.7 Research assumptions and limitations
Chapter 2 Executive Summary
- 2.1 Industry 3600 synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Product trends
- 2.2.3 End use trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Growing incidence of traumatic brain injuries
- 3.2.1.2 Technological advancements in traumatic brain injuries assessment
- 3.2.1.3 Increasing demand for non-invasive devices for TBI assessment
- 3.2.1.4 Rising awareness regarding traumatic brain injury
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Dearth of skilled professionals
- 3.2.2.2 High cost of assessment devices
- 3.2.3 Market opportunities
- 3.2.3.1 Development of portable and point-of-care assessment tools
- 3.2.3.2 Emerging markets with untapped demand
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.5 Technology landscape
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Consumer insights
- 3.7 Pricing analysis, 2024
- 3.8 Future market trends
- 3.9 Value chain analysis
- 3.10 Porter's analysis
- 3.11 PESTEL analysis
- 3.12 Gap analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company matrix analysis
- 4.3 Company market share analysis
- 4.3.1 Global
- 4.3.2 North America
- 4.3.3 Europe
- 4.3.4 Asia Pacific
- 4.4 Competitive positioning matrix
- 4.5 Competitive analysis of major market players
- 4.6 Key developments
- 4.6.1 Mergers & acquisitions
- 4.6.2 Partnerships & collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Screening devices
- 5.2.1 Computerized tomography (CT) scan
- 5.2.2 Magnetic resonance imaging (MRI)
- 5.2.3 Other screening devices
- 5.3 Monitoring devices
- 5.3.1 Intracranial pressure (ICP) monitoring
- 5.3.2 Partial pressure of oxygen in brain tissue (pBrO2)
Chapter 6 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Hospitals
- 6.3 Diagnostic centers
- 6.4 Other end use
Chapter 7 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 North America
- 7.2.1 U.S.
- 7.2.2 Canada
- 7.3 Europe
- 7.3.1 Germany
- 7.3.2 UK
- 7.3.3 France
- 7.3.4 Spain
- 7.3.5 Italy
- 7.3.6 Netherlands
- 7.4 Asia Pacific
- 7.4.1 China
- 7.4.2 Japan
- 7.4.3 India
- 7.4.4 Australia
- 7.4.5 South Korea
- 7.5 Latin America
- 7.5.1 Brazil
- 7.5.2 Mexico
- 7.5.3 Argentina
- 7.6 Middle East and Africa
- 7.6.1 South Africa
- 7.6.2 Saudi Arabia
- 7.6.3 UAE
Chapter 8 Company Profiles
- 8.1 BIOMERIEUX
- 8.2 BrainScope
- 8.3 COMPUMEDICS
- 8.4 FUJIFILM
- 8.5 GE Healthcare
- 8.6 InfraScan
- 8.7 INTEGRA
- 8.8 Natus Medical
- 8.9 NIHON KOHDEN
- 8.10 Oculogica
- 8.11 PHILIPS
- 8.12 RAUMEDIC
- 8.13 SIEMENS Healthineers
- 8.14 ZOLL Medical







