 |
市场调查报告书
商品编码
2019252
猪疫苗市场商机、成长要素、产业趋势分析及2026-2035年预测。Swine Vaccines Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035 |
||||||
预计到 2025 年,全球猪隻疫苗市场价值将达到 16 亿美元,并有望以 6.3% 的复合年增长率成长,到 2035 年达到 29 亿美元。
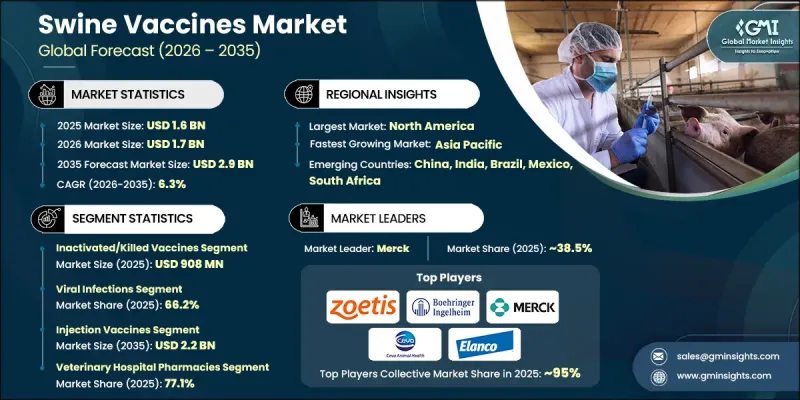
全球市场对猪肉及相关产品的需求不断增长,推动了市场成长,进而促使人们更加重视维护牲畜健康和提高生产力。猪疫苗在保护猪隻方面发挥着至关重要的作用,它能活化猪的免疫系统,使其免受包括病毒、细菌和其他病原体在内的多种传染性病原体的侵害。人口增长、都市化进程加快以及饮食习惯的改变,推动了畜牧业的持续扩张,也进一步刺激了对有效动物保健解决方案的需求。随着生产系统集约化程度的提高,生产者越来越重视动物保健措施。疫苗接种正成为降低疾病风险、提高存活率和保护经济利益的关键策略。此外,对粮食安全的担忧也促使人们采用先进的疾病预防措施,这进一步推动了猪隻疫苗市场的成长。
| 市场范围 | |
|---|---|
| 开始年份 | 2025 |
| 预测期 | 2026-2035 |
| 上市时的市场规模 | 16亿美元 |
| 预测金额 | 29亿美元 |
| 复合年增长率 | 6.3% |
灭活疫苗市场地位强劲,2025年销售额达9.08亿美元。这类疫苗因其安全性高、稳定性佳而广受欢迎。其保质期长、储存条件相对简单,使其适用于大规模免疫接种计划。因此,灭活疫苗仍然是控制猪群常见疾病的可靠选择。
预计到2025年,病毒感染疾病细分市场将占据66.2%的市场份额,并在2026年至2035年间以6.4%的复合年增长率成长。该细分市场的成长主要受猪群病毒性疾病的广泛流行及其经济影响的驱动。这些感染疾病具有高度传染性,会对动物健康、生产力和农场整体盈利造成重大影响。在高密度饲养环境中,感染的快速传播进一步凸显了疫苗接种作为主要预防措施的重要性,从而巩固了该细分市场的主导地位。
预计北美猪疫苗市场规模将在2025年达到6.826亿美元,到2035年将达到12亿美元,2026年至2035年间的复合年增长率(CAGR)为5.8%。该地区的主导地位得益于先进的畜牧管理系统、完善的兽医基础设施以及对优质猪肉产品的强劲需求。美国凭藉其大规模的生猪生产能力以及在国内消费和出口方面的重要地位,仍然是该地区的主要贡献者。持续重视疾病预防以及主要企业的存在,进一步巩固了该地区强劲的市场地位。
目录
第一章:调查方法和范围
第二章执行摘要
第三章业界考察
- 生态系分析
- 供应商情况
- 每个阶段增加的价值
- 影响价值链的因素
- 影响产业的因素
- 促进因素
- 通用感染疾病发生率增加
- 畜牧业的扩张以及人们对粮食安全的担忧
- 疫苗技术的进步
- 动物疾病发生率增加
- 产业潜在风险与挑战
- 高昂的研发成本和漫长的研发週期
- 低温运输基础设施的限制因素
- 监理复杂性和核准延误
- 市场机会
- mRNA和下一代平台的采用
- 耐热疫苗的研发
- 联合疫苗创新
- 促进因素
- 成长潜力分析
- 监理情势(基于初步调查)
- 北美洲
- 我们
- 加拿大
- 欧洲
- 亚太地区
- 北美洲
- 疫苗技术的演进与创新
- 当前技术趋势
- 新兴技术
- 价格分析(基于初步调查)
- 专利分析(基于初步研究)
- 临床试验和研发管线分析(基于初步调查)
- 未来市场趋势
- 人工智慧和生成式人工智慧对市场的影响
- 波特五力分析
- PESTEL 分析
第四章 竞争情势
- 介绍
- 企业市占率分析
- 企业矩阵分析
- 主要市场公司的竞争分析
- 竞争定位矩阵
- 主要进展
- 併购
- 伙伴关係与合作
- 新产品发布
- 业务拓展计划
第五章 市场估计与预测:依类型划分,2022-2035年
- 活病毒疫苗
- 灭活疫苗
- 病毒载体疫苗
- 药效疫苗
- 其他疫苗
第六章 市场估计与预测:依应用领域划分,2022-2035年
- 细菌感染疾病
- 病毒感染疾病
- 寄生虫感染疾病
- 其他用途
第七章 市场估计与预测:依给药途径划分,2022-2023年
- 注射疫苗
- 口服疫苗
- 浸泡式和喷洒式疫苗
第八章 市场估算与预测:依通路划分,2022-2035年
- 兽药
- 普通药房
- 电子商务
第九章 市场估计与预测:依地区划分,2022-2035年
- 北美洲
- 我们
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 西班牙
- 义大利
- 荷兰
- 亚太地区
- 中国
- 印度
- 日本
- 澳洲
- 韩国
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中东和非洲
- 南非
- 阿拉伯聯合大公国
第十章:公司简介
- Addison Biological Laboratory
- Bioveta
- Boehringer Ingelheim
- Ceva Sante Animale
- Colorado Serum Company
- Elanco Animal Health Incorporated
- HIPRA SA
- Indian Immunologicals
- Merck
- Biogenesis Bago
- Vaxxinova(EW Group)
- Vibac
- Zoetis
The Global Swine Vaccines Market was valued at USD 1.6 billion in 2025 and is estimated to grow at a CAGR of 6.3% to reach USD 2.9 billion by 2035.

Market growth is driven by the increasing demand for pork and related products across global markets, which is placing greater emphasis on maintaining herd health and improving productivity. Swine vaccines play a critical role in protecting pigs by activating their immune systems to defend against a range of infectious agents, including viruses, bacteria, and other pathogens. The ongoing expansion of the livestock sector, fueled by population growth, urbanization, and evolving dietary habits, continues to accelerate demand for effective animal healthcare solutions. As production systems become more intensive to enhance output and efficiency, producers are placing stronger focus on preventive healthcare practices. Vaccination is emerging as a key strategy to reduce disease risks, improve survival rates, and protect economic returns. In addition, concerns related to food security are encouraging the adoption of advanced disease prevention measures, further supporting the growth of the swine vaccines market.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $1.6 Billion |
| Forecast Value | $2.9 Billion |
| CAGR | 6.3% |
The inactivated or killed vaccines segment generated USD 908 million in 2025, reflecting its strong position in the market. These vaccines are widely preferred due to their established safety profile and stability. Their extended shelf life and relatively straightforward storage requirements make them suitable for large-scale immunization programs. As a result, they continue to be a reliable option for managing commonly occurring diseases in swine populations.
The viral infections segment accounted for 66.2% share in 2025 and is expected to grow at a CAGR of 6.4% during 2026-2035. This segment's growth is driven by the widespread presence and economic impact of viral diseases affecting swine populations. Such infections are highly transmissible and can significantly affect animal health, productivity, and overall farm profitability. Their rapid spread in high-density farming environments reinforces the importance of vaccination as a primary preventive approach, supporting the segment's dominant position.
North America Swine Vaccines Market captured USD 682.6 million in 2025 and is projected to reach USD 1.2 billion by 2035, growing at a CAGR of 5.8% between 2026 and 2035. The region's leadership is supported by advanced livestock management systems, well-developed veterinary healthcare infrastructure, and strong demand for high-quality pork production. The United States remains a major contributor due to its large-scale swine production capacity and significant role in both domestic consumption and exports. Continued focus on disease prevention and the presence of leading animal health companies further reinforce the region's strong market position.
Key players operating in the Global Swine Vaccines Market include Zoetis, Merck, Boehringer Ingelheim, Elanco Animal Health Incorporated, Ceva Sante Animale, HIPRA S.A., Biogenesis Bago, Vaxxinova (EW Group), Indian Immunologicals, Bioveta, Vibac, Colorado Serum Company, and Addison Biological Laboratory. Companies in the swine vaccines market are strengthening their market position through continuous research and development, strategic partnerships, and expansion into emerging regions. They are focusing on developing advanced vaccines with improved efficacy, broader protection, and enhanced safety profiles. Collaborations with veterinary service providers and livestock producers are helping companies expand their reach and improve product adoption. In addition, manufacturers are investing in production capacity expansion and supply chain optimization to ensure consistent availability. Companies are also emphasizing regulatory compliance and quality standards while exploring innovative vaccine technologies to address evolving disease challenges and maintain a competitive edge in the market.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research approach
- 1.3 Quality commitments
- 1.3.1 GMI AI policy and data integrity commitment
- 1.3.1.1 Source consistency protocol
- 1.3.1 GMI AI policy and data integrity commitment
- 1.4 Research trail and confidence scoring
- 1.4.1 Research trail components
- 1.4.2 Scoring components
- 1.5 Data collection
- 1.5.1 Partial list of primary sources
- 1.6 Data mining sources
- 1.6.1 Paid sources
- 1.6.1.1 Sources, by region
- 1.6.1 Paid sources
- 1.7 Base estimates and calculations
- 1.7.1 Revenue share analysis
- 1.7.2 Base year calculation
- 1.8 Forecast model
- 1.9 Research transparency addendum
- 1.9.1 Source attribution framework
- 1.9.2 Quality assurance metrics
- 1.9.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Type trends
- 2.2.3 Application trends
- 2.2.4 Route of administration trends
- 2.2.5 Distribution channel trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.1.1 Supplier landscape
- 3.1.2 Value addition at each stage
- 3.1.3 Factors affecting the value chain
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising incidence of zoonotic diseases
- 3.2.1.2 Expanding livestock industry and food security concerns
- 3.2.1.3 Advancements in vaccine technology
- 3.2.1.4 Increasing outbreaks of animal diseases
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High R&D costs & long development timelines
- 3.2.2.2 Cold chain infrastructure limitations
- 3.2.2.3 Regulatory complexity & approval delays
- 3.2.3 Market opportunities
- 3.2.3.1 mRNA & next-generation platform adoption
- 3.2.3.2 Thermostable vaccine development
- 3.2.3.3 Combination vaccine innovation
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape (Driven by Primary Research)
- 3.4.1 North America
- 3.4.1.1 U.S.
- 3.4.1.2 Canada
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.1 North America
- 3.5 Vaccine technology evolution and innovation
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Pricing analysis (Driven by Primary Research)
- 3.7 Patent analysis (Driven by Primary Research)
- 3.8 Clinical trial/pipeline analysis (Driven by Primary Research)
- 3.9 Future market trends
- 3.10 Impact of AI & generative AI on the market
- 3.11 Porter's analysis
- 3.12 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Live attenuated vaccines
- 5.3 Inactivated/killed vaccines
- 5.4 Viral vector vaccines
- 5.5 mRNA vaccines
- 5.6 Other vaccines
Chapter 6 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Bacterial infections
- 6.3 Viral infections
- 6.4 Parasitic infections
- 6.5 Other applications
Chapter 7 Market Estimates and Forecast, By Route of Administration, 2022 - 203($ Mn)
- 7.1 Key trends
- 7.2 Injection vaccines
- 7.3 Oral vaccines
- 7.4 Immersion/spray vaccines
Chapter 8 Market Estimates and Forecast, By Distribution Channel, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 Veterinary hospital pharmacies
- 8.3 Retail pharmacies
- 8.4 E-commerce
Chapter 9 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 9.1 Key trends
- 9.2 North America
- 9.2.1 U.S.
- 9.2.2 Canada
- 9.3 Europe
- 9.3.1 Germany
- 9.3.2 UK
- 9.3.3 France
- 9.3.4 Spain
- 9.3.5 Italy
- 9.3.6 Netherlands
- 9.4 Asia Pacific
- 9.4.1 China
- 9.4.2 India
- 9.4.3 Japan
- 9.4.4 Australia
- 9.4.5 South Korea
- 9.5 Latin America
- 9.5.1 Brazil
- 9.5.2 Mexico
- 9.5.3 Argentina
- 9.6 Middle East and Africa
- 9.6.1 South Africa
- 9.6.2 UAE
Chapter 10 Company Profiles
- 10.1 Addison Biological Laboratory
- 10.2 Bioveta
- 10.3 Boehringer Ingelheim
- 10.4 Ceva Sante Animale
- 10.5 Colorado Serum Company
- 10.6 Elanco Animal Health Incorporated
- 10.7 HIPRA S.A.
- 10.8 Indian Immunologicals
- 10.9 Merck
- 10.10 Biogenesis Bago
- 10.11 Vaxxinova (EW Group)
- 10.12 Vibac
- 10.13 Zoetis







