 |
市场调查报告书
商品编码
1877351
全球患者来源异种移植/PDX模型市场(按模型类型和服务、移植方法、肿瘤类型、应用、最终用户和地区划分)—预测至2030年Patient Derived Xenografts/PDX Models Market by Type, Service, Implantation Method, Tumor Type, Application - Global Forecast to 2030 |
||||||
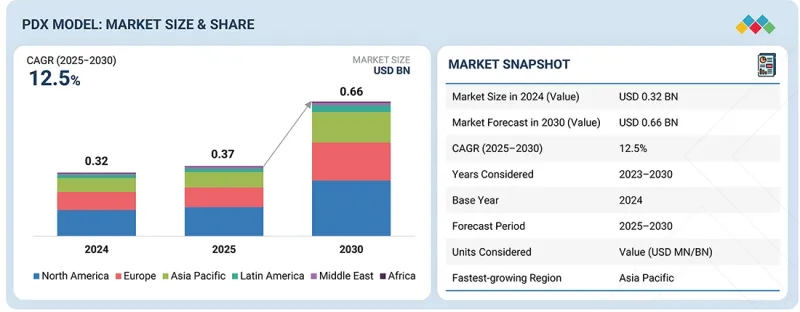
PDX 车型市场预计将从 2025 年的 3.7 亿美元增长到 2030 年的 6.6 亿美元,预测期内复合年增长率为 12.5%。
该市场的主要成长要素是癌症发病率的上升以及对能够忠实模拟人类肿瘤生物学的有效临床前模型日益增长的需求。此外,患者来源的异种移植(PDX)模型在肿瘤学研究中的日益普及,例如用于评估药物疗效、发现生物标记和开发个人化治疗方案,也推动了市场成长。同时,免疫力缺乏小鼠模型的进步及其与次世代定序技术的结合,也提升了PDX模型的转换价值。
| 调查范围 | |
|---|---|
| 调查期 | 2023-2030 |
| 基准年 | 2024 |
| 预测期 | 2025-2030 |
| 目标单元 | 金额(十亿美元) |
| 部分 | 按型号和服务、植入方法、肿瘤类型、应用、最终用户、区域 |
| 目标区域 | 北美、欧洲、亚太地区、拉丁美洲、中东和非洲 |
然而,PDX 模型开发成本高成本、耗时较长,以及伦理方面的考量和模型可重复性的差异,仍限制了其在市场上的广泛应用。
由于原位移植模型能够更好地模拟人类癌症中观察到的自然肿瘤微环境和疾病进展,预计其在患者来源异种移植(PDX)模型市场中将以最高的复合年增长率增长。该技术能够将癌细胞移植到其原始组织部位,更精确地再现肿瘤-基质相互作用、血管生成和转移扩散。因此,与其他模型类型相比,原位移植模型在评估药物疗效和抗药性机制方面具有更高的转化相关性和预测准确性。标靶治疗和免疫疗法的日益普及也进一步推动了对原位移植模型的需求,因为原位移植模型能够提供更接近生理环境的治疗反应评估方法。此外,影像和显微外科技术的进步提高了原位PDX研究的可重复性和监测性,使其在临床前研究中更容易取得且更可靠。这些优势共同推动了全球PDX模型市场中原位移植模型的快速成长。
胃肠道(GI)肿瘤模型在患者来源异种移植(PDX)模型市场中占据最大份额。这主要归因于消化器官系统恶性肿瘤(包括大肠直肠癌、胃癌、胰臟癌和肝癌)在全球范围内的高发生率。这些癌症是癌症相关死亡的主要原因之一,因此对能够准确模拟肿瘤异质性和进展的可靠临床前模型有着强烈的需求。 GI 消化器官系统模型能够忠实地保留患者肿瘤的组织学和遗传学特征,从而能够对治疗效果和抗药性机制进行高度预测性的评估。它们在评估标靶治疗、免疫疗法和联合治疗策略方面的强大应用,使其成为转化肿瘤学研究的基础。此外,精准医疗的日益普及以及针对胃肠道癌症的临床试验数量的不断增加,正在加速这些模型的应用。移植技术的不断进步和精细的分子分析进一步提升了消化器官系统 PDX模型的效用,巩固了其在全球市场的主导地位。
预计在预测期内,美国患者来源异种移植(PDX)模型市场将保持最高的成长率。这一增长主要得益于大型製药和生物技术公司的强大影响力、完善的研究基础设施以及广泛的肿瘤学研究倡议。癌症发生率的上升,以及个人化医疗和标靶治疗的日益普及,显着推动了对PDX模型的需求。此外,对转化和临床前癌症研究投入的不断增加,以及产学研之间的密切合作,也促进了市场的扩张。
此外,美国先进的实验室设施、完善的生物银行系统以及政府对癌症药物研发的大力支持,进一步巩固了其在全球市场的地位。美国的法规环境也促进了创新,同时确保了基于PDX模型的研究的可靠性和伦理合规性。这些因素促成了PDX模型的快速普及,巩固了北美在全球市场的主导地位。
本报告研究了全球患者来源异种移植/PDX 模型市场,并按模型类型和服务、移植方法、肿瘤类型、应用、最终用户、区域趋势和公司概况对其进行了细分。
目录
第一章 引言
第二章执行摘要
第三章重要考察
第四章 市场概览
- 介绍
- 市场动态
- 未满足的需求和閒置频段
- 相互关联的市场与跨产业机会
- 1/2/3级玩家的策略倡议
第五章 产业趋势
- 波特五力分析
- 生态系分析
- 价值链分析
- 定价分析
- 影响客户业务的趋势/颠覆性因素
- 专利分析
- 贸易数据分析
- 2025年美国关税对病人来源异种移植(PDX)模型市场的影响
- 宏观经济展望
- 2025-2026 年主要会议和活动
- 投资和资金筹措方案
- 案例研究分析
第六章:技术进步、人工智慧影响、专利、创新与未来应用
- 关键新兴技术
- 互补技术
- 技术蓝图
- 未来应用
- 人工智慧/生成式人工智慧对病患来源异种移植(PDX)模型市场的影响
第七章永续性和监管环境
- 地方法规和合规性
- 永续性影响和监管政策倡议
- 认证、标籤和环境标准
第八章:顾客状况与购买行为
- 决策流程
- 买方相关人员和采购评估标准
- 招募障碍和内部挑战
- 终端用户产业未满足的需求
9. 病人来源异种移植 (PDX) 模型市场,依模型类型和服务划分
- 介绍
- PDX 型号
- 服务
10. 病人来源异种移植(PDX)模型市场,依移植方法划分
- 介绍
- 皮下植入
- 原位移植
- 其他的
11. 病人来源异种移植(PDX)模型市场,依肿瘤类型划分
- 介绍
- 胃肠道肿瘤模型
- 妇科肿瘤模型
- 呼吸系统肿瘤模型
- 泌尿系统肿瘤模型
- 血液肿瘤模型
- 其他的
12. 病人来源异种移植(PDX)模型市场,依应用领域划分
- 介绍
- 临床前研究
- 生物标记分析
- 转化研究
- 生物银行
13. 病人来源异种移植(PDX)模型市场,依最终用户划分
- 介绍
- 製药和生物技术公司
- CROS 和 CDMOS
- 学术研究机构
14. 病人来源异种移植(PDX)模型市场(按地区划分)
- 介绍
- 北美洲
- 美国
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 义大利
- 西班牙
- 其他的
- 亚太地区
- 中国
- 日本
- 印度
- 澳洲
- 韩国
- 其他的
- 拉丁美洲
- 巴西
- 墨西哥
- 其他的
- 中东
- 海湾合作委员会国家
- 其他的
- 非洲
第十五章 竞争格局
- 介绍
- 主要参与企业的策略/优势
- 2022-2024年收入分析
- 2024年市占率分析
- 公司估值矩阵:主要参与企业,2024 年
- 公司估值矩阵:Start-Ups/中小企业,2024 年
- 估值和财务指标
- 品牌/产品对比
- 竞争场景
第十六章:公司简介
- 主要参与企业
- CHARLES RIVER LABORATORIES
- THE JACKSON LABORATORY
- INOTIV
- JSR CORPORATION
- GENOWAY
- WUXI APPTEC+WUXI BIOLOGICS
- BIOCYTOGEN PHARMACEUTICALS CO., LTD.
- TACONIC BIOSCIENCES, INC.
- ONCODESIGN SERVICES(ONCODESIGN PRECISION MEDICINE)
- CHAMPIONS ONCOLOGY, INC.
- PHARMARON
- SYNGENE INTERNATIONAL LIMITED
- LABCORP
- EUROFINS SCIENTIFIC
- VITALSTAR BIOTECHNOLOGY CO., LTD.
- 其他公司
- CREATIVE ANIMODEL
- JOINN LABORATORIES(CHINA)CO., LTD.
- CREATIVE BIOLABS
- ARAGEN LIFE SCIENCES LTD.
- PHARMTEST SERVICES
- LIDE SHANGHAI BIOTECH CO., LTD.
- CERTIS ONCOLOGY SOLUTIONS
- INNOSER
- SHANGHAI CHEMPARTNER
- IVRS AB
第十七章调查方法
第十八章附录
The PDX model market is expected to reach USD 0.66 billion in 2030 from USD 0.37 billion in 2025, at a CAGR of 12.5% during the forecast period. The market is primarily driven by the increasing incidence of cancer and the growing demand for effective preclinical models that closely replicate human tumor biology. The increasing use of patient-derived xenograft (PDX) models in oncology research to evaluate drug efficacy, discover biomarkers, and inform personalized treatment approaches further supports market growth. Additionally, advancements in immunodeficient mouse models and the integration of PDX platforms with next-generation sequencing technologies are enhancing their translational value.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2023-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD billion) |
| Segments | Implantation Method, Type, Tumor Type, Applications, End User |
| Regions covered | North America, Europe, the Asia Pacific, Latin America, the Middle East, and Africa |
However, the high cost and time-intensive nature of PDX model development, along with ethical considerations and variability in model reproducibility, continue to restrain widespread market adoption.

"Orthotopic implantation segment is expected to grow at the highest CAGR during the forecast period"
The orthotopic implantation segment is expected to grow at the highest CAGR in the patient-derived xenograft (PDX) model market owing to its superior ability to mimic the natural tumor environment and disease progression seen in human cancers. This technique allows tumor cells to be implanted at their original tissue site, enabling a more accurate representation of tumor-stroma interactions, angiogenesis, and metastatic spread. As a result, orthotopic models provide higher translational relevance and predictive accuracy for assessing drug efficacy and resistance mechanisms compared to other model types. The increasing adoption of targeted therapies and immunotherapies has further fueled the demand for orthotopic models, as they provide a more physiologically relevant context for evaluating therapeutic responses. Additionally, advancements in imaging modalities and microsurgical techniques have improved the reproducibility and monitoring of orthotopic PDX studies, making them more accessible and reliable for preclinical research. These advantages collectively drive the rapid growth of the orthotopic implantation segment in the global PDX model market.
"Gastrointestinal tumor model segment holds the largest share of the market"
The gastrointestinal (GI) tumor model segment holds the largest share of the patient-derived xenograft (PDX) model market, driven by the high global incidence of gastrointestinal malignancies, including colorectal, gastric, pancreatic, and liver cancers. These cancers are among the leading causes of cancer-related mortality, creating a strong demand for reliable preclinical models that accurately replicate tumor heterogeneity and progression. Gastrointestinal PDX models closely preserve the histological and genetic characteristics of patient tumors, enabling more predictive evaluation of therapeutic efficacy and resistance mechanisms. Their robust application in assessing targeted therapies, immunotherapies, and combination treatment strategies has made them a cornerstone of translational oncology research. Furthermore, the growing emphasis on precision medicine and the rising volume of clinical trials focused on GI cancers have accelerated the adoption of these models. Continuous advancements in engraftment techniques and molecular profiling further enhance the utility of gastrointestinal PDX models, solidifying their leading position in the global market.
"The US is expected to grow at the highest CAGR during the forecast period"
The US is expected to register the highest growth rate in the patient-derived xenograft (PDX) model market during the forecast period. This growth is primarily driven by the strong presence of leading pharmaceutical and biotechnology companies, well-established research infrastructure, and extensive oncology research initiatives. The rising prevalence of cancer, coupled with increasing adoption of personalized medicine and targeted therapy development, is significantly boosting the demand for PDX models. Furthermore, growing investment in translational and preclinical cancer research, along with strong collaborations between academic institutions and industry players, is accelerating market expansion.
Additionally, the availability of advanced laboratory facilities, the presence of comprehensive biobanking systems, and supportive government funding for cancer drug discovery further strengthen the country's position in the global market. The US regulatory environment also promotes innovation while ensuring the reliability and ethical compliance of PDX-based studies. These factors collectively contribute to the rapid adoption of PDX models, reinforcing North America's leadership in the global market.
The primary interviews conducted for this report can be categorized as follows:
- By Respondent: Tier 1 - 25%, Tier 2 - 35%, and Tier 3 - 40%
- By Designation: Managers - 45%, CXOs and Directors - 30%, and Executives - 25%
- By Region: North America - 35%, Europe - 25%, Asia Pacific - 15%, Latin America - 10%, the Middle East - 10%, and Africa - 5%
JSR Corporation (Japan), WuXi AppTec (China), The Jackson Laboratory (US), Charles River Laboratories (US), Taconic Biosciences, Inc. (US), Oncodesign Precision Medicine (France), Inotiv (US), Pharmatest Services (Finland), Hera Biolabs (US), EPO Berlin-Buch GmbH (Germany), XenTech (France), Urosphere (France), Altogen Labs (US), Abnova Corporation (Taiwan), Genesis Biotechnology Group (US), Biocytogen Pharmaceuticals (Beijing) Co., Ltd. (China), Creative Animodel (US), BioDuro (US), Aragen Life Sciences (India), LIDE Shanghai Biotech, Ltd (China), Certis Oncology Solutions (US), InnoSer (Netherlands), IVRS AB (Sweden), Beijing IDMO Co., Ltd. (China), and Shanghai ChemPartner (China) are some of the key companies offering PDX models.
Research Coverage
This research report categorizes the PDX model market by implantation method (subcutaneous implantation, orthotopic implantation, others), type (mouse model, rat model), tumor type (gastrointestinal, gynecological, respiratory, urological, hematological, other tumor models), application (preclinical drug development, biomarker analysis, translational research, biobanks), end user (pharmaceutical & biotechnology companies, contract research organizations (CROs), academic & research institutes), and region (North America, Europe, Asia Pacific, Latin America, Middle East, And Africa).
The report's scope encompasses detailed information about the primary factors, including drivers, restraints, challenges, and opportunities, that influence the growth of the PDX model market. A comprehensive analysis of key industry players has been performed to provide insights into their business overview, product portfolio, key strategies, new product launches, acquisitions, and recent developments related to the PDX model market. This report also includes a competitive analysis of emerging startups in the PDX model industry ecosystem.
Key Benefits of Buying the Report
The report will assist market leaders and new entrants by providing revenue estimates for the overall market and its subsegments. It will also help stakeholders better understand the competitive landscape and gain more insights to position their businesses effectively and develop suitable go-to-market strategies. This report will enable stakeholders to grasp the market's pulse and offer information on key market drivers, restraints, opportunities, and challenges.
The report provides insights into the following pointers:
- Analysis of key drivers (growing demand for personalized medicine, technological advancements in PDX models, rising investments in cancer research), restraints (discontinuation of animal models for clinical trials by the FDA), opportunities (emergence of CRISPR in biomedical research), and challenges (development of alternative animal testing methods) influencing the market growth
- Product Development/Innovation: Detailed insights into newly launched products and technological assessment of the PDX model market
- Market Development: Comprehensive information about lucrative markets and analysis of the market across varied regions
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the PDX model market
- Competitive Assessment: In-depth assessment of market shares, growth strategies, and product offerings of leading players, including JSR Corporation (Japan), WuXi AppTec (China), The Jackson Laboratory (US), Charles River Laboratories (US), Taconic Biosciences, Inc. (US), among others offering products for the PDX model market. Other companies include BioDuro (US), Aragen Life Sciences (India), LIDE Shanghai Biotech, Ltd (China), Certis Oncology Solutions (US), InnoSer (Netherlands), among others, in the PDX model market
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.2.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.2.2 YEARS CONSIDERED
- 1.3 STUDY SCOPE
- 1.3.1 INCLUSIONS & EXCLUSIONS
- 1.3.2 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS & MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: INSIGHTS & STRATEGIC IMPERATIVES
- 2.3 DISRUPTIVE TRENDS SHAPING PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 2.4 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE AND COUNTRY (2024)
- 3.2 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- 3.3 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SHARE, BY IMPLANTATION, 2025 VS. 2030 (%)
- 3.4 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SHARE, BY SERVICE, 2024 (%)
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Growing demand for immuno-oncology and complex biologics
- 4.2.1.2 Rising preference for humanized PDX models
- 4.2.1.3 Focus on government-funded initiatives for cancer research
- 4.2.2 RESTRAINTS
- 4.2.2.1 Introduction of 3D printed models
- 4.2.2.2 Ethical concerns and regulatory restrictions
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Increasing preference for personalized medicines in oncology
- 4.2.3.2 Growing demand for CDX models for PDX model vendors
- 4.2.4 CHALLENGES
- 4.2.4.1 Limitations of humanized mouse models
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS & WHITE SPACES
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 BARGAINING POWER OF SUPPLIERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 ECOSYSTEM ANALYSIS
- 5.3 VALUE CHAIN ANALYSIS
- 5.4 PRICING ANALYSIS
- 5.4.1 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY KEY PLAYER, 2022-2024
- 5.4.2 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY IMPLANTATION METHOD, 2022-2024
- 5.4.3 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY TUMOR TYPE, 2022-2024
- 5.4.4 AVERAGE SELLING PRICE OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY REGION, 2024
- 5.5 TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS
- 5.6 PATENT ANALYSIS
- 5.6.1 METHODOLOGY
- 5.6.2 NUMBER OF PATENTS FILED, BY DOCUMENT TYPE
- 5.6.3 LIST OF KEY PATENTS
- 5.7 TRADE DATA ANALYSIS
- 5.7.1 IMPORT DATA FOR HS CODE 106.19.90, 2020-2024
- 5.7.2 EXPORT DATA FOR HS CODE 106.19.90, 2020-2024
- 5.8 IMPACT OF 2025 US TARIFF ON PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 5.8.1 KEY TARIFF RATES
- 5.8.2 PRICE IMPACT ANALYSIS
- 5.8.3 KEY IMPACT ON VARIOUS REGIONS
- 5.8.3.1 North America
- 5.8.3.2 Europe
- 5.8.3.3 Asia Pacific
- 5.8.3.4 Rest of the World
- 5.8.4 IMPACT ON END-USE INDUSTRIES
- 5.8.4.1 Pharmaceutical & biotechnology companies
- 5.8.4.2 CROs & CDMOs
- 5.8.4.3 Academic & research institutes
- 5.9 MACROECONOMIC OUTLOOK
- 5.9.1 INTRODUCTION
- 5.9.2 GDP TRENDS AND FORECAST
- 5.9.3 R&D TRENDS IN GLOBAL HEALTHCARE INDUSTRY
- 5.9.4 R&D TRENDS IN GLOBAL PHARMA INDUSTRY
- 5.10 KEY CONFERENCES & EVENTS, 2025-2026
- 5.11 INVESTMENT & FUNDING SCENARIO
- 5.11.1 VC/PRIVATE EQUITY INVESTMENT TRENDS & STARTUP LANDSCAPE
- 5.12 CASE STUDY ANALYSIS
- 5.12.1 IMAGING-READY ORTHOTOPIC PDX FOR PEDIATRIC BRAIN TUMORS
- 5.12.2 HOST-STRAIN SELECTION USING BREAST CANCER PDX
- 5.12.3 RESISTANCE-ANCHORED PDX PANEL TO DESIGN MOUSE CLINICAL TRIAL
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 KEY EMERGING TECHNOLOGIES
- 6.1.1 NEXT-GENERATION SEQUENCING
- 6.1.2 GENE EDITING
- 6.2 COMPLEMENTARY TECHNOLOGIES
- 6.2.1 MULTI-OMICS
- 6.2.2 MULTI-MODEL PRECLINICAL IMAGING
- 6.3 TECHNOLOGY ROADMAP
- 6.4 FUTURE APPLICATIONS
- 6.5 IMPACT OF AI/GEN AI ON PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 6.5.1 TOP USE CASES & MARKET POTENTIAL
- 6.5.2 CASE STUDIES OF AI IMPLEMENTATION
- 6.5.3 BEST PRACTICES IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS CREATION
- 6.5.4 CLIENTS' READINESS TO ADOPT GENERATIVE AI IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 6.5.5 HIGH READINESS VS. ADOPTION BARRIERS
7 SUSTAINABILITY AND REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS & COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 INDUSTRY STANDARDS
- 7.1.2.1 North America
- 7.1.2.2 Europe
- 7.1.2.3 Asia Pacific
- 7.1.2.4 Latin America
- 7.2 SUSTAINABILITY IMPACT & REGULATORY POLICY INITIATIVES
- 7.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS & BUYING EVALUATION CRITERIA
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM END-USE INDUSTRIES
9 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE
- 9.1 INTRODUCTION
- 9.2 PDX MODELS
- 9.2.1 MOUSE MODEL
- 9.2.1.1 Proven efficacy of mouse model in cancer research and drug development to boost segment growth
- 9.2.2 RAT MODEL
- 9.2.2.1 Emergence of advanced technologies in PDX rat models to aid market growth
- 9.2.1 MOUSE MODEL
- 9.3 SERVICES
- 9.3.1 MODEL CREATION & EXPANISON SERVICES
- 9.3.1.1 Need for faster passaging of models to to build study-ready cohorts (P1-P3) to boost segment growth
- 9.3.2 MODEL CHARACTERIZATION SERVICES
- 9.3.2.1 Growing demand for biomarker analysis to drive segment growth
- 9.3.3 CRYOPRESERVATION SERVICES
- 9.3.3.1 Reliable and low-passage biobanking services to offer growth opportunities
- 9.3.4 OTHER PDX SERVICES
- 9.3.1 MODEL CREATION & EXPANISON SERVICES
10 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD
- 10.1 INTRODUCTION
- 10.2 SUBCUTANEOUS IMPLANTATION
- 10.2.1 STANDARDIZED AND SCALABLE PDX MODEL EXPANSION TO DRIVE SEGMENT GROWTH
- 10.3 ORTHOTOPIC IMPLANTATION
- 10.3.1 DEMAND FOR CLINICALLY RELEVANT AND SITE-SPECIFIC PDX MODELS TO BOOST SEGMENT GROWTH
- 10.4 OTHER IMPLANTATION METHODS
11 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE
- 11.1 INTRODUCTION
- 11.2 GASTROINTESTINAL TUMOR MODEL
- 11.2.1 HIGH TISSUE AVAILABILITY AND STANDARDIZED ENGRAFTMENT PROTOCOLS TO FUEL MARKET GROWTH
- 11.3 GYNECOLOGICAL TUMOR MODEL
- 11.3.1 EXPANDING ORTHOTOPIC AND HUMANIZED PDX APPLICATIONS TO DRIVE MARKET
- 11.4 RESPIRATORY TUMOR MODEL
- 11.4.1 INCREASING ADOPTION OF ORTHOTOPIC AND HUMANIZED MODELS TO BOOST SEGMENT GROWTH
- 11.5 UROLOGICAL TUMOR MODEL
- 11.5.1 EXPANSION OF HORMONE-CONTEXT AND IMMUNE-COMPETENT MODELS TO FAVOR MARKET ADOPTION
- 11.6 HEMATOLOGICAL TUMOR MODEL
- 11.6.1 RISING ADOPTION OF HUMANIZED AND RELAPSE-DERIVED MODELS TO DRIVE SEGMENT GROWTH
- 11.7 OTHER TUMOR MODELS
12 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION
- 12.1 INTRODUCTION
- 12.2 PRECLINICAL RESEARCH
- 12.2.1 HIGH IMPORTANCE FOR PDX IN EFFICACY CONFIRMATION TO DRIVE MARKET
- 12.3 BIOMARKER ANALYSIS
- 12.3.1 DEMAND FOR MULTI-OMICS TO AUGMENT MARKET GROWTH
- 12.4 TRANSLATIONAL RESEARCH
- 12.4.1 DEMAND FOR PDX MODELS IN CO-CLINICAL TRIAL DESIGNS TO BOOST SEGMENT GROWTH
- 12.5 BIOBANKING
- 12.5.1 DEMAND FOR PDX MODELS IN SPECIALIZED BANKS TO SPUR MARKET GROWTH
13 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER
- 13.1 INTRODUCTION
- 13.2 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
- 13.2.1 INTEGRATION OF TRANSLATIONALLY RELEVANT PDX WORKFLOWS TO DRIVE MARKET GROWTH
- 13.3 CROS & CDMOS
- 13.3.1 EXPANDED OUTSOURCING THROUGH INTEGRATED WORKFLOWS TO FUEL MARKET GROWTH
- 13.4 ACADEMIC & RESEARCH INSTITUTES
- 13.4.1 ENHANCED INSTITUTIONAL BIOBANKS AND MULTI-OMIC PROFILING TO AUGMENT MARKET GROWTH
14 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY REGION
- 14.1 INTRODUCTION
- 14.2 NORTH AMERICA
- 14.2.1 US
- 14.2.1.1 US to dominate North American PDX models market during study period
- 14.2.2 CANADA
- 14.2.2.1 Growth research and academic activities on oncology to fuel market growth
- 14.2.1 US
- 14.3 EUROPE
- 14.3.1 GERMANY
- 14.3.1.1 Growth in biotechnology industry and strong pharma presence to spur market growth
- 14.3.2 UK
- 14.3.2.1 Increasing focus on cancer research to propel market
- 14.3.3 FRANCE
- 14.3.3.1 Robust research activities in immune-oncology to drive market growth
- 14.3.4 ITALY
- 14.3.4.1 Growing pharmaceutical & biotechnology industry and increasing focus on radiopharmaceuticals to favor market growth
- 14.3.5 SPAIN
- 14.3.5.1 Increasing translational activities to boost market growth
- 14.3.6 REST OF EUROPE
- 14.3.1 GERMANY
- 14.4 ASIA PACIFIC
- 14.4.1 CHINA
- 14.4.1.1 Favorable investments in life sciences research to drive market
- 14.4.2 JAPAN
- 14.4.2.1 Increasing focus on personalized diagnostics products to drive market
- 14.4.3 INDIA
- 14.4.3.1 Favorable initiatives for drug discovery & development to boost market growth
- 14.4.4 AUSTRALIA
- 14.4.4.1 High number of research institutes to favor market growth
- 14.4.5 SOUTH KOREA
- 14.4.5.1 Innovation in health and biotech sectors to support market uptake
- 14.4.6 REST OF ASIA PACIFIC
- 14.4.1 CHINA
- 14.5 LATIN AMERICA
- 14.5.1 BRAZIL
- 14.5.1.1 Increasing investments in biopharmaceutical research to fuel uptake
- 14.5.2 MEXICO
- 14.5.2.1 Expanding healthcare ecosystem to support market growth
- 14.5.3 REST OF LATIN AMERICA
- 14.5.1 BRAZIL
- 14.6 MIDDLE EAST
- 14.6.1 GCC COUNTRIES
- 14.6.1.1 Kingdom of Saudi Arabia
- 14.6.1.1.1 Advanced healthcare sector and prevalence of chronic diseases to boost market
- 14.6.1.2 UAE
- 14.6.1.2.1 Increasing R&D activities and growing demand for personalized medicines to propel market
- 14.6.1.3 Rest of GCC Countries
- 14.6.1.1 Kingdom of Saudi Arabia
- 14.6.2 REST OF MIDDLE EAST
- 14.6.1 GCC COUNTRIES
- 14.7 AFRICA
- 14.7.1 INCREASING FOCUS ON PRECISION MEDICINE INITIATIVES TO SUPPORT MARKET GROWTH
15 COMPETITIVE LANDSCAPE
- 15.1 INTRODUCTION
- 15.2 KEY PLAYERS STRATEGIES/RIGHT TO WIN
- 15.2.1 STRATEGIES ADOPTED BY KEY PLAYERS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, 2022-2025
- 15.3 REVENUE ANALYSIS, 2022-2024
- 15.4 MARKET SHARE ANALYSIS, 2024
- 15.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2024
- 15.5.1 STARS
- 15.5.2 EMERGING LEADERS
- 15.5.3 PERVASIVE PLAYERS
- 15.5.4 PARTICIPANTS
- 15.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2024
- 15.5.5.1 Company footprint
- 15.5.5.2 Region footprint
- 15.5.5.3 Model type & service footprint
- 15.5.5.4 Tumor type footprint
- 15.5.5.5 Implantation method footprint
- 15.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2024
- 15.6.1 PROGRESSIVE COMPANIES
- 15.6.2 RESPONSIVE COMPANIES
- 15.6.3 DYNAMIC COMPANIES
- 15.6.4 STARTING BLOCKS
- 15.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SME PLAYERS, 2024
- 15.6.5.1 Detailed list of key startups/SMEs
- 15.6.5.2 Competitive benchmarking of key startups/SMEs
- 15.7 COMPANY VALUATION & FINANCIAL METRICS
- 15.7.1 FINANCIAL METRICS
- 15.7.2 COMPANY VALUATION
- 15.8 BRAND/PRODUCT COMPARISON
- 15.9 COMPETITIVE SCENARIO
- 15.9.1 DEALS
- 15.9.2 EXPANSIONS
16 COMPANY PROFILES
- 16.1 KEY PLAYERS
- 16.1.1 CHARLES RIVER LABORATORIES
- 16.1.1.1 Business overview
- 16.1.1.2 Products/Services offered
- 16.1.1.3 Recent developments
- 16.1.1.3.1 Deals
- 16.1.1.3.2 Expansions
- 16.1.1.4 MnM view
- 16.1.1.4.1 Key strengths
- 16.1.1.4.2 Strategic choices
- 16.1.1.4.3 Weaknesses & competitive threats
- 16.1.2 THE JACKSON LABORATORY
- 16.1.2.1 Business overview
- 16.1.2.2 Products/Services offered
- 16.1.2.3 Recent developments
- 16.1.2.3.1 Deals
- 16.1.2.3.2 Expansions
- 16.1.2.4 MnM view
- 16.1.2.4.1 Key strengths
- 16.1.2.4.2 Strategic choices
- 16.1.2.4.3 Weaknesses & competitive threats
- 16.1.3 INOTIV
- 16.1.3.1 Business overview
- 16.1.3.2 Products/Services offered
- 16.1.3.3 Recent developments
- 16.1.3.3.1 Expansions
- 16.1.3.4 MnM view
- 16.1.3.4.1 Key strengths
- 16.1.3.4.2 Strategic choices
- 16.1.3.4.3 Weaknesses & competitive threats
- 16.1.4 JSR CORPORATION
- 16.1.4.1 Business overview
- 16.1.4.2 Product/Services offered
- 16.1.4.3 Recent developments
- 16.1.4.3.1 Deals
- 16.1.4.3.2 Expansions
- 16.1.4.4 MnM view
- 16.1.4.4.1 Key strengths
- 16.1.4.4.2 Strategic choices
- 16.1.4.4.3 Weaknesses & competitive threats
- 16.1.5 GENOWAY
- 16.1.5.1 Business overview
- 16.1.5.2 Product/Solutions offered
- 16.1.5.3 MnM view
- 16.1.5.3.1 Key strengths
- 16.1.5.3.2 Strategic choices
- 16.1.5.3.3 Weaknesses & competitive threats
- 16.1.6 WUXI APPTEC + WUXI BIOLOGICS
- 16.1.6.1 Business overview
- 16.1.6.2 Product/Services offered
- 16.1.6.3 Recent developments
- 16.1.6.3.1 Deals
- 16.1.6.3.2 Expansions
- 16.1.7 BIOCYTOGEN PHARMACEUTICALS CO., LTD.
- 16.1.7.1 Business overview
- 16.1.7.2 Product/Services offered
- 16.1.7.3 Recent developments
- 16.1.7.3.1 Product/Service upgrades
- 16.1.7.3.2 Expansions
- 16.1.8 TACONIC BIOSCIENCES, INC.
- 16.1.8.1 Business overview
- 16.1.8.2 Products/Services offered
- 16.1.8.3 Recent developments
- 16.1.8.3.1 Product/Service launches
- 16.1.8.3.2 Deals
- 16.1.8.3.3 Expansions
- 16.1.9 ONCODESIGN SERVICES (ONCODESIGN PRECISION MEDICINE)
- 16.1.9.1 Business overview
- 16.1.9.2 Products/Services offered
- 16.1.10 CHAMPIONS ONCOLOGY, INC.
- 16.1.10.1 Business overview
- 16.1.10.2 Products/Services offered
- 16.1.10.3 Recent developments
- 16.1.10.3.1 Deals
- 16.1.11 PHARMARON
- 16.1.11.1 Business overview
- 16.1.11.2 Products/Services offered
- 16.1.12 SYNGENE INTERNATIONAL LIMITED
- 16.1.12.1 Business overview
- 16.1.12.2 Products/Services offered
- 16.1.13 LABCORP
- 16.1.13.1 Business overview
- 16.1.13.2 Products/Services offered
- 16.1.14 EUROFINS SCIENTIFIC
- 16.1.14.1 Business overview
- 16.1.14.2 Products/Services offered
- 16.1.15 VITALSTAR BIOTECHNOLOGY CO., LTD.
- 16.1.15.1 Business overview
- 16.1.15.2 Products/Services offered
- 16.1.1 CHARLES RIVER LABORATORIES
- 16.2 OTHER PLAYERS
- 16.2.1 CREATIVE ANIMODEL
- 16.2.2 JOINN LABORATORIES (CHINA) CO., LTD.
- 16.2.3 CREATIVE BIOLABS
- 16.2.4 ARAGEN LIFE SCIENCES LTD.
- 16.2.5 PHARMTEST SERVICES
- 16.2.6 LIDE SHANGHAI BIOTECH CO., LTD.
- 16.2.7 CERTIS ONCOLOGY SOLUTIONS
- 16.2.8 INNOSER
- 16.2.9 SHANGHAI CHEMPARTNER
- 16.2.10 IVRS AB
17 RESEARCH METHODOLOGY
- 17.1 RESEARCH DATA
- 17.1.1 SECONDARY DATA
- 17.1.2 KEY OBJECTIVES OF SECONDARY RESEARCH
- 17.1.3 PRIMARY DATA
- 17.2 MARKET ESTIMATION METHODOLOGY
- 17.2.1 COMPANY REVENUE ANALYSIS: BOTTOM-UP APPROACH
- 17.2.2 INSIGHTS FROM PRIMARY EXPERTS
- 17.2.3 TOP-DOWN APPROACH
- 17.3 GROWTH FORECAST
- 17.4 DATA TRIANGULATION
- 17.5 STUDY ASSUMPTIONS
- 17.6 RESEARCH LIMITATIONS
- 17.7 MARKET FORECAST
- 17.8 RISK ANALYSIS
18 APPENDIX
- 18.1 DISCUSSION GUIDE
- 18.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 18.3 CUSTOMIZATION OPTIONS
- 18.4 RELATED REPORTS
- 18.5 AUTHOR DETAILS
List of Tables
- TABLE 1 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: INCLUSIONS & EXCLUSIONS
- TABLE 2 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: IMPACT OF PORTER'S FIVE FORCES
- TABLE 3 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODEL TYPES, BY KEY PLAYER, 2022-2024
- TABLE 4 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY IMPLANTATION METHOD, 2022-2024
- TABLE 5 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY TUMOR TYPE, 2022-2024
- TABLE 6 AVERAGE SELLING PRICE OF PATIENT-DERIVED XENOGRAFT (PDX) MODEL TYPES, BY REGION, 2024
- TABLE 7 NUMBER OF PATENTS FILED, BY DOCUMENT TYPE, 2014-2024
- TABLE 8 LIST OF KEY PATENTS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, 2023-2025
- TABLE 9 IMPORT DATA FOR HS CODE 106.19.90, 2020-2024 (USD THOUSAND)
- TABLE 10 EXPORT DATA FOR HS CODE 106.19.90, 2020-2024 (USD THOUSAND)
- TABLE 11 US-AJUSTED RECIPROCAL TARIFF RATES
- TABLE 12 KEY PRODUCT-RELATED TARIFF: LIVE ANIMALS, PRODUCT OF ANIMAL ORIGIN, MISCELLANEOUS CHEMICAL PRODUCTS
- TABLE 13 CRITICAL COMPONENTS EXPOSED TO TARIFF CHANGES
- TABLE 14 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: KEY CONFERENCES & EVENTS, JANUARY 2025-DECEMBER 2026
- TABLE 15 INVESTMENT & FUNDING ACTIVITY IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY KEY PLAYER, 2022-2025
- TABLE 16 CASE STUDIES OF AI IMPLEMENTATION IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- TABLE 17 HIGH READINESS VS. ADOPTION BARRIERS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- TABLE 18 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 19 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 20 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 21 REST OF THE WORLD: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 22 VENDOR CERTIFICATIONS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- TABLE 23 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 24 PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 25 PDX MODELS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 26 NORTH AMERICA: PDX MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 27 EUROPE: PDX MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 28 ASIA PACIFIC: PDX MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 29 LATIN AMERICA: PDX MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 30 MIDDLE EAST: PDX MODELS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 31 GCC COUNTRIES: PDX MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 32 MOUSE MODEL MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 33 NORTH AMERICA: MOUSE MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 34 EUROPE: MOUSE MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 35 ASIA PACIFIC: MOUSE MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 36 LATIN AMERICA: MOUSE MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 37 MIDDLE EAST: MOUSE MODEL MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 38 GCC COUNTRIES: MOUSE MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 39 RAT MODEL MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 40 NORTH AMERICA: RAT MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 41 EUROPE: RAT MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 42 ASIA PACIFIC: RAT MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 43 LATIN AMERICA: RAT MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 44 MIDDLE EAST: RAT MODEL MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 45 GCC COUNTRIES: RAT MODEL MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 46 PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 47 PDX SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 48 NORTH AMERICA: PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 49 EUROPE: PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 50 ASIA PACIFIC: PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 51 LATIN AMERICA: PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 52 MIDDLE EAST: PDX SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 53 GCC COUNTRIES: PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 54 MODEL CREATION & EXPANSION SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 55 NORTH AMERICA: MODEL CREATION & EXPANSION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 56 EUROPE: MODEL CREATION & EXPANSION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 57 ASIA PACIFIC: MODEL CREATION & EXPANSION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 58 LATIN AMERICA: MODEL CREATION & EXPANSION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 59 MIDDLE EAST: MODEL CREATION & EXPANSION SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 60 GCC COUNTRIES: MODEL CREATION & EXPANSION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 61 MODEL CHARATERIZATION SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 62 NORTH AMERICA: MODEL CHARATERIZATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 63 EUROPE: MODEL CHARATERIZATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 64 ASIA PACIFIC: MODEL CHARATERIZATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 65 LATIN AMERICA: MODEL CHARATERIZATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 66 MIDDLE EAST: MODEL CHARATERIZATION SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 67 GCC COUNTRIES: MODEL CHARATERIZATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 68 CRYOPRESERVATION SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 69 NORTH AMERICA: CRYOPRESERVATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 70 EUROPE: CRYOPRESERVATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 71 ASIA PACIFIC: CRYOPRESERVATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 72 LATIN AMERICA: CRYOPRESERVATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 73 MIDDLE EAST: CRYOPRESERVATION SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 74 GCC COUNTRIES: CRYOPRESERVATION SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 75 OTHER PDX SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 76 NORTH AMERICA: OTHER PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 77 EUROPE: OTHER PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 78 ASIA PACIFIC: OTHER PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 79 LATIN AMERICA: OTHER PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 80 MIDDLE EAST: OTHER PDX SERVICES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 81 GCC COUNTRIES: OTHER PDX SERVICES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 82 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 83 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 84 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 85 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 86 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 87 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 88 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 89 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR SUBCUTANEOUS IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 90 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 91 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 92 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 93 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 94 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 95 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 96 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ORTHOTOPIC IMPLANTATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 97 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 98 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 99 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 100 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 101 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 102 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 103 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER IMPLANTATION METHODS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 104 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 105 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 106 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 107 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 108 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 109 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 110 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 111 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GASTROINTESTINAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 112 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 113 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 114 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 115 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 116 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 117 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 118 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR GYNECOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 119 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 120 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 121 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 122 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 123 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 124 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 125 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR RESPIRATORY TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 126 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 127 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 128 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 129 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 130 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 131 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 132 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR UROLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 133 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 134 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 135 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 136 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 137 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 138 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY REGION, 2023-2030 (USD MILLION)
- TABLE 139 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR HEMATOLOGICAL TUMOR MODEL, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 140 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 141 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 142 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 143 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 144 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 145 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 146 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR OTHER TUMOR MODELS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 147 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 148 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY REGION, 2023-2030 (USD MILLION)
- TABLE 149 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 150 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 151 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 152 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 153 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY REGION, 2023-2030 (USD MILLION)
- TABLE 154 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PRECLINICAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 155 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 156 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 157 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 158 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 159 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 160 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 161 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOMARKER ANALYSIS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 162 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY REGION, 2023-2030 (USD MILLION)
- TABLE 163 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 164 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 165 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 166 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 167 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY REGION, 2023-2030 (USD MILLION)
- TABLE 168 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR TRANSLATIONAL RESEARCH, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 169 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY REGION, 2023-2030 (USD MILLION)
- TABLE 170 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 171 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 172 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 173 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 174 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY REGION, 2023-2030 (USD MILLION)
- TABLE 175 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR BIOBANKING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 176 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 177 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY REGION, 2023-2030 (USD MILLION)
- TABLE 178 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 179 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 180 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 181 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 182 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY REGION, 2023-2030 (USD MILLION)
- TABLE 183 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 184 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 185 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 186 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 187 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 188 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 189 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 190 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR CROS & CDMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 191 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY REGION, 2023-2030 (USD MILLION)
- TABLE 192 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 193 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 194 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 195 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 196 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY REGION, 2023-2030 (USD MILLION)
- TABLE 197 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 198 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 199 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 200 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 201 NORTH AMERICA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 202 NORTH AMERICA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 203 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 204 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 205 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 206 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 207 US: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 208 US: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 209 US: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 210 US: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 211 US: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 212 US: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 213 US: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 214 CANADA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 215 CANADA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 216 CANADA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 217 CANADA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 218 CANADA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 219 CANADA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 220 CANADA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 221 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 222 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 223 EUROPE: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 224 EUROPE: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 225 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 226 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 227 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 228 EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 229 GERMANY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 230 GERMANY: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 231 GERMANY: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 232 GERMANY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 233 GERMANY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 234 GERMANY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 235 GERMANY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 236 UK: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 237 UK: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 238 UK: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 239 UK: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 240 UK: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 241 UK: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 242 UK: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 243 FRANCE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 244 FRANCE: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 245 FRANCE: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 246 FRANCE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 247 FRANCE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 248 FRANCE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 249 FRANCE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 250 ITALY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 251 ITALY: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 252 ITALY: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 253 ITALY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 254 ITALY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 255 ITALY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 256 ITALY: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 257 SPAIN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 258 SPAIN: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 259 SPAIN: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 260 SPAIN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 261 SPAIN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 262 SPAIN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 263 SPAIN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 264 REST OF EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 265 REST OF EUROPE: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 266 REST OF EUROPE: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 267 REST OF EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 268 REST OF EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 269 REST OF EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 270 REST OF EUROPE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 271 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 272 ASIA PACIFIC: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 273 ASIA PACIFIC: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 274 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 275 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 276 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 277 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 278 CHINA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 279 CHINA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 280 CHINA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 281 CHINA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 282 CHINA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 283 CHINA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 284 CHINA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 285 JAPAN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 286 JAPAN: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 287 JAPAN: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 288 JAPAN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 289 JAPAN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 290 JAPAN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 291 JAPAN: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 292 INDIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 293 INDIA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 294 INDIA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 295 INDIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 296 INDIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 297 INDIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 298 INDIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 299 AUSTRALIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 300 AUSTRALIA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 301 AUSTRALIA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 302 AUSTRALIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 303 AUSTRALIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 304 AUSTRALIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 305 AUSTRALIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 306 SOUTH KOREA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 307 SOUTH KOREA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 308 SOUTH KOREA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 309 SOUTH KOREA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 310 SOUTH KOREA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 311 SOUTH KOREA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 312 SOUTH KOREA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 313 REST OF ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 314 REST OF ASIA PACIFIC: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 315 REST OF ASIA PACIFIC: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 316 REST OF ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 317 REST OF ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 318 REST OF ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 319 REST OF ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 320 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 321 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 322 LATIN AMERICA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 323 LATIN AMERICA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 324 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 325 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 326 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 327 LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 328 BRAZIL: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 329 BRAZIL: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 330 BRAZIL: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 331 BRAZIL: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 332 BRAZIL: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 333 BRAZIL: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 334 BRAZIL: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 335 MEXICO: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 336 MEXICO: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 337 MEXICO: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 338 MEXICO: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 339 MEXICO: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 340 MEXICO: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 341 MEXICO: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 342 REST OF LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 343 REST OF LATIN AMERICA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 344 REST OF LATIN AMERICA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 345 REST OF LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 346 REST OF LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 347 REST OF LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 348 REST OF LATIN AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 349 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 350 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 351 MIDDLE EAST: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 352 MIDDLE EAST: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 353 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 354 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 355 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 356 MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 357 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 358 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 359 GCC COUNTRIES: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 360 GCC COUNTRIES: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 361 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 362 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 363 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 364 GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 365 KINGDOM OF SAUDI ARABIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 366 KINGDOM OF SAUDI ARABIA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 367 KINGDOM OF SAUDI ARABIA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 368 KINGDOM OF SAUDI ARABIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 369 KINGDOM OF SAUDI ARABIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 370 KINGDOM OF SAUDI ARABIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 371 KINGDOM OF SAUDI ARABIA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 372 UAE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 373 UAE: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 374 UAE: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 375 UAE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 376 UAE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 377 UAE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 378 UAE: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 379 REST OF GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 380 REST OF GCC COUNTRIES: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 381 REST OF GCC COUNTRIES: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 382 REST OF GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 383 REST OF GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 384 REST OF GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 385 REST OF GCC COUNTRIES: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 386 REST OF MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 387 REST OF MIDDLE EAST: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 388 REST OF MIDDLE EAST: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 389 REST OF MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 390 REST OF MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 391 REST OF MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 392 REST OF MIDDLE EAST: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 393 AFRICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2023-2030 (USD MILLION)
- TABLE 394 AFRICA: PDX MODELS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 395 AFRICA: PDX SERVICES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 396 AFRICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2023-2030 (USD MILLION)
- TABLE 397 AFRICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2023-2030 (USD MILLION)
- TABLE 398 AFRICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 399 AFRICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 400 STRATEGIES ADOPTED BY KEY PLAYERS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, 2022-2025
- TABLE 401 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: DEGREE OF COMPETITION
- TABLE 402 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: REGION FOOTPRINT
- TABLE 403 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: MODEL TYPE & SERVICE FOOTPRINT
- TABLE 404 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: TUMOR TYPE FOOTPRINT
- TABLE 405 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: IMPLANTATION METHOD FOOTPRINT
- TABLE 406 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: DETAILED LIST OF KEY STARTUPS/SME PLAYERS
- TABLE 407 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: COMPETITIVE BENCHMARKING OF KEY STARTUPS/SME PLAYERS, BY MODEL TYPE & SERVICE AND REGION
- TABLE 408 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: DEALS, JANUARY 2022- OCTOBER 2025
- TABLE 409 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: EXPANSIONS, JANUARY 2022- OCTOBER 2025
- TABLE 410 CHARLES RIVER LABORATORIES: COMPANY OVERVIEW
- TABLE 411 CHARLES RIVER LABORATORIES: PRODUCTS/SERVICES OFFERED
- TABLE 412 CHARLES RIVER LABORATORIES: DEALS, JANUARY 2022-OCTOBER 2025
- TABLE 413 CHARLES RIVER LABORATORIES: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 414 THE JACKSON LABORATORY: COMPANY OVERVIEW
- TABLE 415 THE JACKSON LABORATORY: PRODUCTS/SERVICES OFFERED
- TABLE 416 THE JACKSON LABORATORY: DEALS, JANUARY 2022-OCTOBER 2025
- TABLE 417 THE JACKSON LABORATORY: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 418 INOTIV: COMPANY OVERVIEW
- TABLE 419 INOTIV: PRODUCTS/SERVICES OFFERED
- TABLE 420 INOTIV: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 421 JSR CORPORATION: COMPANY OVERVIEW
- TABLE 422 JSR CORPORATION: PRODUCTS/SERVICES OFFERED
- TABLE 423 JSR CORPORATION: DEALS, JANUARY 2022-OCTOBER 2025
- TABLE 424 JSR CORPORATION: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 425 GENOWAY: COMPANY OVERVIEW
- TABLE 426 GENOWAY: PRODUCTS/SERVICES OFFERED
- TABLE 427 WUXI APPTEC + WUXI BIOLOGICS: COMPANY OVERVIEW
- TABLE 428 WUXI APPTEC + WUXI BIOLOGICS: PRODUCTS/SERVICES OFFERED
- TABLE 429 WUXI APPTEC + WUXI BIOLOGICS: DEALS, JANUARY 2022-OCTOBER 2025
- TABLE 430 WUXI APPTEC + WUXI BIOLOGICS: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 431 BIOCYTOGEN PHARMACEUTICALS CO., LTD.: COMPANY OVERVIEW
- TABLE 432 BIOCYTOGEN PHARMACEUTICALS CO., LTD.: PRODUCTS/SERVICES OFFERED
- TABLE 433 BIOCYTOGEN PHARMACEUTICALS CO., LTD.: PRODUCT/SERVICE UPGRADES, JANUARY 2022-OCTOBER 2025
- TABLE 434 BIOCYTOGEN PHARMACEUTICALS CO., LTD.: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 435 TACONIC BIOSCIENCES, INC.: COMPANY OVERVIEW
- TABLE 436 TACONIC BIOSCIENCES, INC.: PRODUCTS/SERVICES OFFERED
- TABLE 437 TACONIC BIOSCIENCES, INC.: PRODUCT/SERVICE LAUNCHES, JANUARY 2022-OCTOBER 2025
- TABLE 438 TACONIC BIOSCIENCES, INC.: DEALS, JANUARY 2022-OCTOBER 2025
- TABLE 439 TACONIC BIOSCIENCES, INC.: EXPANSIONS, JANUARY 2022-OCTOBER 2025
- TABLE 440 ONCODESIGN SERVICES (ONCODESIGN PRECISION MEDICINE): COMPANY OVERVIEW
- TABLE 441 ONCODESIGN SERVICES (ONCODESIGN PRECISION MEDICINE): PRODUCTS/SERVICES OFFERED
- TABLE 442 CHAMPIONS ONCOLOGY, INC.: COMPANY OVERVIEW
- TABLE 443 CHAMPIONS ONCOLOGY, INC.: PRODUCTS/SERVICES OFFERED
- TABLE 444 CHAMPIONS ONCOLOGY, INC.: DEALS, JANUARY 2022-OCTOBER 2025
- TABLE 445 PHARMARON: COMPANY OVERVIEW
- TABLE 446 PHARMARON: PRODUCTS/SERVICES OFFERED
- TABLE 447 SYNGENE INTERNATIONAL LIMITED: COMPANY OVERVIEW
- TABLE 448 SYNGENE INTERNATIONAL LIMITED: PRODUCTS/SERVICES OFFERED
- TABLE 449 LABCORP: COMPANY OVERVIEW
- TABLE 450 LABCORP: PRODUCTS/SERVICES OFFERED
- TABLE 451 EUROFINS SCIENTIFIC: COMPANY OVERVIEW
- TABLE 452 EUROFINS SCIENTIFIC: PRODUCTS/SERVICES OFFERED
- TABLE 453 VITALSTAR BIOTECHNOLOGY CO., LTD.: COMPANY OVERVIEW
- TABLE 454 VITALSTAR BIOTECHNOLOGY CO., LTD.: PRODUCTS/SERVICES OFFERED
- TABLE 455 CREATIVE ANIMODEL: COMPANY OVERVIEW
- TABLE 456 JOINN LABORATORIES (CHINA) CO., LTD.: COMPANY OVERVIEW
- TABLE 457 CREATIVE BIOLABS: COMPANY OVERVIEW
- TABLE 458 ARAGEN LIFE SCIENCES LTD.: BUSINESS OVERVIEW
- TABLE 459 PHARMATEST: BUSINESS OVERVIEW
- TABLE 460 LIDE SHANGHAI BIOTECH CO., LTD.: COMPANY OVERVIEW
- TABLE 461 CERTIS ONCOLOGY SOLUTIONS: COMPANY OVERVIEW
- TABLE 462 INNOSER: COMPANY OVERVIEW
- TABLE 463 SHANGHAI CHEMPARTNER: COMPANY OVERVIEW
- TABLE 464 IVRS AB: COMPANY OVERVIEW
- TABLE 465 PATIENT-DERIVED XENOGRAFTS (PDX) MODELS MARKET: IMPACT ANALYSIS
- TABLE 466 PATIENT-DERIVED XENOGRAFTS (PDX) MODELS MARKET: RISK ANALYSIS
List of Figures
- FIGURE 1 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SEGMENTATION & REGIONAL SCOPE
- FIGURE 2 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: YEARS CONSIDERED
- FIGURE 3 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE, 2025 VS. 2030 (USD MILLION)
- FIGURE 4 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD, 2025 VS. 2030 (USD MILLION)
- FIGURE 5 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE, 2025 VS. 2030 (USD MILLION)
- FIGURE 6 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION, 2025 VS. 2030 (USD MILLION)
- FIGURE 7 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER, 2025 VS. 2030 (USD MILLION)
- FIGURE 8 GROWING DEMAND FOR IMMUNO-ONCOLOGY AND COMPLEX BIOLOGICS TO DRIVE MARKET
- FIGURE 9 US AND PDX MODELS COMMANDED LARGEST MARKET SHARE IN 2024
- FIGURE 10 CHINA TO REGISTER HIGHEST GROWTH RATE DURING FORECAST PERIOD
- FIGURE 11 SUBCUTANEOUS IMPLANTATION TO DOMINATE MARKET DURING FORECAST PERIOD
- FIGURE 12 MODEL CREATION & EXPANSION ACCOUNTED FOR LARGEST MARKET MARKET SHARE IN 2024
- FIGURE 13 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 14 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: PORTER'S FIVE FORCES
- FIGURE 15 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: ECOSYSTEM ANALYSIS
- FIGURE 16 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: VALUE CHAIN ANALYSIS
- FIGURE 17 AVERAGE SELLING PRICE TREND OF MODEL TYPES, BY KEY PLAYER, 2022-2024 (USD)
- FIGURE 18 AVERAGE SELLING PRICE OF PATIENT-DERIVED XENOGRAFT (PDX) MODEL TYPES, BY REGION, 2024 (USD)
- FIGURE 19 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS
- FIGURE 20 PATENT APPLICATIONS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, JANUARY 2014-DECEMBER 2024
- FIGURE 21 NUMBER OF DEALS AND FUNDING IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, 2022-2025 (USD MILLION)
- FIGURE 22 TOP USE CASES IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- FIGURE 23 INFLUENCE OF KEY STAKEHOLDERS ON DECISION-MAKING FOR PATIENT-DERIVED XENOGRAFT (PDX) MODELS
- FIGURE 24 BUYING EVALUATION CRITERIA FOR PATIENT-DERIVED XENOGRAFT (PDX) MODELS
- FIGURE 25 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SNAPSHOT
- FIGURE 26 ASIA PACIFIC: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SNAPSHOT
- FIGURE 27 REVENUE ANALYSIS FOR KEY PLAYERS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, 2022-2024 (USD MILLION)
- FIGURE 28 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SHARE ANALYSIS OF KEY PLAYERS (2024)
- FIGURE 29 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS) 2024,
- FIGURE 30 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: COMPANY FOOTPRINT
- FIGURE 31 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2024
- FIGURE 32 EV/EBITDA OF KEY VENDORS
- FIGURE 33 YEAR-TO-DATE (YTD) PRICE, TOTAL RETURN, AND 5-YEAR STOCK BETA OF KEY VENDORS
- FIGURE 34 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: BRAND/PRODUCT COMPARATIVE ANALYSIS
- FIGURE 35 CHARLES RIVER LABORATORIES: COMPANY SNAPSHOT
- FIGURE 38 JSR CORPORATION: COMPANY SNAPSHOT
- FIGURE 39 GENOWAY: COMPANY SNAPSHOT
- FIGURE 40 WUXI APPTEC + WUXI BIOLOGICS: COMPANY SNAPSHOT
- FIGURE 41 BIOCYTOGEN PHARMACEUTICALS CO., LTD.: COMPANY SNAPSHOT
- FIGURE 42 CHAMPIONS ONCOLOGY, INC.: COMPANY SNAPSHOT
- FIGURE 43 PHARMARON: COMPANY SNAPSHOT
- FIGURE 44 SYNGENE INTERNATIONAL LIMITED: COMPANY SNAPSHOT
- FIGURE 45 LABCORP: COMPANY SNAPSHOT
- FIGURE 46 EUROFINS SCIENTIFIC: COMPANY SNAPSHOT
- FIGURE 47 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: RESEARCH DESIGN
- FIGURE 48 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: BREAKDOWN OF PRIMARIES (SUPPLY- AND DEMAND-SIDE PARTICIPANTS)
- FIGURE 49 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SIZE ESTIMATION (SUPPLY-SIDE ANALYSIS), 2024
- FIGURE 50 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: COMPANY REVENUE ANALYSIS-BASED ESTIMATION (2024)
- FIGURE 51 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: ILLUSTRATIVE REVENUE ANALYSIS OF CHARLES RIVER LABORATORIES (2024)
- FIGURE 52 PATIENT-DERIVED XENOGRAFTS (PDX) MODELS MARKET SIZE VALIDATION FROM PRIMARY SOURCES
- FIGURE 53 PATIENT-DERIVED XENOGRAFTS (PDX) MODELS MARKET SIZE ESTIMATION METHODOLOGY: TOP-DOWN APPROACH
- FIGURE 54 PATIENT-DERIVED XENOGRAFTS (PDX) MODELS MARKET: CAGR PROJECTIONS
- FIGURE 55 PATIENT-DERIVED XENOGRAFTS (PDX) MODELS MARKET: DATA TRIANGULATION METHODOLOGY








