 |
市场调查报告书
商品编码
1936010
全球製药过滤市场按产品、技术、应用、类型、企业规模、最终用户和地区划分-预测至2030年Pharmaceutical Filtration Market by Product (Membrane filter, Depth filter, Virus filter, Air Filter, Assemblies, Systems (Single-use)), Technique (Ultrafiltration), Type (Sterile), Application (API, Protein), Scale, End User - Global Forecast to 2030 |
||||||
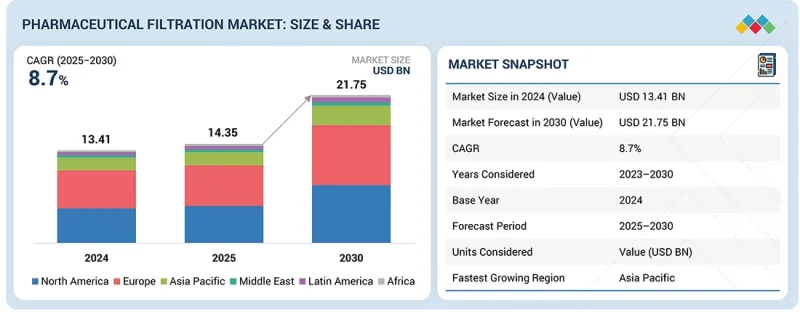
全球医药过滤市场预计到 2030 年将达到 217.5 亿美元,高于 2025 年的 143.5 亿美元。
预计2025年至2030年的年复合成长率(CAGR)为8.7%。
| 调查范围 | |
|---|---|
| 调查期 | 2023-2030 |
| 基准年 | 2024 |
| 预测期 | 2025-2030 |
| 目标单元 | 金额(十亿美元) |
| 部分 | 按产品、技术、应用、类型、业务规模、最终用户和地区划分 |
| 目标区域 | 北美、欧洲、亚太地区、拉丁美洲、中东和非洲 |
製药过滤市场的扩张主要受全球对无菌性和病毒安全性的监管要求不断提高的推动,然而,技术限制预计将限制市场成长。
按类型划分,无菌过滤是製药过滤市场中成长最快的细分市场,这主要归功于对开发先进治疗方案的日益重视以及生物医学研究的快速发展。随着研究活动的不断扩展,在药物研发和生产过程中维持严格的无菌环境变得愈发重要。这极大地推动了无菌过滤技术的应用,该技术在防止微生物污染、保持产品完整性以及确保符合严格的监管标准方面发挥关键作用。这些系统对于保护生物製药在生产各个阶段的品质至关重要,尤其是在细胞培养製备、上游工程和最终製剂阶段。
推动该领域成长的另一个因素是生物製药和生物相似药核准数量的不断增加。随着更复杂、更敏感的生物分子进入市场,对能够在不影响无菌保证的前提下处理高通量工作流程的可靠无菌过滤方法的需求日益增长。这些生物製药需要强大的过滤工艺,以有效去除微生物,同时保持其结构稳定性和治疗效果。
此外,为应对感染疾病和新出现的健康威胁,全球疫苗生产规模不断扩大,这进一步推动了对无菌过滤的需求。疫苗生产高度依赖高性能过滤系统来去除污染物,确保最终产品的安全性和无菌性。这些因素共同巩固了无菌过滤作为医药过滤市场成长最快细分领域的地位。
2024年,最终产品加工领域在应用领域中占据最大份额。
从应用领域来看,医药过滤市场可分为最终产品加工、原料过滤、细胞分离、水净化和空气净化。
2024年,最终产品加工成为製药过滤产业规模最大、成长最快的应用领域。这一主导地位主要源自于药品生产最后阶段对产品纯度、无菌保证和污染控制日益严格的监管要求。生物製药领域,特别是单株抗体、细胞和基因疗法以及疫苗平台的快速扩张,显着提升了对高性能过滤解决方案的需求。此外,生物製药和生物相似药产量的稳定成长,进一步强化了对高效、检验的过滤系统的需求,以确保产品品质稳定并符合法规要求。这些因素共同推动了全球製药过滤市场最终产品加工领域的强劲成长动能。
2024年,北美在全球医药过滤市场中占据最大的市场份额。
北美凭藉其强大的生物製药生产生态系统和对先进过滤技术的快速应用,在製药过滤市场中保持领先地位。该地区汇聚了许多全球製药巨头、创新生物技术公司和大规模合约研发生产机构(CDMO),所有这些企业都需要高性能过滤系统来支援生物製剂、疫苗以及细胞和基因疗法的生产。美国食品药物管理局(FDA)严格的监管标准推动了薄膜技术、无菌过滤系统和自动化品管工具的持续升级。大量的研发投入以及大量的临床试验,支撑了对尖端过滤解决方案的稳定需求。此外,北美还受益于其完善的製药基础设施、经验丰富的生物製程工程师以及主要过滤供应商的大规模产能扩张。政府为增强生物製造韧性、应对疫情以及推广下一代疗法而采取的倡议,进一步巩固了该地区的领先地位,确保了製药过滤领域的持续技术进步和市场成长。
本报告中主要企业概况
製药过滤市场的主要参与企业包括:默克集团(德国)、丹纳赫公司(美国)、赛多利斯股份公司(德国)、Solventum公司(美国)、Repligen公司(美国)、派克汉尼汾公司(美国)、伊顿公司(爱尔兰)、赛默飞世尔科技公司(美国)、唐纳森公司(美国)、Porvair公司(英国)、阿法拉伐公司(瑞典)、康宁公司(美国)、曼胡默尔公司(德国)、圣戈班公司(法国)、STERIS公司(美国)和旭化成株式会社(日本)。
调查范围
本研究报告按产品、技术、类型、应用、企业规模和最终用户对製药过滤市场进行细分,并提供 2030 年的全球预测。
本报告涵盖影响医药过滤市场成长的关键因素的详细信息,包括驱动因素、限制因素、挑战和机会。该报告还对主要产业参与企业进行了详细分析,深入剖析了他们的业务概况、产品、解决方案、关键策略、联盟、伙伴关係和协议。此外,报告还涵盖了医药过滤市场的新核准和上市、合作、收购以及最新发展动态。
购买本报告的主要优势:
本报告将为市场领导和新参与企业提供最准确的医药过滤市场及其细分市场整体收入预测,从而帮助他们更好地掌握市场动态。此外,本报告还将帮助相关人员深入了解竞争格局,并获取有效市场定位和製定合适打入市场策略的见解。透过本报告,相关人员能够掌握市场动态,并了解关键的市场驱动因素、限制因素、机会和挑战。
本报告深入分析了以下内容:
- 该报告分析了关键驱动因素(生物製药和生物相似药的开发和商业化不断加强、全球对无菌性和病毒安全性的监管要求日益严格、一次性过滤系统和组件的日益普及、对无菌和高浓度药品的需求不断增长以及连续生产工艺的采用)、限制因素(先进过滤技术和一次性系统的高成本、技术限制)、机会(智慧过滤与数位化和人工智慧驱动的工艺相结合、日益关注永续和环保的过滤解决方案)以及挑战(严格的监管合规性、技术和运营方面的挑战以及熟练劳动力短缺)。
- 产品开发/创新:深入分析医药过滤市场的新技术趋势与研发活动
- 市场发展:关于盈利市场的全面资讯-该报告分析了各个地区的市场状况。
- 市场多元化:关于医药过滤市场未开发地区、近期趋势和投资的全面讯息
- 竞争格局评估:对主要企业的市场占有率、成长策略和产品供应进行详细评估。对主要产业参与企业进行深入分析,提供有关其关键策略、产品发布和核准情况、研发管线分析、收购、合作、协议、协作和其他近期发展、投资和资金筹措活动、品牌和产品对比分析,以及医药过滤市场供应商估值和财务指标的见解。
目录
第一章 引言
第二章执行摘要
第三章重要考察
第四章 市场概览
- 市场动态
- 未满足的需求和差距
- 相互关联的市场与跨产业机会
- 1/2/3级参与企业的策略倡议
第五章 产业趋势
- 波特五力分析
- 宏观经济展望
- 价值链分析
- 生态系分析
- 定价分析
- 贸易分析
- 大型会议和活动
- 影响客户业务的趋势/干扰因素
- 投资/资金筹措活动
- 案例研究分析
- 2025年美国关税对医药过滤市场的影响
第六章:技术进步、人工智慧的影响、专利、创新与未来应用
- 技术分析
- 技术/产品蓝图
- 专利分析
- 未来应用
- 人工智慧/生成式人工智慧对製药过滤市场的影响
第七章 监理环境与永续性倡议
- 地方法规和合规性
- 永续性影响和监管政策倡议
- 认证、标籤和环境标准
第八章:顾客状况与购买行为
- 决策流程
- 买方相关人员和采购评估标准
- 招募障碍和内部挑战
- 来自各个终端使用者产业的未满足需求
- 市场盈利
9. 医药过滤市场(依产品分类)
- 消耗品
- 系统
第十章:医药过滤市场(依技术分类)
- 微滤
- 超过滤
- 奈米过滤
- 其他的
第十一章 医药过滤市场(按类型划分)
- 无菌过滤
- 非无菌过滤
第十二章 医药过滤市场(依应用领域划分)
- 最终产品加工
- 原料过滤
- 细胞分离
- 水净化
- 空气净化
第十三章 医药过滤市场(依企业规模划分)
- 生产规模
- 中试规模
- 研发规模
第十四章 医药过滤市场(依最终用户划分)
- 製药和生物製药公司
- 合约研究组织 (CRO)、合约行销组织 (CMO)
- 学术研究机构
第十五章 区域性医药过滤市场
- 北美洲
- 北美:宏观经济分析
- 我们
- 加拿大
- 欧洲
- 欧洲:宏观经济分析
- 德国
- 英国
- 法国
- 义大利
- 西班牙
- 瑞士
- 其他的
- 亚太地区
- 亚太地区:宏观经济分析
- 中国
- 日本
- 印度
- 韩国
- 澳洲
- 其他的
- 拉丁美洲
- 拉丁美洲:宏观经济分析
- 巴西
- 墨西哥
- 其他的
- 中东
- 中东:宏观经济分析
- 海湾合作委员会国家
- 其他的
- 非洲
- 非洲:宏观经济分析
第十六章 竞争格局
- 主要参与企业采取的策略
- 收入份额分析
- 市占率分析
- 公司评估矩阵:主要参与企业
- 公司评估矩阵:Start-Ups/中小企业
- 製药过滤供应商估值和财务指标
- 品牌/产品比较分析
- 竞争场景
第十七章:公司简介
- 主要参与企业
- MERCK KGAA
- DANAHER CORPORATION
- SARTORIUS AG
- SOLVENTUM
- PARKER HANNIFIN CORPORATION
- REPLIGEN CORPORATION
- EATON CORPORATION PLC
- THERMO FISHER SCIENTIFIC, INC.
- DONALDSON COMPANY, INC.
- PORVAIR PLC
- ALFA LAVAL CORPORATE AB
- CORNING INCORPORATED
- MANN+HUMMEL
- SAINT-GOBAIN
- STERIS PLC
- ASAHI KASEI CORPORATION
- 其他公司
- MEISSNER FILTRATION PRODUCTS, INC.
- AMAZON FILTERS LTD.
- GRAVER TECHNOLOGIES, LLC
- MMS AG
- ERTELALSOP
- KASAG SWISS AG
- FREUDENBERG FILTRATION TECHNOLOGIES SE & CO. KG
- ANTYLIA SCIENTIFIC
- FILTROX AG
- MEMBRANE SOLUTIONS
第十八章调查方法
第十九章附录
The global pharmaceutical filtration market is projected to reach USD 21.75 billion by 2030 from an estimated USD 14.35 billion in 2025, at a CAGR of 8.7% from 2025 to 2030.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2023-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD billion) |
| Segments | Product, Technique, Type, Application, Scale of Operation, End User. |
| Regions covered | North America, Europe, the Asia Pacific, Latin America, the Middle East, and Africa |
The expansion of the pharmaceutical filtration market has been predominantly fueled by the tightening global regulatory expectations for sterility and viral safety. However, technological limitations are expected to restrict the market.

In 2024, sterile filtration was the fastest-growing segment in the pharmaceutical filtration market by type.
The sterile filtration segment was the fastest-growing segment in the pharmaceutical filtration market, by type, primarily because of the growing emphasis on developing advanced therapeutic solutions and the rapid progress being made in biomedical research. As research activities expand, the need to maintain strictly sterile environments during drug development and manufacturing becomes increasingly critical. This has significantly boosted the adoption of sterile filtration technologies, which play a vital role in preventing microbial contamination, preserving product integrity, and ensuring compliance with stringent regulatory standards. These systems are essential for safeguarding the quality of biopharmaceuticals at various stages of production, particularly during cell culture preparation, upstream processing, and final formulation.
Another major contributor to the segment's growth is the rising number of approvals for biologics and biosimilars. As more complex and sensitive biological molecules enter the market, demand intensifies for reliable sterile filtration methods capable of managing high-throughput workflows without compromising sterility assurance. These biologics require robust filtration processes to remove microorganisms efficiently while maintaining structural stability and therapeutic effectiveness.
Additionally, global expansion in vaccine manufacturing, driven by the need to address infectious diseases and emerging health threats, has further strengthened the demand for sterile filtration. Vaccine production heavily depends on high-performance filtration systems to eliminate contaminants and ensure safe, sterile end products. Together, these factors have firmly positioned sterile filtration as the fastest-growing segment within the pharmaceutical filtration market.
The final product processing segment reported the highest share of the application segment in 2024.
Within the application segment, the pharmaceutical filtration market is divided into final product processing, raw material filtration, cell separation, water purification, and air purification.
In 2024, final product processing emerged as both the largest and the fastest-expanding application area in the pharmaceutical filtration industry. This dominance is primarily driven by rising regulatory expectations for product purity, sterility assurance, and contamination control throughout the final stages of drug manufacturing. As the biopharmaceutical sector continues to expand rapidly, particularly in the production of monoclonal antibodies, cell- and gene-based therapies, and vaccine platforms, the need for high-performance filtration solutions has grown significantly. Additionally, the steady increase in biologics and biosimilar production has intensified the requirement for efficient and validated filtration systems to ensure consistent quality and regulatory compliance. Collectively, these factors reinforce the strong growth trajectory of the final product processing segment within the global pharmaceutical filtration market.
North America accounted for the largest market share in the global pharmaceutical filtration market in 2024.
North America maintains its dominant position in the pharmaceutical filtration market due to its strong biopharmaceutical manufacturing ecosystem and rapid adoption of advanced filtration technologies. The region hosts numerous global pharma leaders, innovative biotech firms, and large CDMOs, all of which require high-performance filtration systems to support biologics, vaccines, and cell and gene therapy production. Strict regulatory standards imposed by the FDA drive continuous upgrades in membrane technologies, sterile filtration systems, and automated quality-control tools. Significant investment in R&D, coupled with a high volume of clinical trials, supports steady demand for cutting-edge filtration solutions. North America also benefits from a well-established pharmaceutical infrastructure, skilled bioprocessing talent, and extensive capacity expansions by major filtration suppliers. Government initiatives promoting biomanufacturing resilience, pandemic preparedness, and next-generation therapies further strengthen the region's leadership, ensuring sustained technological advancement and market growth in pharmaceutical filtration.
The primary interviews conducted for this report can be categorized as follows:
- By Respondent: Supply Side-70% and Demand Side-30%
- By Designation: Managers-45%, CXO and Directors-30%, and Executives-25%
- By Region: North America-40%, Europe-25%, the Asia Pacific-25%, Latin America-5%, and the Middle East & Africa-5%
Key Companies Profiled in the Report
Key players in the Pharmaceutical filtration market include Merck KGaA (Germany), Danaher Corporation (US), Sartorius AG (Germany), Solventum (US), Repligen Corporation (US), Parker Hannifin Corporation (US), Eaton Corporation plc (Ireland), Thermo Fisher Scientific Inc. (US), Donaldson Company, Inc. (US), Porvair PLC (UK), Alfa Laval Corporate AB (Sweden), Corning Incorporated (US), MANN+HUMMEL (Germany), Saint-Gobain (France), STERIS plc (US), Asahi Kasei Corporation(Japan).
Research Coverage
This research report categorizes the Pharmaceutical Filtration Market by Product (Membrane filter, Depth filter, Virus filter, Air Filter, Assemblies, Systems [Single-use]), Technique (Ultrafiltration), Type (Sterile), Application (API, Protein), Scale, End User - Global Forecast to 2030.
The scope of the report covers detailed information regarding the major factors, such as drivers, restraints, challenges, and opportunities, influencing the growth of the pharmaceutical filtration market. A detailed analysis of the key industry players has been done to provide insights into their business overview, products, solutions, key strategies, collaborations, partnerships, and agreements. New approvals/launches, collaborations, acquisitions, and recent developments associated with the pharmaceutical filtration market.
Key Benefits of Buying the Report:
The report will help market leaders and new entrants by providing them with the closest approximations of the revenue numbers for the overall pharmaceutical filtration market and its subsegments. It will also help stakeholders better understand the competitive landscape and gain more insights to better position their businesses and make suitable go-to-market strategies. This report will enable stakeholders to understand the market's pulse and provide them with information on the key market drivers, restraints, opportunities, and challenges.
The report provides insights into the following pointers:
- Analysis of key drivers (Increasing development and commercialization of biologics and biosimilars, tightening global regulatory expectations for sterility and viral safety, growing adoption of single-use filtration systems and assemblies, rising need for sterile and high-concentration drug products, and Adoption of continuous manufacturing process), restraints (High cost of advanced filtration technologies and single-use systems, technological limitations), opportunities (Integration of smart filtration with digitalization and AI-driven process and growing focus on sustainable and eco-friendly filtration solutions) and Challenges (Stringent regulatory compliance, technical and operational challenges, lack of skilled workforce).
- Product Development/Innovation: Detailed insights on upcoming technologies, research & development activities in the pharmaceutical filtration market
- Market Development: Comprehensive information about lucrative markets - the report analyses the market across varied regions.
- Market Diversification: Exhaustive information about untapped geographies, recent developments, and investments in the pharmaceutical filtration market
- Competitive Assessment: In-depth assessment of market shares, growth strategies, and product offerings of leading players. A detailed analysis of the key industry players has been done to provide insights into their key strategies, product launches/approvals, pipeline analysis, acquisitions, partnerships, agreements, collaborations, other recent developments, investment and funding activities, brand/product comparative analysis, and vendor valuation and financial metrics of the pharmaceutical filtration market.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.3.4 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS AND MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: SHARE INSIGHTS AND STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS SHAPING MARKET
- 2.4 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 PHARMACEUTICAL FILTRATION MARKET OVERVIEW
- 3.2 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE & COUNTRY
- 3.3 PHARMACEUTICAL FILTRATION MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Rapid growth in development and commercialization of biologics and biosimilars
- 4.2.1.2 Increasing global regulatory demands for sterility and viral safety
- 4.2.1.3 Growing adoption of single-use filtration systems & assemblies
- 4.2.1.4 Rising need for sterile, high-concentration drugs
- 4.2.1.5 Adoption of continuous manufacturing process
- 4.2.2 RESTRAINTS
- 4.2.2.1 High cost of advanced filtration technologies and single-use systems
- 4.2.2.2 Technological limitations
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Smart filtration with digitalization and innovation
- 4.2.3.2 Growing focus on sustainable and eco-friendly filtration solutions
- 4.2.4 CHALLENGES
- 4.2.4.1 Stringent regulatory compliance
- 4.2.4.2 Technical and operational challenges
- 4.2.4.3 Lack of skilled workforce
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS AND WHITE SPACES
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 BARGAINING POWER OF SUPPLIERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMIC OUTLOOK
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS AND FORECASTS
- 5.2.3 TRENDS IN GLOBAL PHARMACEUTICAL FILTRATION MARKET
- 5.3 VALUE CHAIN ANALYSIS
- 5.4 ECOSYSTEM ANALYSIS
- 5.5 PRICING ANALYSIS
- 5.5.1 AVERAGE SELLING PRICE TREND OF KEY PLAYERS, BY PRODUCT, 2022-2024
- 5.5.2 AVERAGE SELLING PRICE TREND, BY REGION, 2022-2024
- 5.6 TRADE ANALYSIS
- 5.6.1 IMPORT DATA FOR HS CODE 8421, 2020-2024
- 5.6.2 EXPORT DATA FOR HS CODE 8421, 2020-2024
- 5.6.3 EXPORT VOLUME FOR HS CODE 8421, 2020-2024
- 5.6.4 IMPORT VOLUME FOR HS CODE 8421, 2020-2024
- 5.7 KEY CONFERENCES & EVENTS
- 5.8 TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- 5.9 INVESTMENT/FUNDING ACTIVITY
- 5.9.1 VC/PRIVATE EQUITY INVESTMENT TRENDS & STARTUP LANDSCAPE
- 5.10 CASE STUDY ANALYSIS
- 5.11 IMPACT OF 2025 US TARIFF ON PHARMACEUTICAL FILTRATION MARKET
- 5.11.1 INTRODUCTION
- 5.11.2 KEY TARIFF RATES
- 5.11.3 PRICE IMPACT ANALYSIS
- 5.11.4 IMPACT ON COUNTRY/REGION
- 5.11.4.1 US
- 5.11.4.2 Europe
- 5.11.4.3 Asia Pacific
- 5.11.5 IMPACT ON END-USE INDUSTRIES
- 5.11.5.1 Pharmaceutical & biotechnology companies
- 5.11.5.2 CROs and CDMOs/CMOs
- 5.11.5.3 Academic & research institutes
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 TECHNOLOGY ANALYSIS
- 6.1.1 KEY TECHNOLOGIES
- 6.1.1.1 Microfiltration
- 6.1.1.2 Ultrafiltration
- 6.1.1.3 Nanofiltration
- 6.1.2 COMPLEMENTARY TECHNOLOGIES
- 6.1.2.1 Electrospun microfibers
- 6.1.2.2 Photocatalytic filtration
- 6.1.3 ADJACENT TECHNOLOGIES
- 6.1.3.1 Chromatography
- 6.1.3.2 Microfluidics
- 6.1.1 KEY TECHNOLOGIES
- 6.2 TECHNOLOGY/PRODUCT ROADMAP
- 6.3 PATENT ANALYSIS
- 6.3.1 TOP APPLICANTS/OWNERS (COMPANIES) FOR PHARMACEUTICAL FILTRATION PATENTS, 2014-2024
- 6.4 FUTURE APPLICATIONS
- 6.5 IMPACT OF AI/GEN AI ON PHARMACEUTICAL FILTRATION MARKET
- 6.5.1 TOP USE CASES AND MARKET POTENTIAL
- 6.5.2 CASE STUDIES OF AI IMPLEMENTATION IN PHARMACEUTICAL FILTRATION MARKET
- 6.5.3 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS
- 6.5.4 CLIENTS' READINESS TO ADOPT GENERATIVE AI IN PHARMACEUTICAL FILTRATION MARKET
7 REGULATORY LANDSCAPE AND SUSTAINABILITY INITIATIVES
- 7.1 REGIONAL REGULATIONS AND COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 REGULATORY FRAMEWORK
- 7.1.3 INDUSTRY STANDARDS
- 7.2 SUSTAINABILITY IMPACT AND REGULATORY POLICY INITIATIVES
- 7.3 CERTIFICATIONS, LABELING, & ECO-STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS AND BUYING EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 KEY BUYING CRITERIA, BY END USER
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.5 MARKET PROFITABILITY
9 PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT
- 9.1 INTRODUCTION
- 9.2 CONSUMABLES
- 9.2.1 FILTERS
- 9.2.1.1 Membrane filters
- 9.2.1.1.1 Polyethersulfone (PES)
- 9.2.1.1.1.1 Wide use of hydrophilic PES membranes in sterilizing applications to drive growth
- 9.2.1.1.2 Polyvinylidene difluoride (PVDF)
- 9.2.1.1.2.1 Nontoxic nature to increase demand for PVDF filters
- 9.2.1.1.3 Nylon
- 9.2.1.1.3.1 Increased endotoxin adsorption capacity of nylon filters to drive growth
- 9.2.1.1.4 Polytetrafluoroethylene (PTFE)
- 9.2.1.1.4.1 PTFE membrane filters offer retention of ratings as fine as 0.01μ
- 9.2.1.1.5 Mixed cellulose ester & cellulose acetate (MCE & CA)
- 9.2.1.1.5.1 MCE membranes widely used in analytical and research applications
- 9.2.1.1.6 Polycarbonate track-etched (PCTE)
- 9.2.1.1.6.1 PCTE membrane filters widely used in cell biology and analytical testing applications
- 9.2.1.1.7 Other materials
- 9.2.1.1.1 Polyethersulfone (PES)
- 9.2.1.2 Depth filters
- 9.2.1.2.1 Diatomaceous Earth
- 9.2.1.2.1.1 Increasing use in biotechnology industry for cell clarification to drive market
- 9.2.1.2.2 Cellulose
- 9.2.1.2.2.1 Cost-effectiveness of cellulose-based depth filters to drive adoption
- 9.2.1.2.3 Activated carbon
- 9.2.1.2.3.1 Widely used in pharmaceutical industry to remove range of soluble impurities from process streams containing APIs
- 9.2.1.2.4 Perlite
- 9.2.1.2.4.1 Better suited for separating coarse micro-particles from liquids with high solids loading
- 9.2.1.2.5 Other depth filter media
- 9.2.1.2.1 Diatomaceous Earth
- 9.2.1.3 Air filters
- 9.2.1.4 Virus filters
- 9.2.1.5 Other filters
- 9.2.1.1 Membrane filters
- 9.2.2 OTHER CONSUMABLES
- 9.2.2.1 FILTRATION ASSEMBLIES
- 9.2.2.1.1 Microfiltration assemblies
- 9.2.2.1.1.1 Growing demand for high-quality products to support segment growth
- 9.2.2.1.2 Ultrafiltration assemblies
- 9.2.2.1.2.1 Stringent regulatory requirements to support segment growth
- 9.2.2.1.3 Nanofiltration assemblies
- 9.2.2.1.3.1 Increasing demand for high-purity biopharmaceuticals to propel market
- 9.2.2.1.4 Other filtration assemblies
- 9.2.2.1.1 Microfiltration assemblies
- 9.2.2.2 FILTER HOLDERS
- 9.2.2.2.1 Filter holders provide structural support to membrane filters
- 9.2.2.3 FILTRATION ACCESSORIES
- 9.2.2.3.1 Increasing adoption of pharmaceutical filtration products to support growth
- 9.2.2.1 FILTRATION ASSEMBLIES
- 9.2.1 FILTERS
- 9.3 SYSTEMS
- 9.3.1 SINGLE-USE SYSTEMS
- 9.3.1.1 Reduced need for product validation and minimized cross-contamination risk to boost adoption
- 9.3.2 REUSABLE SYSTEMS
- 9.3.2.1 Widely used for large-scale manufacturing to drive adoption
- 9.3.1 SINGLE-USE SYSTEMS
10 PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE
- 10.1 INTRODUCTION
- 10.2 MICROFILTRATION
- 10.2.1 INCREASED ADOPTION OF MICROFILTRATION TO PROPEL GROWTH
- 10.3 ULTRAFILTRATION
- 10.3.1 ADOPTION OF FINE FILTRATION TECHNIQUES TO DRIVE GROWTH
- 10.4 NANOFILTRATION
- 10.4.1 RISING DEMAND IN BIOTECHNOLOGY INDUSTRY TO SUPPORT MARKET GROWTH
- 10.5 OTHER TECHNIQUES
11 PHARMACEUTICAL FILTRATION MARKET, BY TYPE
- 11.1 INTRODUCTION
- 11.2 STERILE FILTRATION
- 11.2.1 ADVANTAGES OF STERILE FILTERS OVER NON-STERILE FILTERS TO DRIVE GROWTH
- 11.3 NON-STERILE FILTRATION
- 11.3.1 GROWTH IN PHARMA R&D ACTIVITIES TO PROPEL MARKET
12 PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION
- 12.1 INTRODUCTION
- 12.2 FINAL PRODUCT PROCESSING
- 12.2.1 ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION
- 12.2.1.1 Continuous requirement for API filtration to drive market
- 12.2.2 PROTEIN PURIFICATION
- 12.2.2.1 Recent advancements in protein therapeutic drugs to propel growth
- 12.2.3 VACCINE AND ANTIBODY PROCESSING
- 12.2.3.1 Development of vaccines for viral diseases to drive growth
- 12.2.4 FORMULATION AND FILLING SOLUTIONS
- 12.2.4.1 Demand for aseptic filling and bioburden reduction to propel growth
- 12.2.5 VIRAL CLEARANCE
- 12.2.5.1 Growing development of therapeutic monoclonal antibodies to drive market
- 12.2.1 ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION
- 12.3 RAW MATERIAL FILTRATION
- 12.3.1 MEDIA AND BUFFER FILTRATION
- 12.3.1.1 Growth in manufacturing of biopharmaceuticals to drive market
- 12.3.2 PREFILTRATION
- 12.3.2.1 Membrane fouling issues to drive growth
- 12.3.3 BIOBURDEN TESTING
- 12.3.3.1 Strict quality control of biopharmaceuticals to drive growth
- 12.3.1 MEDIA AND BUFFER FILTRATION
- 12.4 CELL SEPARATION
- 12.4.1 GROWTH IN PERSONALIZED MEDICINE AND CELL THERAPIES TO PROPEL MARKET
- 12.5 WATER PURIFICATION
- 12.5.1 REQUIREMENT IN RESEARCH AND FORMULATION TO DRIVE MARKET
- 12.6 AIR PURIFICATION
- 12.6.1 INCREASING ADOPTION OF GMP PRACTICES TO DRIVE GROWTH
13 PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION
- 13.1 INTRODUCTION
- 13.2 MANUFACTURING-SCALE
- 13.2.1 GROWTH IN BIOLOGICS AND BIOSIMILARS MANUFACTURING TO DRIVE MARKET
- 13.3 PILOT-SCALE
- 13.3.1 INCREASED OUTSOURCING OF PILOT-SCALE BIOPROCESS DEVELOPMENT TO DRIVE MARKET
- 13.4 R&D-SCALE
- 13.4.1 INCREASING INVESTMENTS IN R&D OF BIOLOGICS TO PROPEL GROWTH
14 PHARMACEUTICAL FILTRATION MARKET, BY END USER
- 14.1 INTRODUCTION
- 14.2 PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES
- 14.2.1 GROWING BIOLOGICS PRODUCTION TO PROPEL DEMAND
- 14.3 CROS & CMOS
- 14.3.1 GROWTH IN PHARMA R&D ACTIVITIES TO DRIVE MARKET
- 14.4 ACADEMIC & RESEARCH INSTITUTES
- 14.4.1 RISING DEMAND FOR LABORATORY FILTRATION ACROSS CROS AND RESEARCH INSTITUTES TO PROPEL MARKET
15 PHARMACEUTICAL FILTRATION MARKET, BY REGION
- 15.1 INTRODUCTION
- 15.2 NORTH AMERICA
- 15.2.1 NORTH AMERICA: MACROECONOMIC ANALYSIS
- 15.2.2 US
- 15.2.2.1 US to dominate North America market during forecast period
- 15.2.3 CANADA
- 15.2.3.1 Growing government support for expanding biologics manufacturing facilities to support market growth
- 15.3 EUROPE
- 15.3.1 EUROPE: MACROECONOMIC ANALYSIS
- 15.3.2 GERMANY
- 15.3.2.1 Growing pharmaceutical R&D and manufacturing to drive market
- 15.3.3 UK
- 15.3.3.1 Increased awareness of drug quality led to greater use of filtration products in drug discovery laboratories
- 15.3.4 FRANCE
- 15.3.4.1 Government investment in pharmaceutical industry to drive growth
- 15.3.5 ITALY
- 15.3.5.1 Investment in biotechnology R&D to boost market growth
- 15.3.6 SPAIN
- 15.3.6.1 Expansion of biomanufacturing facilities to support market growth
- 15.3.7 SWITZERLAND
- 15.3.7.1 Presence of key pharmaceutical and biopharmaceutical companies to support market growth
- 15.3.8 REST OF EUROPE
- 15.4 ASIA PACIFIC
- 15.4.1 ASIA PACIFIC: MACROECONOMIC ANALYSIS
- 15.4.2 CHINA
- 15.4.2.1 China to dominate Asia Pacific market during forecast period
- 15.4.3 JAPAN
- 15.4.3.1 Stringent regulatory guidelines to create need for advanced filtration products
- 15.4.4 INDIA
- 15.4.4.1 Favorable scenario for foreign direct investment to favor market growth
- 15.4.5 SOUTH KOREA
- 15.4.5.1 Increased export of drugs to support market growth
- 15.4.6 AUSTRALIA
- 15.4.6.1 Increasing demand for innovative filtration solutions to propel market
- 15.4.7 REST OF ASIA PACIFIC
- 15.5 LATIN AMERICA
- 15.5.1 LATIN AMERICA: MACROECONOMIC ANALYSIS
- 15.5.2 BRAZIL
- 15.5.2.1 Increased government investments in pharmaceutical R&D to drive market
- 15.5.3 MEXICO
- 15.5.3.1 Rising demand for chronic disease treatment to support market growth
- 15.5.4 REST OF LATIN AMERICA
- 15.6 MIDDLE EAST
- 15.6.1 MIDDLE EAST: MACROECONOMIC ANALYSIS
- 15.6.2 GCC COUNTRIES
- 15.6.2.1 Saudi Arabia
- 15.6.2.1.1 Growing healthcare expenditure to boost market growth
- 15.6.2.2 UAE
- 15.6.2.2.1 Adoption of more efficient and precise filtration processes to aid market growth
- 15.6.2.3 Rest of GCC Countries
- 15.6.2.1 Saudi Arabia
- 15.6.3 REST OF MIDDLE EAST
- 15.7 AFRICA
- 15.7.1 AFRICA: MACROECONOMIC ANALYSIS
16 COMPETITIVE LANDSCAPE
- 16.1 INTRODUCTION
- 16.2 STRATEGIES ADOPTED BY KEY PLAYERS
- 16.3 REVENUE SHARE ANALYSIS
- 16.4 MARKET SHARE ANALYSIS
- 16.5 COMPANY EVALUATION MATRIX: KEY PLAYERS
- 16.5.1 STARS
- 16.5.2 EMERGING LEADERS
- 16.5.3 PERVASIVE PLAYERS
- 16.5.4 PARTICIPANTS
- 16.5.5 COMPETITIVE BENCHMARKING: KEY PLAYERS, 2024
- 16.5.5.1 Company footprint
- 16.5.5.2 Product footprint
- 16.5.5.3 Technique footprint
- 16.5.5.4 Application footprint
- 16.5.5.5 Regional footprint
- 16.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES
- 16.6.1 PROGRESSIVE COMPANIES
- 16.6.2 RESPONSIVE COMPANIES
- 16.6.3 DYNAMIC COMPANIES
- 16.6.4 STARTING BLOCKS
- 16.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2024
- 16.6.5.1 Detailed list of key startups/SMEs, 2024
- 16.6.5.2 Competitive benchmarking of key startups/SMEs, 2024
- 16.7 VALUATION AND FINANCIAL METRICS OF PHARMACEUTICAL FILTRATION VENDORS
- 16.8 BRAND/PRODUCT COMPARATIVE ANALYSIS
- 16.8.1 BRAND/PRODUCT COMPARATIVE ANALYSIS, BY PRODUCT
- 16.9 COMPETITIVE SCENARIO
- 16.9.1 PRODUCT LAUNCHES
- 16.9.2 DEALS
- 16.9.3 EXPANSIONS
17 COMPANY PROFILES
- 17.1 KEY PLAYERS
- 17.1.1 MERCK KGAA
- 17.1.1.1 Business overview
- 17.1.1.2 Products offered
- 17.1.1.3 Recent developments
- 17.1.1.3.1 Expansions
- 17.1.1.4 MnM view
- 17.1.1.4.1 Key strengths
- 17.1.1.4.2 Strategic choices
- 17.1.1.4.3 Weaknesses and competitive threats
- 17.1.2 DANAHER CORPORATION
- 17.1.2.1 Business overview
- 17.1.2.2 Products offered
- 17.1.2.3 Recent developments
- 17.1.2.3.1 Product launches
- 17.1.2.3.2 Deals
- 17.1.2.3.3 Expansions
- 17.1.2.4 MnM view
- 17.1.2.4.1 Key strengths
- 17.1.2.4.2 Strategic choices
- 17.1.2.4.3 Weaknesses and competitive threats
- 17.1.3 SARTORIUS AG
- 17.1.3.1 Business overview
- 17.1.3.2 Products offered
- 17.1.3.3 Recent developments
- 17.1.3.3.1 Product launches
- 17.1.3.3.2 Deals
- 17.1.3.3.3 Expansions
- 17.1.3.4 MnM view
- 17.1.3.4.1 Key strengths
- 17.1.3.4.2 Strategic choices
- 17.1.3.4.3 Weaknesses and competitive threats
- 17.1.4 SOLVENTUM
- 17.1.4.1 Business overview
- 17.1.4.2 Products offered
- 17.1.4.3 Recent developments
- 17.1.4.3.1 Product launches
- 17.1.4.3.2 Deals
- 17.1.4.3.3 Other developments
- 17.1.4.4 MnM view
- 17.1.4.4.1 Key strengths
- 17.1.4.4.2 Strategic choices
- 17.1.4.4.3 Weaknesses and competitive threats
- 17.1.5 PARKER HANNIFIN CORPORATION
- 17.1.5.1 Business overview
- 17.1.5.2 Products offered
- 17.1.5.3 MnM view
- 17.1.5.3.1 Key strengths
- 17.1.5.3.2 Strategic choices
- 17.1.5.3.3 Weaknesses and competitive threats
- 17.1.6 REPLIGEN CORPORATION
- 17.1.6.1 Business overview
- 17.1.6.2 Products offered
- 17.1.6.3 Recent developments
- 17.1.6.3.1 Product launches
- 17.1.6.3.2 Deals
- 17.1.7 EATON CORPORATION PLC
- 17.1.7.1 Business overview
- 17.1.7.2 Products offered
- 17.1.7.3 Recent developments
- 17.1.7.3.1 Product launches
- 17.1.8 THERMO FISHER SCIENTIFIC, INC.
- 17.1.8.1 Business overview
- 17.1.8.2 Products offered
- 17.1.8.3 Recent developments
- 17.1.8.3.1 Deals
- 17.1.8.3.2 Expansions
- 17.1.9 DONALDSON COMPANY, INC.
- 17.1.9.1 Business overview
- 17.1.9.2 Products offered
- 17.1.9.3 Recent developments
- 17.1.9.3.1 Deals
- 17.1.9.3.2 Expansions
- 17.1.10 PORVAIR PLC
- 17.1.10.1 Business overview
- 17.1.10.2 Products offered
- 17.1.10.3 Recent developments
- 17.1.10.3.1 Product launches
- 17.1.11 ALFA LAVAL CORPORATE AB
- 17.1.11.1 Business overview
- 17.1.11.2 Products offered
- 17.1.11.3 Recent developments
- 17.1.11.3.1 Product launches
- 17.1.12 CORNING INCORPORATED
- 17.1.12.1 Business overview
- 17.1.12.2 Products offered
- 17.1.13 MANN+HUMMEL
- 17.1.13.1 Business overview
- 17.1.13.2 Products offered
- 17.1.13.3 Recent developments
- 17.1.13.3.1 Deals
- 17.1.14 SAINT-GOBAIN
- 17.1.14.1 Business overview
- 17.1.14.2 Products offered
- 17.1.14.3 Recent developments
- 17.1.14.3.1 Product launches
- 17.1.14.3.2 Expansions
- 17.1.15 STERIS PLC
- 17.1.15.1 Business overview
- 17.1.15.2 Products offered
- 17.1.16 ASAHI KASEI CORPORATION
- 17.1.16.1 Business overview
- 17.1.16.2 Products offered
- 17.1.16.3 Recent developments
- 17.1.16.3.1 Product launches
- 17.1.16.3.2 Deals
- 17.1.16.3.3 Expansions
- 17.1.1 MERCK KGAA
- 17.2 OTHER PLAYERS
- 17.2.1 MEISSNER FILTRATION PRODUCTS, INC.
- 17.2.2 AMAZON FILTERS LTD.
- 17.2.3 GRAVER TECHNOLOGIES, LLC
- 17.2.4 MMS AG
- 17.2.5 ERTELALSOP
- 17.2.6 KASAG SWISS AG
- 17.2.7 FREUDENBERG FILTRATION TECHNOLOGIES SE & CO. KG
- 17.2.8 ANTYLIA SCIENTIFIC
- 17.2.9 FILTROX AG
- 17.2.10 MEMBRANE SOLUTIONS
18 RESEARCH METHODOLOGY
- 18.1 RESEARCH DATA
- 18.1.1 SECONDARY DATA
- 18.1.2 PRIMARY DATA
- 18.2 MARKET ESTIMATION METHODOLOGY
- 18.2.1 MARKET SIZE ESTIMATION
- 18.2.2 INSIGHTS OF PRIMARY EXPERTS
- 18.2.3 TOP-DOWN APPROACH
- 18.3 MARKET GROWTH RATE PROJECTIONS
- 18.4 MARKET BREAKDOWN AND DATA TRIANGULATION
- 18.5 RESEARCH ASSUMPTIONS
- 18.6 RESEARCH LIMITATIONS
- 18.7 RISK ANALYSIS
19 APPENDIX
- 19.1 DISCUSSION GUIDE
- 19.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 19.3 CUSTOMIZATION OPTIONS
- 19.4 RELATED REPORTS
- 19.5 AUTHOR DETAILS
List of Tables
- TABLE 1 PHARMACEUTICAL FILTRATION MARKET: IMPACT ANALYSIS
- TABLE 2 LIST OF KEY BIOSIMILARS APPROVED, 2023-2025
- TABLE 3 PHARMACEUTICAL FILTRATION MARKET: PORTER'S FIVE FORCES ANALYSIS
- TABLE 4 PHARMACEUTICAL FILTRATION MARKET: ROLE IN ECOSYSTEM
- TABLE 5 AVERAGE SELLING PRICE TREND OF KEY PLAYERS, BY PRODUCT, 2022-2024
- TABLE 6 AVERAGE SELLING PRICE, BY REGION, 2022-2024
- TABLE 7 IMPORT DATA FOR HS CODE 8421, BY COUNTRY, 2020-2024 (USD THOUSAND)
- TABLE 8 EXPORT DATA FOR HS CODE 8421, BY COUNTRY, 2020-2024 (USD THOUSAND)
- TABLE 9 EXPORT VOLUME FOR HS CODE 8421, BY COUNTRY, 2020-2024 (TON)
- TABLE 10 IMPORT VOLUME FOR HS CODE 8421, BY COUNTRY, 2020-2024 (TON)
- TABLE 11 PHARMACEUTICAL FILTRATION MARKET: KEY CONFERENCES & EVENTS, 2025-2027
- TABLE 12 ADVANCED VIRAL FILTRATION ENABLING SAFE MAB PRODUCTION SCALE-UP
- TABLE 13 RAPID SCALE-UP OF VIRAL VECTOR FILTRATION FOR COVID-19 VACCINE
- TABLE 14 HIGH-THROUGHPUT VIRAL FILTRATION OPTIMIZATION
- TABLE 15 US-ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 16 CRITICAL COMPONENTS LIKELY EXPOSED TO TARIFF CHANGES
- TABLE 17 INDICATIVE LIST OF PATENTS IN PHARMACEUTICAL FILTRATION MARKET, 2025
- TABLE 18 CASE STUDIES OF AI IMPLEMENTATION IN PHARMACEUTICAL FILTRATION MARKET
- TABLE 19 CLIENT'S READINESS TO ADOPT GENERATIVE AI IN PHARMACEUTICAL FILTRATION MARKET
- TABLE 20 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 21 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 22 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 23 REST OF THE WORLD: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 24 COUNTRY-WISE REGULATORY SCENARIO FOR PHARMACEUTICAL FILTER MANUFACTURERS
- TABLE 25 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS, BY PRODUCTS (%)
- TABLE 26 KEY BUYING CRITERIA, BY END USER
- TABLE 27 PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 28 PHARMACEUTICAL CONSUMABLES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 29 NORTH AMERICA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 30 EUROPE: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 31 ASIA PACIFIC: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 32 LATIN AMERICA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 33 MIDDLE EAST: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 34 GCC COUNTRIES: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 35 PHARMACEUTICAL FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 36 NORTH AMERICA: PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 37 EUROPE: PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 38 ASIA PACIFIC: PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 39 LATIN AMERICA: PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 40 MIDDLE EAST: PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 41 GCC COUNTRIES: PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 42 PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 43 PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 44 NORTH AMERICA: PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 45 EUROPE: PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 46 ASIA PACIFIC: PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 47 LATIN AMERICA: PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 48 MIDDLE EAST: PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 49 GCC COUNTRIES: PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 50 PHARMACEUTICAL MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 51 POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 52 NORTH AMERICA: POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 53 EUROPE: POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 54 ASIA PACIFIC: POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 55 LATIN AMERICA: POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 56 MIDDLE EAST: POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 57 GCC COUNTRIES: POLYETHERSULFONE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 58 POLYVINYLIDENE DIFLUORIDE MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 59 NORTH AMERICA: PVDF MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 60 EUROPE: PVDF MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 61 ASIA PACIFIC: PVDF MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 62 LATIN AMERICA: PVDF MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 63 MIDDLE EAST: PVDF MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 64 GCC COUNTRIES: PVDF MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 65 NYLON MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 66 NORTH AMERICA: NYLON MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 67 EUROPE: NYLON MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 68 ASIA PACIFIC: NYLON MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 69 LATIN AMERICA: NYLON MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 70 MIDDLE EAST: NYLON MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 71 GCC COUNTRIES: NYLON MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 72 POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 73 NORTH AMERICA: POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 74 EUROPE: POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 75 ASIA PACIFIC: POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 76 LATIN AMERICA: POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 77 MIDDLE EAST: POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 78 GCC COUNTRIES: POLYTETRAFLUOROETHYLENE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 79 MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 80 NORTH AMERICA: MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 81 EUROPE: MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 82 ASIA PACIFIC: MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 83 LATIN AMERICA: MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 84 MIDDLE EAST: MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 85 GCC COUNTRIES: MIXED CELLULOSE ESTER & CELLULOSE ACETATE MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 86 POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 87 NORTH AMERICA: POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 88 EUROPE: POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 89 ASIA PACIFIC: POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 90 LATIN AMERICA: POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 91 MIDDLE EAST: POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 92 GCC COUNTRIES: POLYCARBONATE TRACK-ETCHED MEMBRANE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 93 OTHER MEMBRANE FILTER MATERIALS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 94 NORTH AMERICA: OTHER MEMBRANE FILTER MATERIALS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 95 EUROPE: OTHER MEMBRANE FILTER MATERIALS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 96 ASIA PACIFIC: OTHER MEMBRANE FILTER MATERIALS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 97 LATIN AMERICA: OTHER MEMBRANE FILTER MATERIALS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 98 MIDDLE EAST: OTHER MEMBRANE FILTER MATERIALS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 99 GCC COUNTRIES: OTHER MEMBRANE FILTER MATERIALS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 100 DEPTH FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 101 NORTH AMERICA: DEPTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 102 EUROPE: DEPTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 103 ASIA PACIFIC: DEPTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 104 LATIN AMERICA: DEPTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 105 MIDDLE EAST: DEPTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 106 GCC COUNTRIES: DEPTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 107 DEPTH FILTERS MARKET, BY MEDIA, 2023-2030 (USD MILLION)
- TABLE 108 DIATOMACEOUS EARTH FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 109 NORTH AMERICA: DIATOMACEOUS EARTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 110 EUROPE: DIATOMACEOUS EARTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 111 ASIA PACIFIC: DIATOMACEOUS EARTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 112 LATIN AMERICA: DIATOMACEOUS EARTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 113 MIDDLE EAST: DIATOMACEOUS EARTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 114 GCC COUNTRIES: DIATOMACEOUS EARTH FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 115 CELLULOSE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 116 NORTH AMERICA: CELLULOSE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 117 EUROPE: CELLULOSE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 118 ASIA PACIFIC: CELLULOSE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 119 LATIN AMERICA: CELLULOSE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 120 MIDDLE EAST: CELLULOSE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 121 GCC COUNTRIES: CELLULOSE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 122 ACTIVATED CARBON FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 123 NORTH AMERICA: ACTIVATED CARBON FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 124 EUROPE: ACTIVATED CARBON FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 125 ASIA PACIFIC: ACTIVATED CARBON FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 126 LATIN AMERICA: ACTIVATED CARBON FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 127 MIDDLE EAST: ACTIVATED CARBON FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 128 GCC COUNTRIES: ACTIVATED CARBON FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 129 PERLITE FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 130 NORTH AMERICA: PERLITE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 131 EUROPE: PERLITE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 132 ASIA PACIFIC: PERLITE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 133 LATIN AMERICA: PERLITE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 134 MIDDLE EAST: PERLITE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 135 GCC COUNTRIES: PERLITE FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 136 OTHER DEPTH FILTER MEDIA MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 137 NORTH AMERICA: OTHER DEPTH FILTER MEDIA MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 138 EUROPE: OTHER DEPTH FILTER MEDIA MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 139 ASIA PACIFIC: OTHER DEPTH FILTER MEDIA MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 140 LATIN AMERICA: OTHER DEPTH FILTER MEDIA MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 141 MIDDLE EAST: OTHER DEPTH FILTER MEDIA MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 142 GCC COUNTRIES: OTHER DEPTH FILTER MEDIA MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 143 AIR FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 144 NORTH AMERICA: AIR FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 145 EUROPE: AIR FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 146 ASIA PACIFIC: AIR FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 147 LATIN AMERICA: AIR FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 148 MIDDLE EAST: AIR FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 149 GCC COUNTRIES: AIR FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 150 VIRUS FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 151 NORTH AMERICA: VIRUS FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 152 EUROPE: VIRUS FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 153 ASIA PACIFIC: VIRUS FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 154 LATIN AMERICA: VIRUS FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 155 MIDDLE EAST: VIRUS FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 156 GCC COUNTRIES: VIRUS FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 157 OTHER PHARMACEUTICAL FILTERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 158 NORTH AMERICA: OTHER PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 159 EUROPE: OTHER PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 160 ASIA PACIFIC: OTHER PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 161 LATIN AMERICA: OTHER PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 162 MIDDLE EAST: OTHER PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 163 GCC COUNTRIES: OTHER PHARMACEUTICAL FILTERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 164 OTHER CONSUMABLES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 165 NORTH AMERICA: OTHER CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 166 EUROPE: OTHER CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 167 ASIA PACIFIC: OTHER CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 168 LATIN AMERICA: OTHER CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 169 MIDDLE EAST: OTHER CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 170 GCC COUNTRIES: OTHER CONSUMABLES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 171 OTHER CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 172 FILTRATION ASSEMBLIES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 173 NORTH AMERICA: FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 174 EUROPE: FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 175 ASIA PACIFIC: FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 176 LATIN AMERICA: FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 177 MIDDLE EAST: FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 178 GCC COUNTRIES: FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 179 FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 180 MICROFILTRATION ASSEMBLIES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 181 NORTH AMERICA: MICROFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 182 EUROPE: MICROFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 183 ASIA PACIFIC: MICROFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 184 LATIN AMERICA: MICROFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 185 MIDDLE EAST: MICROFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 186 GCC COUNTRIES: MICROFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 187 ULTRAFILTRATION ASSEMBLIES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 188 NORTH AMERICA: ULTRAFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 189 EUROPE: ULTRAFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 190 ASIA PACIFIC: ULTRAFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 191 LATIN AMERICA: ULTRAFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 192 MIDDLE EAST: ULTRAFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 193 GCC COUNTRIES: ULTRAFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 194 NANOFILTRATION ASSEMBLIES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 195 NORTH AMERICA: NANOFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 196 EUROPE: NANOFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 197 ASIA PACIFIC: NANOFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 198 LATIN AMERICA: NANOFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 199 MIDDLE EAST: NANOFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 200 GCC COUNTRIES: NANOFILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 201 OTHER FILTRATION ASSEMBLIES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 202 NORTH AMERICA: OTHER FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 203 EUROPE: OTHER FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 204 ASIA PACIFIC: OTHER FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 205 LATIN AMERICA: OTHER FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 206 MIDDLE EAST: OTHER FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 207 GCC COUNTRIES: OTHER FILTRATION ASSEMBLIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 208 FILTER HOLDERS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 209 NORTH AMERICA: FILTER HOLDERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 210 EUROPE: FILTER HOLDERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 211 ASIA PACIFIC: FILTER HOLDERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 212 LATIN AMERICA: FILTER HOLDERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 213 MIDDLE EAST: FILTER HOLDERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 214 GCC COUNTRIES: FILTER HOLDERS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 215 FILTRATION ACCESSORIES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 216 NORTH AMERICA: FILTRATION ACCESSORIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 217 EUROPE: FILTRATION ACCESSORIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 218 ASIA PACIFIC: FILTRATION ACCESSORIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 219 LATIN AMERICA: FILTRATION ACCESSORIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 220 MIDDLE EAST: FILTRATION ACCESSORIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 221 GCC COUNTRIES: FILTRATION ACCESSORIES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 222 FILTRATION SYSTEMS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 223 NORTH AMERICA: FILTRATION SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 224 EUROPE: FILTRATION SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 225 ASIA PACIFIC: FILTRATION SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 226 LATIN AMERICA: FILTRATION SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 227 MIDDLE EAST: FILTRATION SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 228 GCC COUNTRIES: FILTRATION SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 229 FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 230 SINGLE-USE SYSTEMS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 231 NORTH AMERICA: SINGLE-USE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 232 EUROPE: SINGLE-USE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 233 ASIA PACIFIC: SINGLE-USE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 234 LATIN AMERICA: SINGLE-USE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 235 MIDDLE EAST: SINGLE-USE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 236 GCC COUNTRIES: SINGLE-USE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 237 REUSABLE SYSTEMS MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 238 NORTH AMERICA: REUSABLE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 239 EUROPE: REUSABLE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 240 ASIA PACIFIC: REUSABLE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 241 LATIN AMERICA: REUSABLE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 242 MIDDLE EAST: REUSABLE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 243 GCC COUNTRIES: REUSABLE SYSTEMS MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 244 PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 245 MICROFILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 246 NORTH AMERICA: MICROFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 247 EUROPE: MICROFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 248 ASIA PACIFIC: MICROFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 249 LATIN AMERICA: MICROFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 250 MIDDLE EAST: MICROFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 251 GCC COUNTRIES: MICROFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 252 ULTRAFILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 253 NORTH AMERICA: ULTRAFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 254 EUROPE: ULTRAFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 255 ASIA PACIFIC: ULTRAFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 256 LATIN AMERICA: ULTRAFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 257 MIDDLE EAST: ULTRAFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 258 GCC COUNTRIES: ULTRAFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 259 NANOFILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 260 NORTH AMERICA: NANOFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 261 EUROPE: NANOFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 262 ASIA PACIFIC: NANOFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 263 LATIN AMERICA: NANOFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 264 MIDDLE EAST: NANOFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 265 GCC COUNTRIES: NANOFILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 266 OTHER FILTRATION TECHNIQUES MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 267 NORTH AMERICA: OTHER FILTRATION TECHNIQUES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 268 EUROPE: OTHER FILTRATION TECHNIQUES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 269 ASIA PACIFIC: OTHER FILTRATION TECHNIQUES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 270 LATIN AMERICA: OTHER FILTRATION TECHNIQUES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 271 MIDDLE EAST: OTHER FILTRATION TECHNIQUES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 272 GCC COUNTRIES: OTHER FILTRATION TECHNIQUES MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 273 PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 274 STERILE FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 275 NORTH AMERICA: STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 276 EUROPE: STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 277 ASIA PACIFIC: STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 278 LATIN AMERICA: STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 279 MIDDLE EAST: STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 280 GCC COUNTRIES: STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 281 NON-STERILE FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 282 NORTH AMERICA: NON-STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 283 EUROPE: NON-STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 284 ASIA PACIFIC: NON-STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 285 LATIN AMERICA: NON-STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 286 MIDDLE EAST: NON-STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 287 GCC COUNTRIES: NON-STERILE FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 288 PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 289 PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY REGION, 2023-2030 (USD MILLION)
- TABLE 290 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 291 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 292 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 293 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 294 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 295 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 296 PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 297 ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 298 NORTH AMERICA: ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 299 EUROPE: ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 300 ASIA PACIFIC: ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 301 LATIN AMERICA: ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 302 MIDDLE EAST: ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 303 GCC COUNTRIES: ACTIVE PHARMACEUTICAL INGREDIENT FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 304 PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 305 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 306 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 307 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 308 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 309 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 310 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR PROTEIN PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 311 PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY REGION, 2023-2030 (USD MILLION)
- TABLE 312 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 313 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 314 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 315 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 316 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 317 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR VACCINE AND ANTIBODY PROCESSING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 318 PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 319 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 320 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 321 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 322 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 323 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 324 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR FORMULATION AND FILLING SOLUTIONS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 325 PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY REGION, 2023-2030 (USD MILLION)
- TABLE 326 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 327 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 328 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 329 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 330 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 331 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR VIRAL CLEARANCE, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 332 PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 333 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 334 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 335 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 336 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 337 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 338 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 339 PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 340 PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 341 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 342 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 343 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 344 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 345 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 346 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR MEDIA AND BUFFER FILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 347 PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 348 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 349 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 350 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 351 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 352 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 353 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR PREFILTRATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 354 PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY REGION, 2023-2030 (USD MILLION)
- TABLE 355 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 356 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 357 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 358 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 359 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 360 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR BIOBURDEN TESTING, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 361 PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 362 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 363 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 364 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 365 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 366 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 367 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR CELL SEPARATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 368 PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 369 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 370 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 371 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 372 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 373 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 374 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR WATER PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 375 PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY REGION, 2023-2030 (USD MILLION)
- TABLE 376 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 377 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 378 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 379 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 380 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 381 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR AIR PURIFICATION, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 382 PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 383 LIST OF FDA-APPROVED BIOSIMILAR PRODUCTS, 2022-2024
- TABLE 384 MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 385 NORTH AMERICA: MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 386 EUROPE: MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 387 ASIA PACIFIC: MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 388 LATIN AMERICA: MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 389 MIDDLE EAST: MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 390 GCC COUNTRIES: MANUFACTURING-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 391 PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 392 NORTH AMERICA: PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 393 EUROPE: PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 394 ASIA PACIFIC: PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 395 LATIN AMERICA: PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 396 MIDDLE EAST: PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 397 GCC COUNTRIES: PILOT-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 398 R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 399 NORTH AMERICA: R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 400 EUROPE: R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 401 ASIA PACIFIC: R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 402 LATIN AMERICA: R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 403 MIDDLE EAST: R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 404 GCC COUNTRIES: R&D-SCALE PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 405 PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 406 PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY REGION, 2023-2030 (USD MILLION)
- TABLE 407 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 408 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 409 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 410 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 411 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 412 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 413 PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY REGION, 2023-2030 (USD MILLION)
- TABLE 414 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 415 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 416 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 417 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 418 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 419 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR CROS & CMOS, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 420 PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY REGION, 2023-2030 (USD MILLION)
- TABLE 421 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 422 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 423 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 424 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 425 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 426 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR ACADEMIC & RESEARCH INSTITUTES, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 427 PHARMACEUTICAL FILTRATION MARKET, BY REGION, 2023-2030 (USD MILLION)
- TABLE 428 MACROECONOMIC OUTLOOK FOR PHARMACEUTICAL FILTRATION MARKET IN NORTH AMERICA
- TABLE 429 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 430 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 431 NORTH AMERICA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 432 NORTH AMERICA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 433 NORTH AMERICA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 434 NORTH AMERICA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 435 NORTH AMERICA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 436 NORTH AMERICA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 437 NORTH AMERICA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 438 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 439 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 440 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 441 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 442 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 443 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 444 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 445 US: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 446 US: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 447 US: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 448 US: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 449 US: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 450 US: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 451 US: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 452 US: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 453 US: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 454 US: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 455 US: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 456 US: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 457 US: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 458 US: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 459 US: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 460 CANADA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 461 CANADA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 462 CANADA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 463 CANADA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 464 CANADA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 465 CANADA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 466 CANADA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 467 CANADA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 468 CANADA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 469 CANADA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 470 CANADA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 471 CANADA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 472 CANADA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 473 CANADA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 474 CANADA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 475 MACROECONOMIC OUTLOOK FOR PHARMACEUTICAL FILTRATION MARKET IN EUROPE
- TABLE 476 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 477 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 478 EUROPE: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 479 EUROPE: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 480 EUROPE: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 481 EUROPE: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 482 EUROPE: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 483 EUROPE: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 484 EUROPE: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 485 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 486 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 487 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 488 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 489 EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 490 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 491 EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 492 GERMANY: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 493 GERMANY: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 494 GERMANY: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 495 GERMANY: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 496 GERMANY: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 497 GERMANY: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 498 GERMANY: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 499 GERMANY: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 500 GERMANY: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 501 GERMANY: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 502 GERMANY: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 503 GERMANY: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 504 GERMANY: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 505 GERMANY: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 506 GERMANY: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 507 UK: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 508 UK: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 509 UK: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 510 UK: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 511 UK: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 512 UK: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 513 UK: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 514 UK: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 515 UK: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 516 UK: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 517 UK: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 518 UK: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 519 UK: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 520 UK: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 521 UK: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 522 FRANCE: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 523 FRANCE: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 524 FRANCE: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 525 FRANCE: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 526 FRANCE: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 527 FRANCE: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 528 FRANCE: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 529 FRANCE: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 530 FRANCE: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 531 FRANCE: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 532 FRANCE: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 533 FRANCE: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 534 FRANCE: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 535 FRANCE: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 536 FRANCE: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 537 ITALY: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 538 ITALY: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 539 ITALY: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 540 ITALY: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 541 ITALY: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 542 ITALY: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 543 ITALY: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 544 ITALY: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 545 ITALY: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 546 ITALY: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 547 ITALY: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 548 ITALY: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 549 ITALY: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 550 ITALY: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 551 ITALY: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 552 SPAIN: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 553 SPAIN: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 554 SPAIN: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 555 SPAIN: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 556 SPAIN: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 557 SPAIN: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 558 SPAIN: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 559 SPAIN: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 560 SPAIN: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 561 SPAIN: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 562 SPAIN: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 563 SPAIN: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 564 SPAIN: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 565 SPAIN: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 566 SPAIN: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 567 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 568 SWITZERLAND: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 569 SWITZERLAND: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 570 SWITZERLAND: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 571 SWITZERLAND: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 572 SWITZERLAND: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 573 SWITZERLAND: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 574 SWITZERLAND: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 575 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 576 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 577 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 578 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 579 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 580 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 581 SWITZERLAND: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 582 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 583 REST OF EUROPE: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 584 REST OF EUROPE: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 585 REST OF EUROPE: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 586 REST OF EUROPE: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 587 REST OF EUROPE: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 588 REST OF EUROPE: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 589 REST OF EUROPE: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 590 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 591 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 592 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 593 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 594 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 595 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 596 REST OF EUROPE: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 597 MACROECONOMIC OUTLOOK FOR PHARMACEUTICAL FILTRATION MARKET IN ASIA PACIFIC
- TABLE 598 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 599 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 600 ASIA PACIFIC: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 601 ASIA PACIFIC: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 602 ASIA PACIFIC: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 603 ASIA PACIFIC: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 604 ASIA PACIFIC: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 605 ASIA PACIFIC: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 606 ASIA PACIFIC: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 607 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 608 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 609 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 610 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 611 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 612 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 613 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 614 CHINA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 615 CHINA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 616 CHINA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 617 CHINA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 618 CHINA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 619 CHINA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 620 CHINA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 621 CHINA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 622 CHINA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 623 CHINA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 624 CHINA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 625 CHINA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 626 CHINA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 627 CHINA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 628 CHINA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 629 JAPAN: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 630 JAPAN: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 631 JAPAN: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 632 JAPAN: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 633 JAPAN: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 634 JAPAN: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 635 JAPAN: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 636 JAPAN: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 637 JAPAN: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 638 JAPAN: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 639 JAPAN: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 640 JAPAN: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 641 JAPAN: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 642 JAPAN: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 643 JAPAN: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 644 INDIA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 645 INDIA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 646 INDIA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 647 INDIA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 648 INDIA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 649 INDIA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 650 INDIA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 651 INDIA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 652 INDIA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 653 INDIA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 654 INDIA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 655 INDIA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 656 INDIA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 657 INDIA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 658 INDIA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 659 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 660 SOUTH KOREA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 661 SOUTH KOREA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 662 SOUTH KOREA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 663 SOUTH KOREA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 664 SOUTH KOREA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 665 SOUTH KOREA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 666 SOUTH KOREA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 667 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 668 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 669 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 670 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 671 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 672 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 673 SOUTH KOREA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 674 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 675 AUSTRALIA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 676 AUSTRALIA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 677 AUSTRALIA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 678 AUSTRALIA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 679 AUSTRALIA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 680 AUSTRALIA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 681 AUSTRALIA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 682 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 683 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 684 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 685 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 686 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 687 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 688 AUSTRALIA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 689 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 690 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 691 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 692 REST OF ASIA PACIFIC: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 693 REST OF ASIA PACIFIC: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 694 REST OF ASIA PACIFIC: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 695 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 696 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 697 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 698 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 699 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 700 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 701 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 702 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 703 REST OF ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 704 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 705 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 706 LATIN AMERICA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 707 LATIN AMERICA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 708 LATIN AMERICA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 709 LATIN AMERICA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 710 LATIN AMERICA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 711 LATIN AMERICA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 712 LATIN AMERICA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 713 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 714 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 715 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 716 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 717 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 718 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 719 LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 720 BRAZIL: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 721 BRAZIL: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 722 BRAZIL: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 723 BRAZIL: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 724 BRAZIL: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 725 BRAZIL : OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 726 BRAZIL: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 727 BRAZIL: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 728 BRAZIL: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 729 BRAZIL: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 730 BRAZIL: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 731 BRAZIL: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 732 BRAZIL: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 733 BRAZIL: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 734 BRAZIL: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 735 MEXICO: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 736 MEXICO: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 737 MEXICO: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 738 MEXICO: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 739 MEXICO: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 740 MEXICO: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 741 MEXICO: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 742 MEXICO: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 743 MEXICO: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 744 MEXICO: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 745 MEXICO: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 746 MEXICO: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 747 MEXICO: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 748 MEXICO: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 749 MEXICO: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 750 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 751 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 752 REST OF LATIN AMERICA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 753 REST OF LATIN AMERICA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 754 REST OF LATIN AMERICA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 755 REST OF LATIN AMERICA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 756 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 757 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 758 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 759 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 760 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 761 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 762 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 763 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 764 REST OF LATIN AMERICA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 765 MACROECONOMIC OUTLOOK FOR PHARMACEUTICAL FILTRATION MARKET IN MIDDLE EAST
- TABLE 766 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY REGION, 2023-2030 (SD MILLION)
- TABLE 767 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 768 MIDDLE EAST: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 769 MIDDLE EAST: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 770 MIDDLE EAST: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 771 MIDDLE EAST: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 772 MIDDLE EAST: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 773 MIDDLE EAST: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 774 MIDDLE EAST: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 775 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 776 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 777 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 778 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 779 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 780 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 781 MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 782 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY COUNTRY, 2023-2030 (USD MILLION)
- TABLE 783 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 784 GCC COUNTRIES: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 785 GCC COUNTRIES: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 786 GCC COUNTRIES: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 787 GCC COUNTRIES: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 788 GCC COUNTRIES: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 789 GCC COUNTRIES: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 790 GCC COUNTRIES: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 791 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 792 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 793 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 794 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 795 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 796 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 797 GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 798 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 799 SAUDI ARABIA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 800 SAUDI ARABIA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 801 SAUDI ARABIA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 802 SAUDI ARABIA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 803 SAUDI ARABIA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 804 SAUDI ARABIA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 805 SAUDI ARABIA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 806 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 807 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 808 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 809 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 810 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 811 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 812 SAUDI ARABIA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 813 UAE: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 814 UAE: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 815 UAE: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 816 UAE: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 817 UAE: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 818 UAE: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 819 UAE: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 820 UAE: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 821 UAE: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 822 UAE: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 823 UAE: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 824 UAE: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 825 UAE: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 826 UAE: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 827 UAE: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 828 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 829 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 830 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 831 REST OF GCC COUNTRIES: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 832 REST OF GCC COUNTRIES: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 833 REST OF GCC COUNTRIES: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 834 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 835 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 836 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 837 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 838 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 839 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 840 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 841 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 842 REST OF GCC COUNTRIES: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 843 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 844 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 845 REST OF MIDDLE EAST: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 846 REST OF MIDDLE EAST: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 847 REST OF MIDDLE EAST: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 848 REST OF MIDDLE EAST: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 849 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 850 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 851 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 852 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 853 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 854 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 855 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 856 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 857 REST OF MIDDLE EAST: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 858 MACROECONOMIC OUTLOOK FOR PHARMACEUTICAL FILTRATION MARKET IN AFRICA
- TABLE 859 AFRICA: PHARMACEUTICAL FILTRATION MARKET, BY PRODUCT, 2023-2030 (USD MILLION)
- TABLE 860 AFRICA: PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 861 AFRICA: PHARMACEUTICAL FILTERS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 862 AFRICA: MEMBRANE FILTERS MARKET, BY MATERIAL, 2023-2030 (USD MILLION)
- TABLE 863 AFRICA: DEPTH FILTERS MARKET, BY TYPE OF MEDIA, 2023-2030 (USD MILLION)
- TABLE 864 AFRICA: OTHER PHARMACEUTICAL FILTRATION CONSUMABLES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 865 AFRICA: PHARMACEUTICAL FILTRATION ASSEMBLIES MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 866 AFRICA: PHARMACEUTICAL FILTRATION SYSTEMS MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 867 AFRICA: PHARMACEUTICAL FILTRATION MARKET, BY TECHNIQUE, 2023-2030 (USD MILLION)
- TABLE 868 AFRICA: PHARMACEUTICAL FILTRATION MARKET, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 869 AFRICA: PHARMACEUTICAL FILTRATION MARKET, BY APPLICATION, 2023-2030 (USD MILLION)
- TABLE 870 AFRICA: PHARMACEUTICAL FILTRATION MARKET FOR FINAL PRODUCT PROCESSING, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 871 AFRICA: PHARMACEUTICAL FILTRATION MARKET FOR RAW MATERIAL FILTRATION, BY TYPE, 2023-2030 (USD MILLION)
- TABLE 872 AFRICA: PHARMACEUTICAL FILTRATION MARKET, BY SCALE OF OPERATION, 2023-2030 (USD MILLION)
- TABLE 873 AFRICA: PHARMACEUTICAL FILTRATION MARKET, BY END USER, 2023-2030 (USD MILLION)
- TABLE 874 STRATEGIES ADOPTED BY KEY PLAYERS IN PHARMACEUTICAL FILTRATION MARKET, 2022-2024
- TABLE 875 PHARMACEUTICAL FILTRATION MARKET: DEGREE OF COMPETITION
- TABLE 876 PHARMACEUTICAL FILTRATION MARKET: PRODUCT FOOTPRINT
- TABLE 877 PHARMACEUTICAL FILTRATION MARKET: TECHNIQUE FOOTPRINT
- TABLE 878 PHARMACEUTICAL FILTRATION MARKET: APPLICATION FOOTPRINT
- TABLE 879 PHARMACEUTICAL FILTRATION MARKET: REGIONAL FOOTPRINT
- TABLE 880 PHARMACEUTICAL FILTRATION MARKET: DETAILED LIST OF KEY STARTUPS/SMES
- TABLE 881 PHARMACEUTICAL FILTRATION: COMPETITIVE BENCHMARKING OF KEY STARTUPS/SMES
- TABLE 882 PHARMACEUTICAL FILTRATION MARKET: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 883 PHARMACEUTICAL FILTRATION MARKET: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 884 PHARMACEUTICAL FILTRATION MARKET: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 885 MERCK KGAA: COMPANY OVERVIEW
- TABLE 886 MERCK KGAA: PRODUCTS OFFERED
- TABLE 887 MERCK KGAA: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 888 DANAHER CORPORATION: COMPANY OVERVIEW
- TABLE 889 DANAHER CORPORATION: PRODUCTS OFFERED
- TABLE 890 DANAHER CORPORATION: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 891 DANAHER CORPORATION: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 892 DANAHER CORPORATION: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 893 SARTORIUS AG: COMPANY OVERVIEW
- TABLE 894 SARTORIUS AG: PRODUCTS OFFERED
- TABLE 895 SARTORIUS AG: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 896 SARTORIUS AG: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 897 SARTORIUS AG: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 898 :SOLVENTUM: COMPANY OVERVIEW
- TABLE 899 SOLVENTUM: PRODUCTS OFFERED
- TABLE 900 SOLVENTUM: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 901 SOLVENTUM: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 902 SOLVENTUM: OTHER DEVELOPMENTS, JANUARY 2022-NOVEMBER 2025
- TABLE 903 PARKER HANNIFIN CORPORATION: COMPANY OVERVIEW
- TABLE 904 PARKER HANNIFIN CORPORATION: PRODUCTS OFFERED
- TABLE 905 REPLIGEN CORPORATION: COMPANY OVERVIEW
- TABLE 906 REPLIGEN CORPORATION: PRODUCTS OFFERED
- TABLE 907 REPLIGEN CORPORATION: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 908 REPLIGEN CORPORATION: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 909 EATON CORPORATION PLC: COMPANY OVERVIEW
- TABLE 910 EATON CORPORATION PLC: PRODUCTS OFFERED
- TABLE 911 EATON CORPORATION PLC: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 912 THERMO FISHER SCIENTIFIC, INC.: COMPANY OVERVIEW
- TABLE 913 THERMO FISHER SCIENTIFIC, INC.: PRODUCTS OFFERED
- TABLE 914 THERMO FISHER SCIENTIFIC, INC.: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 915 THERMO FISHER SCIENTIFIC, INC.: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 916 DONALDSON COMPANY, INC.: COMPANY OVERVIEW
- TABLE 917 DONALDSON COMPANY, INC.: PRODUCTS OFFERED
- TABLE 918 DONALDSON COMPANY, INC.: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 919 DONALDSON COMPANY, INC.: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 920 PORVAIR PLC: COMPANY OVERVIEW
- TABLE 921 PORVAIR PLC: PRODUCTS OFFERED
- TABLE 922 PORVAIR PLC: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 923 ALFA LAVAL CORPORATE AB: COMPANY OVERVIEW
- TABLE 924 ALFA LAVAL CORPORATE AB: PRODUCTS OFFERED
- TABLE 925 ALFA LAVAL CORPORATE AB: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 926 CORNING INCORPORATED: COMPANY OVERVIEW
- TABLE 927 CORNING INCORPORATED: PRODUCTS OFFERED
- TABLE 928 MANN+HUMMEL: COMPANY OVERVIEW
- TABLE 929 MANN+HUMMEL: PRODUCTS OFFERED
- TABLE 930 MANN+HUMMEL: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 931 SAINT-GOBAIN: COMPANY OVERVIEW
- TABLE 932 SAINT-GOBAIN: PRODUCTS OFFERED
- TABLE 933 SAINT-GOBAIN: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 934 SAINT-GOBAIN: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 935 STERIS PLC: COMPANY OVERVIEW
- TABLE 936 STERIS PLC: PRODUCTS OFFERED
- TABLE 937 ASAHI KASEI CORPORATION: COMPANY OVERVIEW
- TABLE 938 ASAHI KASEI CORPORATION: PRODUCTS OFFERED
- TABLE 939 ASAHI KASEI CORPORATION: PRODUCT LAUNCHES, JANUARY 2022-NOVEMBER 2025
- TABLE 940 ASAHI KASEI CORPORATION: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 941 ASAHI KASEI CORPORATION: EXPANSIONS, JANUARY 2022-NOVEMBER 2025
- TABLE 942 MEISSNER FILTRATION PRODUCTS, INC.: COMPANY OVERVIEW
- TABLE 943 AMAZON FILTERS LTD.: COMPANY OVERVIEW
- TABLE 944 GRAVER TECHNOLOGIES, LLC: COMPANY OVERVIEW
- TABLE 945 MMS AG: COMPANY OVERVIEW
- TABLE 946 ERTELALSOP: COMPANY OVERVIEW
- TABLE 947 KASAG SWISS AG: COMPANY OVERVIEW
- TABLE 948 FREUDENBERG FILTRATION TECHNOLOGIES SE & CO. KG: COMPANY OVERVIEW
- TABLE 949 ANTYLIA SCIENTIFIC: COMPANY OVERVIEW
- TABLE 950 FILTROX AG: COMPANY OVERVIEW
- TABLE 951 MEMBRANE SOLUTIONS: COMPANY OVERVIEW
- TABLE 952 IMPACT ANALYSIS OF SUPPLY-SIDE AND DEMAND-SIDE FACTORS
List of Figures
- FIGURE 1 PHARMACEUTICAL FILTRATION MARKET SEGMENTATION & REGIONAL SCOPE
- FIGURE 2 PHARMACEUTICAL FILTRATION MARKET: YEARS CONSIDERED
- FIGURE 3 MARKET SCENARIO
- FIGURE 4 GLOBAL PHARMACEUTICAL FILTRATION MARKET, 2023-2030
- FIGURE 5 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN PHARMACEUTICAL FILTRATION MARKET, 2020-2025
- FIGURE 6 DISRUPTIONS INFLUENCING GROWTH OF PHARMACEUTICAL FILTRATION MARKET
- FIGURE 7 HIGH-GROWTH SEGMENTS IN PHARMACEUTICAL FILTRATION MARKET, 2025-2030
- FIGURE 8 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN PHARMACEUTICAL FILTRATION MARKET DURING FORECAST PERIOD
- FIGURE 9 ADOPTION OF CONTINUOUS MANUFACTURING PROCESS TO DRIVE MARKET
- FIGURE 10 STERILE FILTRATION SEGMENT ACCOUNTED FOR LARGEST SHARE OF NORTH AMERICA MARKET IN 2024
- FIGURE 11 CHINA TO REGISTER HIGHEST GROWTH DURING FORECAST PERIOD
- FIGURE 12 PHARMACEUTICAL FILTRATION MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 13 PHARMACEUTICAL FILTRATION MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 14 PHARMACEUTICAL FILTRATION MARKET: VALUE CHAIN ANALYSIS
- FIGURE 15 PHARMACEUTICAL FILTRATION MARKET: ECOSYSTEM ANALYSIS
- FIGURE 16 AVERAGE SELLING PRICE TREND OF KEY PLAYERS, BY PRODUCT, 2022-2024
- FIGURE 17 PHARMACEUTICAL FILTRATION MARKET: TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- FIGURE 18 INVESTOR DEALS AND FUNDING, 2023-2025 (USD MILLION)
- FIGURE 19 TOP APPLICANTS/OWNERS (COMPANIES) FOR PHARMACEUTICAL FILTRATION PATENTS, 2014-2024
- FIGURE 20 IMPACT OF AI/GEN AI ON PHARMACEUTICAL FILTRATION MARKET
- FIGURE 21 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS, BY PRODUCT
- FIGURE 22 KEY BUYING CRITERIA, BY END USER
- FIGURE 23 NORTH AMERICA: PHARMACEUTICAL FILTRATION MARKET SNAPSHOT
- FIGURE 24 NIH FUNDING IN LAST 10 YEARS, 2015-2025
- FIGURE 25 ASIA PACIFIC: PHARMACEUTICAL FILTRATION MARKET SNAPSHOT
- FIGURE 26 PHARMACEUTICAL FILTRATION MARKET: REVENUE ANALYSIS OF KEY PLAYERS, 2020-2024 (USD BILLION)
- FIGURE 27 PHARMACEUTICAL FILTRATION MARKET SHARE ANALYSIS, 2024
- FIGURE 28 PHARMACEUTICAL FILTRATION MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2024
- FIGURE 29 PHARMACEUTICAL FILTRATION MARKET: COMPANY FOOTPRINT, 2024
- FIGURE 30 PHARMACEUTICAL FILTRATION MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2024
- FIGURE 31 EV/EBITDA OF KEY VENDORS
- FIGURE 32 YEAR-TO-DATE (YTD) PRICE, TOTAL RETURN, AND 5-YEAR STOCK BETA OF KEY VENDORS
- FIGURE 33 BRAND/PRODUCT COMPARATIVE ANALYSIS, BY PRODUCT
- FIGURE 34 MERCK KGAA: COMPANY SNAPSHOT (2024)
- FIGURE 35 DANAHER CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 36 SARTORIUS AG: COMPANY SNAPSHOT (2024)
- FIGURE 37 SOLVENTUM: COMPANY SNAPSHOT (2024)
- FIGURE 38 PARKER HANNIFIN CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 39 REPLIGEN CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 40 EATON CORPORATION PLC: COMPANY SNAPSHOT (2024)
- FIGURE 41 THERMO FISHER SCIENTIFIC, INC.: COMPANY SNAPSHOT (2024)
- FIGURE 42 DONALDSON COMPANY, INC.: COMPANY SNAPSHOT (2024)
- FIGURE 43 PORVAIR PLC: COMPANY SNAPSHOT (2024)
- FIGURE 44 ALFA LAVAL CORPORATE AB: COMPANY SNAPSHOT (2024)
- FIGURE 45 CORNING INCORPORATED: COMPANY SNAPSHOT (2024)
- FIGURE 46 MANN+HUMMEL: COMPANY SNAPSHOT (2024)
- FIGURE 47 SAINT-GOBAIN: COMPANY SNAPSHOT (2024)
- FIGURE 48 STERIS PLC: COMPANY SNAPSHOT (2024)
- FIGURE 49 ASAHI KASEI CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 50 RESEARCH DESIGN
- FIGURE 51 PHARMACEUTICAL FILTRATION MARKET: BREAKDOWN OF PRIMARIES
- FIGURE 52 PHARMACEUTICAL FILTRATION MARKET SIZE ESTIMATION (SUPPLY-SIDE ANALYSIS), 2024
- FIGURE 53 MARKET SIZE ESTIMATION: APPROACH 1 (COMPANY REVENUE ANALYSIS-BASED ESTIMATION), 2024
- FIGURE 54 REVENUE SHARE ANALYSIS: ILLUSTRATIVE EXAMPLE OF SARTORIUS AG (2024)
- FIGURE 55 MARKET SIZE VALIDATION FROM PRIMARY SOURCES
- FIGURE 56 MARKET SIZE ESTIMATION METHODOLOGY: TOP-DOWN APPROACH
- FIGURE 57 PHARMACEUTICAL FILTRATION MARKET: CAGR PROJECTIONS
- FIGURE 58 DATA TRIANGULATION METHODOLOGY










