 |
市场调查报告书
商品编码
1950784
全球过敏诊断市场按产品/服务、检测类型、过敏原、最终用户和地区划分-预测至2031年Allergy Diagnostic Market by Product & Service (Consumables, Instruments [Immunoassay Analyzers, Luminometers]), Test Type (In Vivo Tests, In Vitro Tests), Allergen (Food Allergens), End User (Hospital-Based Laboratories) - Global Forecast to 2031 |
||||||
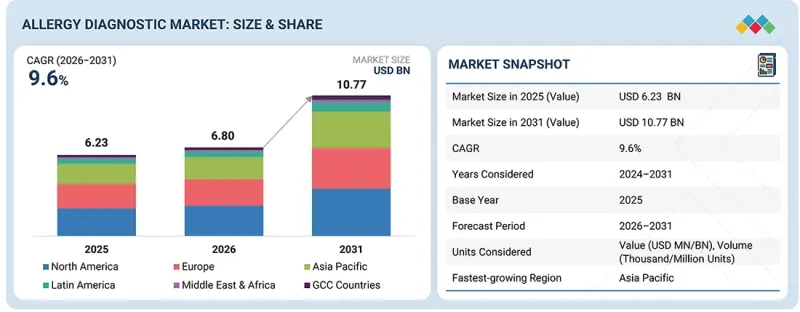
预计到 2030 年,过敏诊断市场规模将从 2025 年的 68 亿美元成长到 107.7 亿美元,预测期内复合年增长率为 9.6%。
| 调查范围 | |
|---|---|
| 调查期 | 2024-2031 |
| 基准年 | 2025 |
| 预测期 | 2026-2031 |
| 目标单元 | 金额(十亿美元) |
| 部分 | 按服务、测试类型、过敏原、最终用户、地区 |
| 目标区域 | 北美、欧洲、亚太地区、拉丁美洲、中东和非洲、海湾合作委员会国家 |
过敏诊断领域的技术创新是推动成长的主要动力,使检测更加准确、快速和全面。虽然传统的诊断方法,例如皮肤点刺试验 (SPT) 和单一过敏原血清 IgE 检测仍然有用,但多重免疫检测、组件解析诊断 (CRD) 和分子过敏等先进平台在许多方面已经超越了它们。多重平台可以从单一检体同时检测数十种甚至数百种过敏原的 IgE 反应,节省患者的时间、金钱和精力。 CRD 更进一步,能够辨识过敏原中导致致敏的确切蛋白质分子。这提高了检测的临床效用和风险分层能力。例如,它们可以区分无害的花粉致敏和交叉反应,而交叉反应是严重过敏反应的预测指标。
分子诊断在个人化医疗中也发挥关键作用,它能产生个人化的致敏谱图,供医生用于制定个人化的免疫治疗方案。自动化和与电子健康记录的数位化整合简化了临床检查室流程,并支持大规模筛检计画的实施。可在医生诊室和药房快速获得检测结果的照护现场(POC)创新技术,提高了患者的便利性和医疗效率。此外,人工智慧驱动的分析工具正在开发中,以辅助解读复杂的过敏原谱。这些创新不仅提高了检测性能和患者体验,也代表着一个长期的市场拓展,在这个市场中,诊断准确性将成为医疗服务的标准。
就产品和服务而言,全球过敏性疾病的增加是耗材在过敏诊断市场占据主要份额的原因。城市爆炸式增长、环境恶化、生活方式改变、饮食习惯转变以及全球暖化等多种因素共同导致了食物过敏、过敏性鼻炎、气喘和异位性皮肤炎的增加。由此导致的患者数量增加,进而推动了诊断测试数量的成长,尤其是高度依赖耗材的检查室体外诊断测试。每次诊断都需要大量的试剂和过敏原特异性试剂盒,这进一步增加了对耗材的需求。此外,由于儿童和老年人群最容易患过敏症,且通常需要重复进行确诊测试,因此在全球疾病负担日益加重的情况下,耗材仍然是受影响最大且增长最快的收入来源,尤其是在亚太地区和新兴市场。
在所有检测类型中,体内检测推广应用的关键驱动因素之一是患者只需一次就诊即可在15至30分钟内获得检测结果。这种即时回馈使医生能够识别过敏原、为患者提供咨询并立即启动治疗方案。与需要检查室处理且週转时间更长的体外检测相比,体内检测的工作流程更有效率,诊断速度更快。除了提高患者满意度外,快速诊断对于减少患者復诊次数也至关重要。这些因素使得体内检测在门诊和医院环境中更具吸引力,提高了其使用率和市场份额。
北美地区占据过敏诊断市场最大份额。该地区拥有完善的医疗保健生态系统,极大地促进了过敏诊断的广泛应用。这里汇集了许多配备先进检测设备的医院、专业的过敏诊所、诊断检查室和参考检查室。自动化免疫检测设备、多重检测系统和集中式检测服务的存在,确保了检测的高准确性和速度。此外,诊断流程已与临床工作流程深度整合,使医生能够轻鬆地将过敏检测作为标准诊疗项目,从而巩固了市场领导的优势。
过敏诊断市场的主要企业包括 Thermo Fisher Scientific Inc.(美国)、Siemens Healthineers AG(德国)、Danaher Corporatio(美国)、Minaris Medical America, Inc.(美国)、Omega Diagnostics Group Plc(英国)、bioMerieux SA(法国)、Romer Labs AG(德国)、HollisterStier Allergy(美国)、Eurofins Scientific(卢森堡)和 Stallergenes Greer(英国)。
调查对象
本报告评估了过敏诊断市场,并估算了其在产品和服务、检测类型、过敏原、最终用户和地区等各个细分市场的规模和未来成长潜力。报告还提供了竞争分析、公司简介、产品组合、最新发展动态以及主要企业的关键市场策略。
为什么要购买这份报告?
本报告提供了整体过敏诊断市场及其细分市场预计收入的宝贵数据,对市场领导企业和新参与企业均有裨益。它帮助相关人员了解竞争格局,并获得有效定位业务和製定合适打入市场策略的见解。此外,该报告还帮助相关人员了解市场趋势,包括市场中的关键驱动因素、挑战、障碍和机会。
本报告深入分析了以下内容:
- 对关键驱动因素(过敏性疾病的高发病率和沈重的经济负担、人们对过敏的认识不断提高、环境污染程度加剧、医疗保险系统对过敏诊断的覆盖范围)、阻碍因素(过敏诊断设备成本高、缺乏知识和过敏测试方法实施不力、医疗服务获取有限)、机会(医疗服务获取有限、将过敏诊断诊断)和实验专家的研究
- 产品改良与创新:全球过敏诊断市场产品发布详细资讯及预测趋势
- 市场发展:按产品/服务、检测类型、过敏原、最终用户和地区,对盈利且不断增长的市场进行详细洞察和分析
- 市场多元化:提供全球过敏诊断市场新产品发布、市场扩张、最新发展和投资趋势的全面信息
- 竞争格局分析:对全球过敏诊断市场主要竞争对手的市场份额、成长计画、产品组合和产能进行深入评估
目录
第一章 引言
第二章执行摘要
第三章重要考察
第四章 市场概览
- 市场动态
- 司机
- 抑制因素
- 机会
- 任务
- 未满足的需求和閒置频段
- 相互关联的市场与跨产业机会
- 一级/二级/三级公司的策略性倡议
第五章 产业趋势
- 波特五力分析
- 宏观经济展望
- 供应链分析
- 价值链分析
- 生态系分析
- 定价分析
- 贸易分析
- 2026-2027 年主要会议和活动
- 影响客户业务的趋势/颠覆性因素
- 投资和资金筹措方案
- 案例研究分析
- 2025年美国关税对过敏诊断市场的影响
第六章:技术进步、人工智慧的影响、专利、创新与未来应用
- 关键新兴技术
- 互补技术
- 技术/产品蓝图
- 专利分析
- 未来应用
- 人工智慧/生成式人工智慧对过敏诊断市场的影响
第七章 监理环境
- 地方法规和合规性
- 监管机构、政府机构和其他组织
- 业界标准
第八章:顾客状况与购买行为
- 决策流程
- 主要相关人员和采购标准
- 招募障碍和内部挑战
- 来自各个终端使用者产业的未满足需求
- 市场盈利
第九章 过敏诊断市场(依产品/服务划分)
- 消耗品
- 装置
- 服务
第十章 过敏诊断市场(依检测类型划分)
- 体内试验
- 体外试验
第十一章 过敏诊断市场(依过敏原分类)
- 吸入性过敏原
- 食物过敏原
- 药物过敏原
- 其他的
第十二章 过敏诊断市场(依最终用户划分)
- 院内检查室
- 诊断检查室
- 学术研究机构
- 其他的
第十三章 过敏诊断市场(按地区划分)
- 北美洲
- 我们
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 义大利
- 西班牙
- 其他的
- 亚太地区
- 中国
- 日本
- 印度
- 韩国
- 澳洲
- 其他的
- 拉丁美洲
- 巴西
- 墨西哥
- 其他的
- 中东和非洲
- 海湾合作委员会国家
- 医疗成本上涨推动市场成长
第十四章 竞争格局
- 主要参与企业的策略/优势
- 2021-2025年收入分析
- 市占率分析
- 公司估值矩阵:主要参与企业,2025 年
- 竞争力评估矩阵:Start-Ups/中小企业,2025 年
- 估值和财务指标
- 品牌/产品对比
- 竞争场景
第十五章:公司简介
- 主要参与企业
- THERMO FISHER SCIENTIFIC INC.
- DANAHER CORPORATION
- SIEMENS HEALTHINEERS AG
- CANON, INC.(MINARIS MEDICAL AMERICA, INC.)
- REVVITY, INC.(EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG)
- EUROFINS SCIENTIFIC
- BIOMERIEUX SA
- DSM ROYAL(ROMER LABS DIVISION HOLDING GMBH)
- HOLLISTERSTIER ALLERGY(JUBILANT PHARMA)
- OMEGA DIAGNOSTICS GROUP PLC
- STALLERGENES GREER LTD.
- 其他公司
- HOB BIOTECH GROUP CORP., LTD.
- HYCOR BIOMEDICAL
- LINCOLN DIAGNOSTICS, INC.
- R-BIOPHARM AG
- ASTRA BIOTECH GMBH
- ERBA GROUP
- AESKU.GROUP GMBH
- ACON LABORATORIES, INC.
- ALCIT INDIA PVT. LTD.
- BIOPANDA REAGENTS LTD.
- BIOSIDE SRL
- CREATIVE DIAGNOSTIC MEDICARE PVT. LTD.
- DST DIAGNOSTISCHE SYSTEME & TECHNOLOGIEN GMBH
- DR. FOOKE LABORATORIEN GMBH
第十六章调查方法
第十七章附录
The allergy diagnostic market is projected to reach USD 10.77 billion by 2030 from USD 6.80 billion in 2025, at a CAGR of 9.6% during the forecast period.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD billion) |
| Segments | Product & Service, Test Type, Allergen, End user, and Region |
| Regions covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa, and GCC Countries |
Technological innovation in allergy diagnostics is a key driver of growth, making testing more accurate, faster, and comprehensive. Traditional diagnostic methods such as skin prick tests (SPT) and single, allergen blood IgE tests are still of use, but the advanced platforms like multiplex immunoassays, component resolved diagnostics (CRD), and molecular allergology have surpassed them in a variety of ways. Multiplex platforms allow simultaneous detection of IgE responses to dozens or even hundreds of allergens in a single sample, thereby decreasing the time, cost, and effort for the patient. CRD is a step further in that it pinpoints the exact protein molecules within the allergens that are the cause of sensitization, thus increasing how clinically relevant the test is and risk stratification; e.g. , it can differentiate between innocent pollen sensitization and cross-reactivity that is a predictor of severe reactions.

Molecular diagnostics are also instrumental in personalized medicine-they generate the individual sensitization profile map, which doctors use for the personalization of immunotherapy treatments. Automation and digital integration with electronic health records facilitate the working processes in clinical laboratories and are also of assistance when it comes to undertaking large-scale screening programs. Point-of-care (POC) innovations that deliver quick results right in doctors' or pharmacies' offices enhance patients' convenience and the effectiveness of medical care. Apart from that, AI-powered interpretation tools are being developed to help doctors make sense of complicated allergen profiles. Such technological innovations not only improve test performance and patient experience but also represent a long-term market expansion in which diagnostic precision becomes the standard of care.
Based on product & service: The global increase in allergic diseases is a significant factor that explains why consumables have the leading share in the allergy diagnostic market. A combination of factors such as the explosive growth of cities, the deterioration of the environment, alterations in lifestyles, changes in diet, and global warming has, among other things, led to a rise in food allergies, allergic rhinitis, asthma, and atopic dermatitis. Correspondingly, the growing patient population results in higher diagnostic testing volumes, particularly laboratory-based in vitro tests, which are highly dependent on consumables. Numerous reagents and allergen-specific kits are required for each diagnostic episode; thus, the demand for consumables is further increased. Besides that, both children and elderly pediatric and geriatric populations are among the most susceptible to allergies and, therefore, often necessitate repeated and confirmatory tests. Consumables remain the most directly affected and fastest-growing source of revenue as the disease burden escalates worldwide, especially in the Asia Pacific and emerging markets.
Based on test type: A powerful incentive for the widespread adoption of in vivo testing, among other things, is the possibility of delivering test results within 15-30 minutes during a single patient visit. Such instant feedback enables doctors to identify allergies, advise patients, and start therapy plans right away. In vivo tests offer a workflow that is much more efficient, with rapid diagnosis compared to in vitro tests, which are subjected to laboratory processing and require a longer turnaround time. Besides patient satisfaction, fast diagnosis is important because it reduces patient revisits, both of which factors make the tests more appealing to outpatient clinics and hospital settings, thereby leading to higher utilization and market share for these tests.
North America holds the largest market share in the allergy diagnostic market. The North American region has a sophisticated healthcare ecosystem that strongly promotes the use of allergy diagnostics at scale. The area is well-equipped with a variety of hospitals, specialty allergy clinics, diagnostic laboratories, and reference labs equipped with advanced testing equipment. The presence of automated immunoassay analyzers, multiplex testing systems, and centralized lab services ensures high test accuracy and speed. In addition, the diagnostics are so well integrated into clinical workflows that doctors can easily order allergy tests as standard of care, thereby supporting the market leader's dominance.
A breakdown of the primary participants (supply-side) for the allergy diagnostic market referred to in this report is provided below:
- By Company Type: Tier 1: 34%, Tier 2: 38%, and Tier 3: 28%
- By Designation: C-level: 26%, Director Level: 35%, and Others: 39%
- By Region: North America: 35%, Europe: 30%, Asia Pacific: 25%, Latin America: 6%, Middle East & Africa: 2%, GCC Countries: 3%
Prominent players in the allergy diagnostic market are Thermo Fisher Scientific Inc. (US), Siemens Healthineers AG (Germany), Danaher Corporation (US), Minaris Medical America, Inc. (US), Omega Diagnostics Group Plc (UK), bioMerieux SA (France), Romer Labs Division Holding (Austria), EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG (Germany), HollisterStier Allergy (US), Eurofins Scientific (Luxembourg), and Stallergenes Greer (UK), among others.
Research Coverage
The report evaluates the allergy diagnostic market and estimates its size and future growth potential across various segments, including product & service, test type, allergen, end user, and region. The report also includes a competitive analysis of the major players in this market, along with company profiles, product offerings, recent developments, and key market strategies.
Reasons to Buy the Report
The report provides valuable data on estimated revenue figures for the overall allergy diagnostic market and its subsegments, benefiting both market leaders and new entrants. It helps stakeholders understand the competitive landscape and gain insights for effectively positioning their businesses and developing appropriate go-to-market strategies. Additionally, the report offers stakeholders an understanding of market trends, including key drivers, challenges, obstacles, and opportunities within the market.
This report provides insights into the following points:
- Analysis of key drivers (High incidence and heavy economic burden of allergic diseases, Increasing awareness about allergies, Rising environmental pollution levels, Coverage of allergy diagnosis under health insurance schemes), restraints (Premium cost of allergy diagnostic instruments, Lack of adequate knowledge and poor implementation of allergy testing methods, Limited access to healthcare services), opportunities (Limited access to healthcare services, Integration of artificial intelligence in allergy diagnosis), and challenges (Shortage of allergists and lack of training programs, Diagnostic challenges in allergic patients)
- Product Enhancement/Innovation: Comprehensive details about product launches and anticipated trends in the global allergy diagnostic market
- Market Development: Thorough knowledge and analysis of the profitable rising markets by product & service, test type, allergen, end user, and region
- Market Diversification: Comprehensive information about newly launched products, expanding markets, current advancements, and investments in the global allergy diagnostic market
- Competitive Assessment: Thorough evaluation of the market shares, growth plans, offerings, and capacities of the major competitors in the global allergy diagnostic market
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.2.1 INCLUSIONS & EXCLUSIONS
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 YEARS CONSIDERED
- 1.3.3 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS & MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: SHARE INSIGHTS AND DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS SHAPING MARKET
- 2.4 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 ALLERGY DIAGNOSTIC MARKET OVERVIEW
- 3.2 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY END USER AND COUNTRY
- 3.3 ALLERGY DIAGNOSTIC MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- 3.4 ALLERGY DIAGNOSTIC MARKET: REGIONAL MIX
- 3.5 ALLERGY DIAGNOSTIC MARKET: DEVELOPED VS. EMERGING ECONOMIES
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 High incidence and heavy economic burden of allergic diseases
- 4.2.1.2 Increasing awareness about allergies
- 4.2.1.3 Rising environmental pollution levels
- 4.2.1.4 Coverage of allergy diagnosis under health insurance schemes
- 4.2.2 RESTRAINTS
- 4.2.2.1 Premium cost of allergy diagnostic instruments
- 4.2.2.2 Lack of adequate knowledge and poor implementation of allergy testing methods
- 4.2.2.3 Limited access to healthcare services
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Use of mHealth in allergy diagnosis
- 4.2.3.2 Integration of artificial intelligence in allergy diagnosis
- 4.2.4 CHALLENGES
- 4.2.4.1 Shortage of allergists and lack of training programs
- 4.2.4.2 Diagnostic challenges in allergic patients
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS & WHITE SPACES
- 4.3.1 UNMET NEEDS IN ALLERGY DIAGNOSTIC MARKET
- 4.3.2 WHITE SPACE OPPORTUNITIES
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.4.1 INTERCONNECTED MARKETS
- 4.4.2 CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 INTENSITY OF COMPETITIVE RIVALRY
- 5.1.2 BARGAINING POWER OF SUPPLIERS
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 THREAT OF SUBSTITUTES
- 5.1.5 THREAT OF NEW ENTRANTS
- 5.2 MACROECONOMIC OUTLOOK
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS AND FORECAST
- 5.2.3 TRENDS IN GLOBAL IVD INDUSTRY
- 5.2.4 TRENDS IN GLOBAL IMMUNOASSAY INDUSTRY
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.4 VALUE CHAIN ANALYSIS
- 5.5 ECOSYSTEM ANALYSIS
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE OF ALLERGY DIAGNOSTIC PRODUCTS, BY PRODUCT & SERVICE, 2025
- 5.6.2 AVERAGE SELLING PRICE TREND OF ALLERGY DIAGNOSTIC PRODUCTS, BY REGION, 2024-2026
- 5.7 TRADE ANALYSIS
- 5.7.1 IMPORT DATA FOR HS CODE 902750, 2020-2024
- 5.7.2 EXPORT DATA FOR HS CODE 902750, 2020-2024
- 5.8 KEY CONFERENCES & EVENTS, 2026-2027
- 5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS
- 5.10 INVESTMENT & FUNDING SCENARIO
- 5.11 CASE STUDY ANALYSIS
- 5.11.1 EXPANDING ACCESS TO ALLERGY DIAGNOSTICS IN EMERGING MARKETS
- 5.11.2 SCALING HIGH-THROUGHPUT ALLERGY TESTING IN CENTRALIZED LABORATORIES
- 5.11.3 IMPROVING PEDIATRIC ALLERGY DIAGNOSIS THROUGH MULTIPLEX TESTING
- 5.12 IMPACT OF 2025 US TARIFF ON ALLERGY DIAGNOSTIC MARKET
- 5.12.1 INTRODUCTION
- 5.12.2 KEY TARIFF RATES
- 5.12.3 PRICE IMPACT ANALYSIS
- 5.12.4 IMPACT ON COUNTRIES/REGIONS
- 5.12.4.1 North America
- 5.12.4.2 Europe
- 5.12.4.3 Asia Pacific
- 5.12.5 END-USE INDUSTRY IMPACT
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 KEY EMERGING TECHNOLOGIES
- 6.1.1 SPECIFIC IGE IMMUNOASSAYS
- 6.1.2 SKIN PRICK TESTING (SPT)
- 6.2 COMPLEMENTARY TECHNOLOGIES
- 6.2.1 AUTOMATED IMMUNOASSAY ANALYZERS
- 6.3 TECHNOLOGY/PRODUCT ROADMAP
- 6.3.1 SHORT-TERM (2025-2027) | FOUNDATION & EARLY COMMERCIALIZATION
- 6.3.2 MID-TERM (2027-2030) | EXPANSION & STANDARDIZATION
- 6.3.3 LONG-TERM (2030-2035+) | MASS COMMERCIALIZATION & DISRUPTION
- 6.4 PATENT ANALYSIS
- 6.4.1 INSIGHTS: JURISDICTION AND TOP APPLICANT ANALYSIS
- 6.5 FUTURE APPLICATIONS
- 6.5.1 ADVANCED MULTIPLEX AND PERSONALIZED ALLERGY DIAGNOSTICS
- 6.5.2 DIGITAL & AI-ENABLED ALLERGY MONITORING PLATFORMS
- 6.5.3 AI-INTEGRATED DIGITAL SURGICAL ECOSYSTEMS FOR IOLS
- 6.6 IMPACT OF AI/GEN AI ON ALLERGY DIAGNOSTIC MARKET
- 6.6.1 TOP USE CASES AND MARKET POTENTIAL
- 6.6.2 CASE STUDIES OF AI IMPLEMENTATION
- 6.6.3 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS
- 6.6.4 CLIENTS' READINESS TO ADOPT GENERATIVE AI
7 REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS & COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 INDUSTRY STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 KEY STAKEHOLDERS AND BUYING EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 KEY BUYING CRITERIA
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.5 MARKET PROFITABILITY
- 8.5.1 REVENUE POTENTIAL
- 8.5.2 COST DYNAMICS
- 8.5.3 MARGIN OPPORTUNITIES IN KEY APPLICATIONS
9 ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE
- 9.1 INTRODUCTION
- 9.2 CONSUMABLES
- 9.2.1 RECURRENT USE OF CONSUMABLES FOR ALLERGY DIAGNOSTICS TO DRIVE MARKET
- 9.3 INSTRUMENTS
- 9.3.1 IMMUNOASSAY ANALYZERS
- 9.3.1.1 High preference for testing to boost market demand
- 9.3.2 LUMINOMETERS
- 9.3.2.1 Convenience and ease of use to propel market growth
- 9.3.3 OTHER INSTRUMENTS
- 9.3.1 IMMUNOASSAY ANALYZERS
- 9.4 SERVICES
- 9.4.1 RISING NEED TO ENSURE EFFECTIVE UTILIZATION OF ALLERGY DIAGNOSTIC PRODUCTS TO SUPPORT MARKET GROWTH
10 ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE
- 10.1 INTRODUCTION
- 10.2 IN VIVO TESTS
- 10.2.1 SKIN PRICK TESTS
- 10.2.1.1 Skin prick tests to be most widely preferred in vivo allergy tests
- 10.2.2 PATCH TESTS
- 10.2.2.1 Rapid diagnosis of contact dermatitis to boost market demand
- 10.2.3 OTHER IN VIVO TESTS
- 10.2.1 SKIN PRICK TESTS
- 10.3 IN VITRO TESTS
- 10.3.1 ABILITY TO IDENTIFY ALLERGEN-SPECIFIC IGE MOLECULES IN PATIENT SERUM TO DRIVE MARKET
11 ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN
- 11.1 INTRODUCTION
- 11.2 INHALED ALLERGENS
- 11.2.1 HIGH INCIDENCE OF ASTHMA TO BOOST MARKET GROWTH
- 11.3 FOOD ALLERGENS
- 11.3.1 HIGH PREVALENCE OF FOOD-RELATED ALLERGIES AMONG YOUNG CHILDREN TO SUPPORT MARKET GROWTH
- 11.4 DRUG ALLERGENS
- 11.4.1 INCREASING INCIDENCE OF DRUG ALLERGIES AND ADVERSE REACTIONS TO DRIVE MARKET
- 11.5 OTHER ALLERGENS
12 ALLERGY DIAGNOSTIC MARKET, BY END USER
- 12.1 INTRODUCTION
- 12.2 HOSPITAL-BASED LABORATORIES
- 12.2.1 HOSPIATAL-BASED LABORATORIES TO BE MORE ACCESSIBLE TO PATIENTS AND OFFER RAPID RESULTS
- 12.3 DIAGNOSTIC LABORATORIES
- 12.3.1 DIAGNOSTIC LABORATORIES TO PIVOTAL FOR DIAGNOSIS AND MANAGEMENT OF ALLERGIES
- 12.4 ACADEMIC RESEARCH INSTITUTES
- 12.4.1 RISING NUMBER OF INSTITUTES FOR ALLERGY TESTING TRAINING TO SUPPORT MARKET GROWTH
- 12.5 OTHER END USERS
13 ALLERGY DIAGNOSTIC MARKET, BY REGION
- 13.1 INTRODUCTION
- 13.2 NORTH AMERICA
- 13.2.1 US
- 13.2.1.1 US to dominate North American allergy diagnostic market
- 13.2.2 CANADA
- 13.2.2.1 Rising funding activities in allergy diagnostics to drive market
- 13.2.1 US
- 13.3 EUROPE
- 13.3.1 GERMANY
- 13.3.1.1 Increasing prevalence of allergic conditions and rising presence of allergy diagnostic product manufacturers to aid market growth
- 13.3.2 UK
- 13.3.2.1 Increasing prevalence of food anaphylaxis to support market growth
- 13.3.3 FRANCE
- 13.3.3.1 Increasing reimbursements for allergy testing to spur market growth
- 13.3.4 ITALY
- 13.3.4.1 Presence of allergy diagnostic organizations to support increased demand for products
- 13.3.5 SPAIN
- 13.3.5.1 Government initiatives and healthcare infrastructural development to drive market
- 13.3.6 REST OF EUROPE
- 13.3.1 GERMANY
- 13.4 ASIA PACIFIC
- 13.4.1 CHINA
- 13.4.1.1 Rising allergy prevalence and growing research activities to augment market growth
- 13.4.2 JAPAN
- 13.4.2.1 Increasing prevalence of allergies among children to propel demand for allergy diagnostics
- 13.4.3 INDIA
- 13.4.3.1 Limited awareness regarding allergy management to limit market growth
- 13.4.4 SOUTH KOREA
- 13.4.4.1 Rising allergy burden and high diagnostic uptake to fuel South Korean market growth
- 13.4.5 AUSTRALIA
- 13.4.5.1 High prevalence of food allergies and major lifestyle changes to drive market
- 13.4.6 REST OF ASIA PACIFIC
- 13.4.1 CHINA
- 13.5 LATIN AMERICA
- 13.5.1 BRAZIL
- 13.5.1.1 High prevalence of childhood asthma to augment market growth
- 13.5.2 MEXICO
- 13.5.2.1 Growing focus on awareness training programs to boost market demand
- 13.5.3 REST OF LATIN AMERICA
- 13.5.1 BRAZIL
- 13.6 MIDDLE EAST & AFRICA
- 13.7 GCC COUNTRIES
- 13.7.1 RISING HEALTHCARE SPENDING TO SPUR MARKET GROWTH
14 COMPETITIVE LANDSCAPE
- 14.1 INTRODUCTION
- 14.2 KEY PLAYER STRATEGY/RIGHT TO WIN
- 14.3 REVENUE ANALYSIS, 2021-2025
- 14.4 MARKET SHARE ANALYSIS
- 14.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 14.5.1 STARS
- 14.5.2 EMERGING LEADERS
- 14.5.3 PERVASIVE PLAYERS
- 14.5.4 PARTICIPANTS
- 14.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 14.5.5.1 Company footprint
- 14.5.5.2 Region footprint
- 14.5.5.3 Product & service footprint
- 14.5.5.4 Test type footprint
- 14.5.5.5 Allergen footprint
- 14.6 COMPETITIVE EVALUATION MATRIX: STARTUPS/SMES, 2025
- 14.6.1 PROGRESSIVE COMPANIES
- 14.6.2 RESPONSIVE COMPANIES
- 14.6.3 DYNAMIC COMPANIES
- 14.6.4 STARTING BLOCKS
- 14.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2025
- 14.6.5.1 Detailed list of key startups/SMEs
- 14.6.5.2 Competitive benchmarking of startups/SMEs
- 14.7 COMPANY VALUATION & FINANCIAL METRICS
- 14.7.1 FINANCIAL METRICS
- 14.7.2 COMPANY VALUATION
- 14.8 BRAND/PRODUCT COMPARISON
- 14.9 COMPETITIVE SCENARIO
- 14.9.1 PRODUCT LAUNCHES & APPROVALS
- 14.9.2 DEALS
- 14.9.3 EXPANSIONS
15 COMPANY PROFILES
- 15.1 KEY PLAYERS
- 15.1.1 THERMO FISHER SCIENTIFIC INC.
- 15.1.1.1 Business overview
- 15.1.1.2 Products offered
- 15.1.1.3 Recent developments
- 15.1.1.3.1 Product launches & approvals
- 15.1.1.3.2 Deals
- 15.1.1.4 MnM view
- 15.1.1.4.1 Right to win
- 15.1.1.4.2 Strategic choices
- 15.1.1.4.3 Weaknesses & competitive threats
- 15.1.2 DANAHER CORPORATION
- 15.1.2.1 Business overview
- 15.1.2.2 Products offered
- 15.1.2.3 MnM view
- 15.1.2.3.1 Right to win
- 15.1.2.3.2 Strategic choices
- 15.1.2.3.3 Weaknesses & competitive threats
- 15.1.3 SIEMENS HEALTHINEERS AG
- 15.1.3.1 Business overview
- 15.1.3.2 Products offered
- 15.1.3.3 MnM view
- 15.1.3.3.1 Right to win
- 15.1.3.3.2 Strategic choices
- 15.1.3.3.3 Weaknesses & competitive threats
- 15.1.4 CANON, INC. (MINARIS MEDICAL AMERICA, INC.)
- 15.1.4.1 Business overview
- 15.1.4.2 Products offered
- 15.1.4.3 Recent developments
- 15.1.4.3.1 Deals
- 15.1.4.4 MnM view
- 15.1.4.4.1 Right to win
- 15.1.4.4.2 Strategic choices
- 15.1.4.4.3 Weaknesses & competitive threats
- 15.1.5 REVVITY, INC. (EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG)
- 15.1.5.1 Business overview
- 15.1.5.2 Products offered
- 15.1.5.3 Recent developments
- 15.1.5.3.1 Deals
- 15.1.5.4 MnM view
- 15.1.5.4.1 Right to win
- 15.1.5.4.2 Strategic choices
- 15.1.5.4.3 Weaknesses & competitive threats
- 15.1.6 EUROFINS SCIENTIFIC
- 15.1.6.1 Business overview
- 15.1.6.2 Products offered
- 15.1.6.3 Recent developments
- 15.1.6.3.1 Expansions
- 15.1.7 BIOMERIEUX SA
- 15.1.7.1 Business overview
- 15.1.7.2 Products offered
- 15.1.8 DSM ROYAL (ROMER LABS DIVISION HOLDING GMBH)
- 15.1.8.1 Business overview
- 15.1.8.2 Products offered
- 15.1.8.3 Recent developments
- 15.1.8.3.1 Deals
- 15.1.9 HOLLISTERSTIER ALLERGY (JUBILANT PHARMA)
- 15.1.9.1 Business overview
- 15.1.9.2 Products offered
- 15.1.10 OMEGA DIAGNOSTICS GROUP PLC
- 15.1.10.1 Business overview
- 15.1.10.2 Products offered
- 15.1.11 STALLERGENES GREER LTD.
- 15.1.11.1 Business overview
- 15.1.11.2 Products offered
- 15.1.11.3 Recent developments
- 15.1.11.3.1 Deals
- 15.1.1 THERMO FISHER SCIENTIFIC INC.
- 15.2 OTHER PLAYERS
- 15.2.1 HOB BIOTECH GROUP CORP., LTD.
- 15.2.2 HYCOR BIOMEDICAL
- 15.2.3 LINCOLN DIAGNOSTICS, INC.
- 15.2.4 R-BIOPHARM AG
- 15.2.5 ASTRA BIOTECH GMBH
- 15.2.6 ERBA GROUP
- 15.2.7 AESKU.GROUP GMBH
- 15.2.8 ACON LABORATORIES, INC.
- 15.2.9 ALCIT INDIA PVT. LTD.
- 15.2.10 BIOPANDA REAGENTS LTD.
- 15.2.11 BIOSIDE S.R.L.
- 15.2.12 CREATIVE DIAGNOSTIC MEDICARE PVT. LTD.
- 15.2.13 DST DIAGNOSTISCHE SYSTEME & TECHNOLOGIEN GMBH
- 15.2.14 DR. FOOKE LABORATORIEN GMBH
16 RESEARCH METHODOLOGY
- 16.1 RESEARCH DATA
- 16.1.1 SECONDARY DATA
- 16.1.1.1 Key secondary sources
- 16.1.1.2 Key data from secondary sources
- 16.1.2 PRIMARY DATA
- 16.1.2.1 Objectives of primary research
- 16.1.2.2 Key data from primary sources
- 16.1.2.3 Key industry insights
- 16.1.2.4 Breakdown of primaries
- 16.1.1 SECONDARY DATA
- 16.2 MARKET SIZE ESTIMATION
- 16.2.1 SUPPLY-SIDE ANALYSIS (REVENUE SHARE ANALYSIS)
- 16.2.2 COMPANY INVESTOR PRESENTATIONS AND PRIMARY INTERVIEWS
- 16.2.3 TOP-DOWN APPROACH
- 16.2.4 DEMAND-SIDE ANALYSIS
- 16.3 DATA TRIANGULATION
- 16.4 MARKET SHARE ESTIMATION
- 16.5 STUDY ASSUMPTIONS
- 16.6 RISK ANALYSIS
- 16.7 RESEARCH LIMITATIONS
17 APPENDIX
- 17.1 DISCUSSION GUIDE
- 17.2 KNOWLEDGESTORE: MARKETSANDMARKETS. SUBSCRIPTION PORTAL
- 17.3 CUSTOMIZATION OPTIONS
- 17.4 RELATED REPORTS
- 17.5 AUTHOR DETAILS
List of Tables
- TABLE 1 ALLERGY DIAGNOSTIC MARKET: INCLUSIONS & EXCLUSIONS
- TABLE 2 STANDARD CURRENCY CONVERSION RATES (USD)
- TABLE 3 IMPACT OF PORTER'S FIVE FORCES IN ALLERGY DIAGNOSTIC MARKET
- TABLE 4 GDP PERCENTAGE CHANGE, BY COUNTRY, 2021-2030
- TABLE 5 AVERAGE SELLING PRICE OF ALLERGY DIAGNOSTIC PRODUCTS, BY PRODUCT & SERVICE, 2025 (USD)
- TABLE 6 AVERAGE PRICING TREND OF ALLERGY DIAGNOSTIC PRODUCTS, BY REGION, 2024-2026 (USD)
- TABLE 7 IMPORT DATA FOR HS CODE 902750, BY COUNTRY, 2020-2024 (USD THOUSAND)
- TABLE 8 EXPORT DATA FOR HS CODE 902750, BY COUNTRY, 2020-2024 (USD THOUSAND)
- TABLE 9 KEY MAJOR CONFERENCES AND EVENTS IN ALLERGY DIAGNOSTIC MARKET, JANUARY 2026-DECEMBER 2027
- TABLE 10 CASE STUDY 1: EXPANDING ACCESS TO ALLERGY DIAGNOSTICS IN EMERGING MARKETS
- TABLE 11 CASE STUDY 2: SCALING HIGH-THROUGHPUT ALLERGY TESTING IN CENTRALIZED LABORATORIES
- TABLE 12 CASE STUDY 3: IMPROVING PEDIATRIC ALLERGY DIAGNOSIS THROUGH MULTIPLEX TESTING
- TABLE 13 US-ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 14 KEY PRODUCT-RELATED TARIFF EFFECTIVE FOR ALLERGY DIAGNOSTIC
- TABLE 15 TOP USE CASES AND POTENTIAL IN ALLERGY DIAGNOSTIC MARKET
- TABLE 16 CASE STUDIES RELATED TO AI IMPLEMENTATION IN ALLERGY DIAGNOSTIC MARKET
- TABLE 17 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS IN ALLERGY DIAGNOSTIC MARKET
- TABLE 18 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 19 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 20 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 21 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 22 MIDDLE EAST & AFRICA AND GCC COUNTRIES: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 23 GLOBAL STANDARDS IN ALLERGY DIAGNOSTIC MARKET
- TABLE 24 INFLUENCE OF KEY STAKEHOLDERS ON BUYING PROCESS OF ALLERGY DIAGNOSTIC PRODUCTS
- TABLE 25 KEY BUYING CRITERIA FOR ALLERGY DIAGNOSTIC PRODUCTS
- TABLE 26 UNMET NEEDS IN ALLERGY DIAGNOSTIC MARKET
- TABLE 27 ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 28 ALLERGY DIAGNOSTIC CONSUMABLES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 29 ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 30 ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 31 ALLERGY DIAGNOSTIC INSTRUMENTS MARKET FOR IMMUNOASSAY ANALYZERS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 32 ALLERGY DIAGNOSTIC INSTRUMENTS MARKET FOR IMMUNOASSAY ANALYZERS, BY REGION, 2024-2031 (UNITS)
- TABLE 33 ALLERGY DIAGNOSTIC INSTRUMENTS MARKET FOR LUMINOMETERS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 34 ALLERGY DIAGNOSTIC INSTRUMENTS MARKET FOR LUMINOMETERS, BY REGION, 2024-2031 (UNITS)
- TABLE 35 OTHER ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 36 ALLERGY DIAGNOSTIC SERVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 37 ADVANTAGES AND LIMITATIONS OF ALLERGY TESTS
- TABLE 38 ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 39 ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 40 ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 41 ALLERGY DIAGNOSTIC MARKET FOR SKIN PRICK TESTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 42 ALLERGY DIAGNOSTIC MARKET FOR PATCH TESTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 43 ALLERGY DIAGNOSTIC MARKET FOR OTHER IN VIVO TESTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 44 ALLERGY DIAGNOSTIC MARKET FOR IN VITRO TESTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 45 ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 46 ALLERGY DIAGNOSTIC MARKET FOR INHALED ALLERGENS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 47 SPECIFIC FOOD-INDUCED ALLERGIC CONDITIONS
- TABLE 48 ALLERGY DIAGNOSTIC MARKET FOR FOOD ALLERGENS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 49 ALLERGY DIAGNOSTIC MARKET FOR DRUG ALLERGENS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 50 ALLERGY DIAGNOSTIC MARKET FOR OTHER ALLERGENS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 51 ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 52 ALLERGY DIAGNOSTIC MARKET FOR HOSPITAL-BASED LABORATORIES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 53 ALLERGY DIAGNOSTIC MARKET FOR DIAGNOSTIC LABORATORIES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 54 ALLERGY DIAGNOSTIC MARKET FOR ACADEMIC RESEARCH INSTITUTES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 55 ALLERGY DIAGNOSTIC MARKET FOR OTHER END USERS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 56 ALLERGY DIAGNOSTIC MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 57 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 58 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 59 NORTH AMERICA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 60 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 61 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 62 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 63 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 64 US: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 65 US: ALLERGY DIAGNOSTICS INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 66 US: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 67 US: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 68 US: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 69 US: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 70 CANADA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 71 CANADA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 72 CANADA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 73 CANADA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 74 CANADA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 75 CANADA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 76 EUROPE: ALLERGY DIAGNOSTIC MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 77 EUROPE: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 78 EUROPE: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 79 EUROPE: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 80 EUROPE: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 81 EUROPE: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 82 EUROPE: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 83 GERMANY: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 84 GERMANY: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 85 GERMANY: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 86 GERMANY: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 87 GERMANY: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 88 GERMANY: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 89 UK: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 90 UK: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 91 UK: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 92 UK: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 93 UK: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 94 UK: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 95 FRANCE: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 96 FRANCE: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 97 FRANCE: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 98 FRANCE: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 99 FRANCE: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 100 FRANCE: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 101 ITALY: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 102 ITALY: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 103 ITALY: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 104 ITALY: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 105 ITALY: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 106 ITALY: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 107 SPAIN: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 108 SPAIN: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 109 SPAIN: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 110 SPAIN: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 111 SPAIN: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 112 SPAIN: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 113 REST OF EUROPE: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 114 REST OF EUROPE: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 115 REST OF EUROPE: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 116 REST OF EUROPE: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 117 REST OF EUROPE: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 118 REST OF EUROPE: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 119 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 120 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 121 ASIA PACIFIC: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 122 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 123 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 124 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 125 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 126 CHINA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 127 CHINA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 128 CHINA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 129 CHINA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 130 CHINA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 131 CHINA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 132 JAPAN: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 133 JAPAN: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 134 JAPAN: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 135 JAPAN: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 136 JAPAN: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 137 JAPAN: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 138 INDIA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 139 INDIA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 140 INDIA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 141 INDIA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 142 INDIA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 143 INDIA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 144 SOUTH KOREA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 145 SOUTH KOREA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 146 SOUTH KOREA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 147 SOUTH KOREA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 148 SOUTH KOREA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 149 SOUTH KOREA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 150 AUSTRALIA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 151 AUSTRALIA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 152 AUSTRALIA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 153 AUSTRALIA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 154 AUSTRALIA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 155 AUSTRALIA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 156 REST OF ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 157 REST OF ASIA PACIFIC: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 158 REST OF ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 159 REST OF ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 160 REST OF ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 161 REST OF ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 162 LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 163 LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 164 LATIN AMERICA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 165 LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 166 LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 167 LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 168 LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 169 BRAZIL: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 170 BRAZIL: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 171 BRAZIL: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 172 BRAZIL: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 173 BRAZIL: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 174 BRAZIL: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 175 MEXICO: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 176 MEXICO: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 177 MEXICO: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 178 MEXICO: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 179 MEXICO: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 180 MEXICO: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 181 REST OF LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 182 REST OF LATIN AMERICA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 183 REST OF LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 184 REST OF LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 185 REST OF LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 186 REST OF LATIN AMERICA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 187 MIDDLE EAST & AFRICA: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 188 MIDDLE EAST & AFRICA: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 189 MIDDLE EAST & AFRICA: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 190 MIDDLE EAST & AFRICA: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 191 MIDDLE EAST & AFRICA: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 192 MIDDLE EAST & AFRICA: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 193 GCC COUNTRIES: ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, 2024-2031 (USD MILLION)
- TABLE 194 GCC COUNTRIES: ALLERGY DIAGNOSTIC INSTRUMENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 195 GCC COUNTRIES: ALLERGY DIAGNOSTIC MARKET, BY TEST TYPE, 2024-2031 (USD MILLION)
- TABLE 196 GCC COUNTRIES: ALLERGY DIAGNOSTIC MARKET FOR IN VIVO TESTS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 197 GCC COUNTRIES: ALLERGY DIAGNOSTIC MARKET, BY ALLERGEN, 2024-2031 (USD MILLION)
- TABLE 198 GCC COUNTRIES: ALLERGY DIAGNOSTIC MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 199 KEY DEVELOPMENTS IN ALLERGY DIAGNOSTIC MARKET, JANUARY 2021-JANUARY 2026
- TABLE 200 ALLERGY DIAGNOSTIC MARKET: DEGREE OF COMPETITION
- TABLE 201 ALLERGY DIAGNOSTIC MARKET: REGION FOOTPRINT
- TABLE 202 ALLERGY DIAGNOSTIC MARKET: PRODUCT & SERVICE FOOTPRINT
- TABLE 203 ALLERGY DIAGNOSTIC MARKET: TEST TYPE FOOTPRINT
- TABLE 204 ALLERGY DIAGNOSTIC MARKET: ALLERGEN FOOTPRINT
- TABLE 205 ALLERGY DIAGNOSTIC MARKET: DETAILED LIST OF KEY STARTUPS/SME PLAYERS
- TABLE 206 ALLERGY DIAGNOSTIC MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SME PLAYERS, BY PRODUCT & SERVICE AND REGION
- TABLE 207 ALLERGY DIAGNOSTIC MARKET: PRODUCT LAUNCHES & APPROVALS, JANUARY 2021-JANUARY 2026
- TABLE 208 ALLERGY DIAGNOSTIC MARKET: DEALS, JANUARY 2021-JANUARY 2026
- TABLE 209 ALLERGY DIAGNOSTIC MARKET: EXPANSIONS, JANUARY 2021-JANUARY 2026
- TABLE 210 THERMO FISHER SCIENTIFIC INC.: COMPANY OVERVIEW
- TABLE 211 THERMO FISHER SCIENTIFIC INC.: PRODUCTS OFFERED
- TABLE 212 THERMO FISHER SCIENTIFIC INC.: PRODUCT LAUNCHES & APPROVALS, JANUARY 2021-JANUARY 2026
- TABLE 213 THERMO FISHER SCIENTIFIC INC.: DEALS, JANUARY 2021-JANUARY 2026
- TABLE 214 DANAHER CORPORATION: COMPANY OVERVIEW
- TABLE 215 DANAHER CORPORATION: PRODUCTS OFFERED
- TABLE 216 SIEMENS HEALTHINEERS AG: COMPANY OVERVIEW
- TABLE 217 SIEMENS HEALTHINEERS AG: PRODUCTS OFFERED
- TABLE 218 CANON, INC. (MINARIS MEDICAL AMERICA, INC.): COMPANY OVERVIEW
- TABLE 219 CANON, INC. (MINARIS MEDICAL AMERICA, INC.): PRODUCTS OFFERED
- TABLE 220 CANON, INC. (MINARIS MEDICAL AMERICA, INC.): DEALS, JANUARY 2021-JANUARY 2026
- TABLE 221 REVVITY, INC. (EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG): COMPANY OVERVIEW
- TABLE 222 REVVITY, INC. (EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG): PRODUCTS OFFERED
- TABLE 223 REVVITY, INC. (EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG): DEALS, JANUARY 2021-JANUARY 2026
- TABLE 224 EUROFINS SCIENTIFIC: COMPANY OVERVIEW
- TABLE 225 EUROFINS SCIENTIFIC: PRODUCTS OFFERED
- TABLE 226 EUROFINS SCIENTIFIC: EXPANSIONS, JANUARY 2021-JANUARY 2026
- TABLE 227 BIOMERIEUX SA: COMPANY OVERVIEW
- TABLE 228 BIOMERIEUX SA: PRODUCTS OFFERED
- TABLE 229 DSM ROYAL (ROMER LABS DIVISION HOLDING GMBH): COMPANY OVERVIEW
- TABLE 230 DSM ROYAL (ROMER LABS DIVISION HOLDING GMBH): PRODUCTS OFFERED
- TABLE 231 DSM ROYAL (ROMER LABS DIVISION HOLDING GMBH): DEALS, JANUARY 2021-JANUARY 2026
- TABLE 232 HOLLISTERSTIER ALLERGY (JUBILANT PHARMA): COMPANY OVERVIEW
- TABLE 233 HOLLISTERSTIER ALLERGY (JUBILANT PHARMA): PRODUCTS OFFERED
- TABLE 234 OMEGA DIAGNOSTICS GROUP PLC: COMPANY OVERVIEW
- TABLE 235 OMEGA DIAGNOSTICS GROUP PLC: PRODUCTS OFFERED
- TABLE 236 STALLERGENES GREER LTD.: COMPANY OVERVIEW
- TABLE 237 STALLERGENES GREER LTD.: PRODUCTS OFFERED
- TABLE 238 STALLERGENES GREER LTD.: DEALS, JANUARY 2021-JANUARY 2026
- TABLE 239 HOB BIOTECH GROUP CORP., LTD.: COMPANY OVERVIEW
- TABLE 240 HYCOR BIOMEDICAL: COMPANY OVERVIEW
- TABLE 241 LINCOLN DIAGNOSTICS, INC.: COMPANY OVERVIEW
- TABLE 242 R-BIOPHARM AG: COMPANY OVERVIEW
- TABLE 243 ASTRA BIOTECH GMBH: COMPANY OVERVIEW
- TABLE 244 ERBA GROUP: COMPANY OVERVIEW
- TABLE 245 AESKU.GROUP GMBH: COMPANY OVERVIEW
- TABLE 246 ACON LABORATORIES, INC.: COMPANY OVERVIEW
- TABLE 247 ALCIT INDIA PVT. LTD.: COMPANY OVERVIEW
- TABLE 248 BIOPANDA REAGENTS LTD.: COMPANY OVERVIEW
- TABLE 249 BIOSIDE S.R.L.: COMPANY OVERVIEW
- TABLE 250 CREATIVE DIAGNOSTIC MEDICARE PVT. LTD.: COMPANY OVERVIEW
- TABLE 251 DST DIAGNOSTISCHE SYSTEME & TECHNOLOGIEN GMBH: COMPANY OVERVIEW
- TABLE 252 DR. FOOKE LABORATORIEN GMBH: COMPANY OVERVIEW
- TABLE 253 ALLERGY DIAGNOSTIC MARKET: STUDY ASSUMPTIONS
- TABLE 254 ALLERGY DIAGNOSTIC MARKET: RISK ANALYSIS
List of Figures
- FIGURE 1 ALLERGY DIAGNOSTIC MARKET SEGMENTATION & REGIONAL SCOPE
- FIGURE 2 ALLERGY DIAGNOSTIC MARKET: YEARS CONSIDERED
- FIGURE 3 KEY INSIGHTS & MARKET HIGHLIGHTS
- FIGURE 4 ALLERGY DIAGNOSTIC MARKET SIZE, 2026-2031 (USD MILLION)
- FIGURE 5 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN ALLERGY DIAGNOSTIC MARKET
- FIGURE 6 DISRUPTIVE TRENDS IMPACTING GROWTH OF ALLERGY DIAGNOSTIC MARKET
- FIGURE 7 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS IN ALLERGY DIAGNOSTIC MARKET, BY PRODUCT & SERVICE, TEST TYPE, END USER, AND REGION (2025)
- FIGURE 8 NORTH AMERICA TO REGISTER HIGHEST GROWTH DURING FORECAST PERIOD
- FIGURE 9 INCREASING GOVERNMENT INITIATIVES AND RISING INVESTMENTS TO IMPROVE MARKET GROWTH
- FIGURE 10 CHINA AND HOSPITAL-BASED LABORATORIES ACCOUNTED FOR LARGEST MARKET SHARE IN 2025
- FIGURE 11 INDIA TO REGISTER HIGHEST CAGR DURING FORECAST PERIOD
- FIGURE 12 NORTH AMERICA ACCOUNTED FOR LARGEST MARKET SHARE FROM 2024 TO 2031
- FIGURE 13 EMERGING ECONOMIES TO REGISTER HIGHER GROWTH RATES DURING STUDY PERIOD
- FIGURE 14 ALLERGY DIAGNOSTIC MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 15 ALLERGY DIAGNOSTIC MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 16 ALLERGY DIAGNOSTIC MARKET: SUPPLY CHAIN ANALYSIS
- FIGURE 17 ALLERGY DIAGNOSTIC MARKET: VALUE CHAIN ANALYSIS
- FIGURE 18 ALLERGY DIAGNOSTIC MARKET: ECOSYSTEM ANALYSIS
- FIGURE 19 TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS IN ALLERGY DIAGNOSTIC MARKET
- FIGURE 20 FUNDING AND NUMBER OF DEALS IN ALLERGY DIAGNOSTIC MARKET (USD MILLION)
- FIGURE 21 NUMBER OF PATENTS PUBLISHED IN ALLERGY DIAGNOSTIC MARKET, JANUARY 2016-JANUARY 2026
- FIGURE 22 TOP APPLICANT COUNTRIES/REGIONS FOR ALLERGY DIAGNOSTIC (JANUARY 2015-JANUARY 2026)
- FIGURE 23 INFLUENCE OF KEY STAKEHOLDERS ON BUYING PROCESS OF ALLERGY DIAGNOSTIC PRODUCTS
- FIGURE 24 KEY BUYING CRITERIA FOR ALLERGY DIAGNOSTIC PRODUCTS
- FIGURE 25 NORTH AMERICA: ALLERGY DIAGNOSTIC MARKET SNAPSHOT
- FIGURE 26 ASIA PACIFIC: ALLERGY DIAGNOSTIC MARKET SNAPSHOT
- FIGURE 27 REVENUE ANALYSIS OF TOP PLAYERS IN ALLERGY DIAGNOSTIC MARKET, 2021-2025 (USD MILLION)
- FIGURE 28 MARKET SHARE ANALYSIS OF KEY PLAYERS IN ALLERGY DIAGNOSTIC MARKET, 2025
- FIGURE 29 ALLERGY DIAGNOSTIC MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
- FIGURE 30 ALLERGY DIAGNOSTIC MARKET: COMPANY FOOTPRINT
- FIGURE 31 ALLERGY DIAGNOSTIC MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 32 EV/EBITDA OF KEY VENDORS
- FIGURE 33 YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS
- FIGURE 34 ALLERGY DIAGNOSTIC MARKET: BRAND/PRODUCT COMPARATIVE ANALYSIS
- FIGURE 35 THERMO FISHER SCIENTIFIC INC.: COMPANY SNAPSHOT
- FIGURE 36 DANAHER CORPORATION: COMPANY SNAPSHOT
- FIGURE 37 SIEMENS HEALTHINEERS AG: COMPANY SNAPSHOT
- FIGURE 38 CANON, INC. (MINARIS MEDICAL AMERICA, INC.): COMPANY SNAPSHOT
- FIGURE 39 REVVITY, INC. (EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG): COMPANY SNAPSHOT
- FIGURE 40 EUROFINS SCIENTIFIC: COMPANY SNAPSHOT
- FIGURE 41 BIOMERIEUX SA: COMPANY SNAPSHOT
- FIGURE 42 DSM ROYAL (ROMER LABS DIVISION HOLDING GMBH): COMPANY SNAPSHOT
- FIGURE 43 HOLLISTERSTIER ALLERGY (JUBILANT PHARMA): COMPANY SNAPSHOT
- FIGURE 44 OMEGA DIAGNOSTICS GROUP PLC: COMPANY SNAPSHOT
- FIGURE 45 ALLERGY DIAGNOSTIC MARKET: RESEARCH DESIGN
- FIGURE 46 ALLERGY DIAGNOSTIC MARKET: KEY DATA FROM SECONDARY SOURCES
- FIGURE 47 ALLERGY DIAGNOSTIC MARKET: PRIMARY SOURCES
- FIGURE 48 ALLERGY DIAGNOSTIC MARKET: KEY DATA FROM PRIMARY SOURCES
- FIGURE 49 ALLERGY DIAGNOSTIC MARKET: KEY INSIGHTS FROM INDUSTRY EXPERTS
- FIGURE 50 BREAKDOWN OF PRIMARY INTERVIEWS: SUPPLY/DEMAND-SIDE PARTICIPANTS
- FIGURE 51 BREAKDOWN OF PRIMARY INTERVIEWS: SUPPLY-SIDE PARTICIPANTS, BY COMPANY TYPE, DESIGNATION, AND REGION
- FIGURE 52 BREAKDOWN OF PRIMARY INTERVIEWS: DEMAND-SIDE PARTICIPANTS, BY END USER, DESIGNATION, AND REGION
- FIGURE 53 ALLERGY DIAGNOSTIC MARKET SIZE ESTIMATION
- FIGURE 54 REVENUE SHARE ANALYSIS ILLUSTRATION: THERMO FISHER SCIENTIFIC INC. (2025)
- FIGURE 55 SUPPLY-SIDE ANALYSIS OF KEY PLAYERS IN ALLERGY DIAGNOSTIC MARKET (2025)
- FIGURE 56 ALLERGY DIAGNOSTIC MARKET: TOP-DOWN APPROACH
- FIGURE 57 ALLERGY DIAGNOSTIC MARKET: CAGR PROJECTIONS OF DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES (2024-2031)
- FIGURE 58 ALLERGY DIAGNOSTIC MARKET: CAGR PROJECTIONS (SUPPLY-SIDE ANALYSIS)
- FIGURE 59 ALLERGY DIAGNOSTIC MARKET: DATA TRIANGULATION










