 |
市场调查报告书
商品编码
1961000
全球静脉注射(IV)医疗设备市场:按类型、应用、最终用户和地区划分-预测至2031年IV Equipment Market by Type (IV Catheter, IV Administration Set, Needle-free Connectors), Application (Medication Administration, Parenteral Nutrition, Diagnostic Testing, Blood & Blood Product Transfusion), End User (Hospital) - Global Forecast to 2031 |
||||||
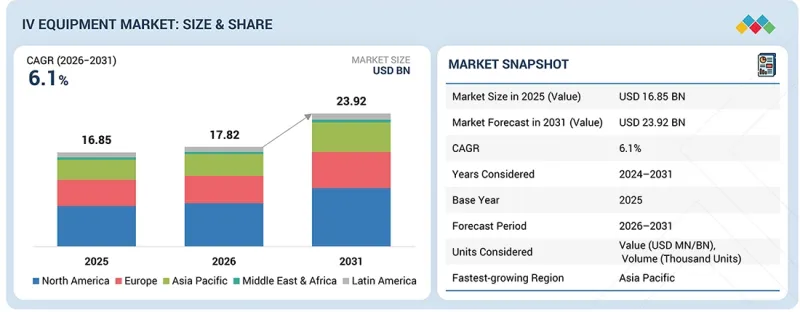
全球静脉注射(IV)医疗设备市场预计将从 2026 年的 178.2 亿美元成长到 2031 年的 239.2 亿美元,预测期内复合年增长率为 6.1%。
| 调查范围 | |
|---|---|
| 调查期 | 2024-2031 |
| 基准年 | 2025 |
| 预测期 | 2026-2031 |
| 目标单元 | 金额(10亿美元) |
| 部分 | 按类型、按应用程式、按最终用户、按地区 |
| 目标区域 | 北美、欧洲、亚太地区、拉丁美洲、中东和非洲。 |
受全球慢性病和住院人数增加的推动,静脉输液(IV)医疗设备的需求正在稳定成长。静脉输液疗法在药物输注、补液和营养补充方面的应用日益广泛,加上居家医疗和门诊照护模式的转变,进一步促进了产品的普及。医院和诊所优先采用高品质、安全且易于使用的静脉输液医疗设备,以确保药物精准输送和病人安全。无针连接器、智慧输液帮浦和相容的多功能静脉输液系统等技术创新正在进一步推动市场渗透。此外,新兴国家医疗基础设施的扩建以及政府为推广现代医疗实践而製定的利好政策,预计将在未来几年内维持市场的强劲成长。
按类型划分,无针连接器和延长管是静脉输液(IV)医疗设备市场中成长最快的细分市场。这主要得益于其更高的安全性、更便捷的使用方式以及显着降低的感染风险和针刺伤风险。这些连接器能够提高药物输注效率,减少污染,并简化医院、诊所和居家医疗机构的工作流程。严格的感染控制法规和医护人员日益增强的病人安全意识也推动了其应用,使其成为现代医疗机构的首选。
按应用领域划分,药物输注在静脉注射装置市场中占据最大份额。这是因为静脉输液疗法可将药物直接输送到血液中,以达到快速起效、剂量精准和最佳治疗效果。这种应用在加护病房、化疗、疼痛管理和严重感染疾病治疗中尤其重要,因为快速且精准的药物传递能够显着改善患者的治疗效果。随着慢性病和急性病患病率的上升以及住院率的增加,基于静脉注射的药物输注需求持续成长。
静脉注射设备市场按地区划分为北美、欧洲、拉丁美洲、亚太以及中东和非洲。北美在全球静脉注射设备市场占有领先地位。这项优势主要归功于慢性病的高发生率、完善的医疗基础设施以及先进医疗技术的快速普及。此外,主要市场参与企业的强大实力、不断增长的医疗费用支出以及医院和居家医疗机构对先进输液系统日益增长的需求,也进一步巩固了该地区的市场主导地位。
静脉注射装置市场的主要企业包括:贝克顿·迪金森公司(美国)、贝朗公司(德国)、费森尤斯公司(德国)、ICU Medical公司(美国)、泰尔茂株式会社(日本)、穆格公司(美国)、泰利福公司(美国)、康德乐公司(美国)、深圳生尔生物医疗电子有限公司(中国)、百特公司(美国)、阿瓦诺斯医疗公司(美国)、尼普洛公司(日本)、JMS公司(日本)、维卡雷尔公司(法国)和米切尔(日本)。
调查范围
本报告按类型、应用、最终用户和地区分析了静脉注射(IV)医疗设备市场。该报告还涵盖了影响市场成长的因素,分析了市场中的各种机会和挑战,并为市场领导提供了详细的竞争格局资讯。此外,报告还分析了各个细分市场的成长趋势,并提供了五个主要地区(以及这些地区内的各个国家)的市场细分收入预测。
购买本报告的好处
本报告将帮助现有企业和新参与企业/中小企业了解市场趋势,最终获得更大的市场份额。购买本报告的企业可以透过运用以下一种或多种策略来增强其市场地位。
本报告深入分析了以下几点:
- 影响静脉输液(IV)设备市场成长的关键驱动因素(全球疾病负担加重和由此导致的老年人口激增、转向家庭和门诊输液)、限制因素(产品召回和故障)、机会(新兴市场尚未开发的成长潜力)和挑战(供应商合作)分析。
- 市场渗透率:全面介绍静脉注射设备市场主要企业提供的产品系列。
- 产品开发/创新:对静脉注射设备市场的未来趋势、研发活动和产品开发进行详细分析。
- 市场发展:提供盈利地区的全面资讯。
- 市场多元化:全面介绍静脉注射(IV)医疗设备市场的新产品、成长区域和最新趋势。
- 竞争评估:对主要市场参与企业的市场区隔、成长策略、收入分析和产品进行详细评估。
目录
第一章:引言
第二章执行摘要
第三章重要考察
第四章 市场概览
- 市场动态
- 未满足的需求和閒置频段
- 相互关联的市场与跨产业机会
- 参与企业一级/二级/三级市场的策略性倡议
第五章 产业趋势
- 波特五力分析
- 总体经济指标
- 供应链分析
- 价值链分析
- 市场生态系统
- 价格分析
- 贸易分析
- 退款方案
- 2026-2027 年主要会议和活动
- 影响客户业务的趋势/颠覆性因素
- 投资和资金筹措场景
- 案例研究分析
- 2025年美国关税对静脉注射(IV)医疗设备市场的影响
第六章:透过科技、专利和数位化/人工智慧应用实现策略颠覆
- 技术分析
- 技术/产品蓝图
- 专利分析
- 未来应用
- 人工智慧/第三代人工智慧对静脉注射(IV)医疗设备市场的影响
第七章永续性和监管情势
- 监理情势
- 对永续性的承诺
第八章:顾客趋势与购买行为
- 主要相关人员和采购标准
- 客户趋势和购买行为
第九章:静脉注射(IV)医疗设备市场(按类型划分)
- 静脉导管
- 点滴帮浦
- 输液器和输液管
- 固定和稳定装置
- 无针连接器和延长套件
- 滴水室
- 其他的
第十章:静脉注射(IV)医疗设备市场(依应用领域划分)
- 药物管理
- 经肠营养(PN)
- 补充体液和电解质
- 输血及血液製品
- 诊断测试
- 其他的
第十一章:静脉注射(IV)医疗设备市场(依最终用户划分)
- 医院
- 门诊手术中心
- 门诊静脉输液治疗及专科诊所
- 居家照护
- 其他的
第十二章 静脉注射(IV)医疗设备市场(按地区划分)
- 北美洲
- 北美宏观经济展望
- 我们
- 加拿大
- 欧洲
- 欧洲宏观经济展望
- 德国
- 英国
- 法国
- 义大利
- 西班牙
- 荷兰
- 丹麦
- 瑞典
- 其他的
- 亚太地区
- 亚太地区宏观经济展望
- 中国
- 日本
- 印度
- 韩国
- 澳洲
- 菲律宾
- 马来西亚
- 越南
- 新加坡
- 印尼
- 泰国
- 其他的
- 拉丁美洲
- 拉丁美洲宏观经济展望
- 巴西
- 墨西哥
- 阿根廷
- 哥伦比亚
- 智利
- 厄瓜多
- 秘鲁
- 其他的
- 中东和非洲
- 中东和非洲宏观经济展望
- 海湾合作委员会国家
- 其他的
第十三章 竞争格局
- 概述
- 主要参与企业的策略/优势
- 2023-2025年收入分析
- 2025年全球市占率分析
- 企业估值矩阵:主要参与企业,2025 年
- 公司估值矩阵:Start-Ups/中小企业,2025 年
- 企业估值和财务指标
- 品牌/产品对比
- 竞争格局
第十四章:公司简介
- 主要参与企业
- BECTON, DICKINSON AND COMPANY
- B. BRAUN SE
- BAXTER
- FRESENIUS SE & CO. KGAA
- ICU MEDICAL, INC.
- TERUMO CORPORATION
- MOOG, INC.
- TELEFLEX INCORPORATED
- SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
- CARDINAL HEALTH
- AVANOS MEDICAL, INC.
- NIPRO
- JMS CO., LTD.
- VYGON
- MICREL MEDICAL DEVICES SA
- 其他公司
- ARCOMED
- HINDUSTAN SYRINGES & MEDICAL DEVICES LTD.
- EITAN MEDICAL
- ANGIPLAST PRIVATE LIMITED
- PRIMEGUARD MEDICAL
- IRADIMED CORPORATION
- EPIC MEDICAL
- ROMSONS
- BEIJING KELLYMED CO., LTD.
- INTUVIE HOLDINGS LLC
- PROMED TECHNOLOGY CO., LIMITED
- MEDCAPTAIN MEDICAL TECHNOLOGY CO., LTD.
第十五章:调查方法
第十六章附录
The global IV equipment market is projected to reach USD 23.92 billion by 2031 from USD 17.82 billion in 2026, growing at a CAGR of 6.1% during the forecast period.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD billion) |
| Segments | Type, Application, End User, and Region |
| Regions covered | North America, Europe, the Asia Pacific, Latin America, and the Middle East & Africa. |
The demand for IV equipment is growing steadily, driven by the rising prevalence of chronic diseases and increasing hospitalizations worldwide. The growing adoption of IV therapy for medication administration, hydration, and nutrition, along with the shift toward home-based and outpatient care, is boosting product utilization. Hospitals and clinics are prioritizing high-quality, safe, and user-friendly IV devices to ensure accurate drug delivery and patient safety. Technological innovations, such as needle-free connectors, smart infusion pumps, and compatible multi-parameter IV systems, are further enhancing market adoption. Moreover, expanding healthcare infrastructure in emerging economies and favorable government initiatives promoting modern healthcare practices are expected to sustain strong market growth in the coming years.

"By type, the needle-free connectors & extension sets segment is projected to grow significantly during the forecast period."
Based on type, needle-free connectors & extension sets are the fastest-growing segment in the IV equipment market, driven by their enhanced safety, ease of use, and ability to significantly reduce the risk of infections and needlestick injuries. These connectors streamline medication administration, reduce contamination, and improve workflow efficiency in hospitals, clinics, and home care settings. Their growing adoption is also supported by stringent infection control regulations and increasing awareness among healthcare providers about patient safety, making them a highly preferred choice in modern healthcare facilities.
"By application, the medication administration segment held the largest market share in 2025."
By application, medication administration holds the largest market share in the IV equipment market, as IV therapy allows drugs to be delivered directly into the bloodstream, ensuring rapid onset of action, precise dosing, and optimal therapeutic effectiveness. This application is particularly critical in intensive care units, chemotherapy treatments, pain management, and severe infection management, where timely and accurate drug delivery can significantly improve patient outcomes. The growing prevalence of chronic and acute diseases, along with increasing hospital admissions, continues to drive the demand for IV-based medication administration.
"By region, North America accounted for the largest market share in 2025."
The IV equipment market is segmented into North America, Europe, Latin America, the Asia Pacific, and the Middle East & Africa. North America leads the global IV equipment market. This dominance is attributed to the high prevalence of chronic diseases, well-established healthcare infrastructure, and rapid adoption of advanced medical technologies. Additionally, the strong presence of key market players, increased healthcare spending, and growing demand for advanced infusion systems in hospitals and home healthcare settings further contribute to the region's leading market position.
A breakdown of the primary participants referred to for this report is provided below:
- By Company Type: Tier 1 (35%), Tier 2 (40%), and Tier 3 (25%)
- By Designation: Directors (25%), Managers (50%), and Others (25%)
- By Region: North America (35%), Europe (30%), the Asia Pacific (15%), and the Rest of the World (20%)
The prominent players in the IV equipment market are Becton, Dickinson and Company (US), B. Braun SE (Germany), Fresenius SE & Co. KGaA (Germany), ICU Medical, Inc. (US), Terumo Corporation (Japan), Moog Inc. (US), Teleflex Incorporated (US), Cardinal Health (US), Shenzhen Mindray Bio-Medical Electronics Co., Ltd. (China), Baxter (US), Avanos Medical, Inc. (US), Nipro (Japan), JMS Co., Ltd. (Japan), Vygon (France), and Micrel Medical Devices SA (Greece), among others.
Research Coverage
This report analyzes the IV equipment market by type, application, end user, and region. It also covers the factors affecting market growth, analyzes the various opportunities and challenges in the market, and provides details of the competitive landscape for market leaders. Furthermore, the report analyzes micromarkets by their individual growth trends and forecasts market segment revenues across five main regions (and the respective countries in these regions).
Reasons to Buy the Report
The report will enable established firms as well as entrants/smaller firms to gauge the pulse of the market, which, in turn, would help them to garner a larger market share. Firms purchasing the report could use one or a combination of the following strategies to strengthen their market presence.
This report provides insights into the following pointers:
- Analysis of key drivers (rising disease burden and the subsequent surge in the geriatric population globally as well as the shift toward home and ambulatory infusion), restraints (product recalls and failures), opportunities (untapped growth potential in emerging markets), and challenges (vendor collaborations) influencing the growth of the IV equipment market.
- Market Penetration: Comprehensive information on the product portfolios offered by the top players in the IV equipment market.
- Product Development/Innovation: Detailed insights on the upcoming trends, R&D activities, and product developments in the IV equipment market.
- Market Development: Comprehensive information on lucrative emerging regions.
- Market Diversification: Exhaustive information about new products, growing geographies, and recent developments in the IV equipment market.
- Competitive Assessment: In-depth assessment of market segments, growth strategies, revenue analysis, and products of the leading market players.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 SEGMENTS CONSIDERED & GEOGRAPHICAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.4 CURRENCY CONSIDERED
- 1.5 STAKEHOLDERS
- 1.6 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 MARKET HIGHLIGHTS & KEY INSIGHTS
- 2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS IN IV EQUIPMENT MARKET
- 2.4 HIGH-GROWTH SEGMENTS
- 2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 IV EQUIPMENT MARKET OVERVIEW
- 3.2 ASIA PACIFIC: IV EQUIPMENT MARKET, BY COUNTRY AND END USER
- 3.3 GEOGRAPHIC SNAPSHOT OF IV EQUIPMENT MARKET
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Rising disease burden and subsequent surge in geriatric population globally
- 4.2.1.2 Shift toward home and ambulatory infusion
- 4.2.1.3 Smart pumps, improved safety features, and precision infusion systems
- 4.2.2 RESTRAINTS
- 4.2.2.1 Product recalls and failures
- 4.2.2.2 Infection and complication risk associated with IV therapy
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Untapped growth potential in emerging markets
- 4.2.3.2 Technological advancements and vendor collaborations
- 4.2.4 CHALLENGES
- 4.2.4.1 Device reliability, maintenance requirements, and user-related errors
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS & WHITE SPACES
- 4.3.1 UNMET NEEDS IN IV EQUIPMENT MARKET
- 4.3.1.1 Interoperable & smart IV systems
- 4.3.1.2 Affordable, reliable equipment for emerging markets
- 4.3.1.3 Better infection-prevention designs
- 4.3.1.4 Home care & ambulatory-friendly IV devices
- 4.3.1.5 Streamlined regulatory pathways
- 4.3.1.6 Wider access & supply consistency
- 4.3.2 WHITE SPACE OPPORTUNITIES
- 4.3.2.1 Low-cost smart infusion pumps for emerging markets
- 4.3.2.2 Integrated IV ecosystem
- 4.3.2.3 Home infusion & ambulatory-first devices
- 4.3.2.4 Infection-prevention IV consumables
- 4.3.2.5 Disposable/Single-use smart components
- 4.3.1 UNMET NEEDS IN IV EQUIPMENT MARKET
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.4.1 INTERCONNECTED MARKETS
- 4.4.2 CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
- 4.5.1 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF SUPPLIERS
- 5.1.4 BARGAINING POWER OF BUYERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMIC INDICATORS
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS & FORECAST
- 5.2.3 TRENDS IN GLOBAL HEALTHCARE INDUSTRY
- 5.2.4 TRENDS IN GLOBAL MEDICAL DEVICE INDUSTRY
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.4 VALUE CHAIN ANALYSIS
- 5.5 MARKET ECOSYSTEM
- 5.5.1 ROLE IN ECOSYSTEM
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE TREND OF IV EQUIPMENT, BY KEY PLAYER, 2023-2025 (USD)
- 5.6.2 AVERAGE SELLING PRICE TREND OF INFUSION PUMPS, BY REGION, 2023-2025 (USD)
- 5.7 TRADE ANALYSIS
- 5.7.1 IMPORT DATA (HS CODE 901890)
- 5.7.2 EXPORT DATA (HS CODE 901890)
- 5.8 REIMBURSEMENT SCENARIO
- 5.9 KEY CONFERENCES & EVENTS, 2026-2027
- 5.10 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- 5.11 INVESTMENT & FUNDING SCENARIO
- 5.12 CASE STUDY ANALYSIS
- 5.13 IMPACT OF 2025 US TARIFFS ON IV EQUIPMENT MARKET
- 5.13.1 INTRODUCTION
- 5.13.2 KEY TARIFF RATES
- 5.13.3 PRICE IMPACT ANALYSIS
- 5.13.4 IMPACT ON COUNTRY/REGION
- 5.13.5 IMPACT ON END-USE SEGMENTS
- 5.13.5.1 Hospitals
- 5.13.5.2 Outpatient infusion & specialty clinics
- 5.13.5.3 Ambulatory surgical centers (ASCs)
- 5.13.5.4 Home care settings
- 5.13.5.5 Other end users (long-term care facilities, emergency services, and specialty clinics)
6 STRATEGIC DISRUPTIONS THROUGH TECHNOLOGY, PATENTS, AND DIGITAL & AI ADOPTION
- 6.1 TECHNOLOGY ANALYSIS
- 6.1.1 KEY EMERGING TECHNOLOGIES
- 6.1.1.1 Smart infusion pumps
- 6.1.1.2 Closed-loop infusion control systems
- 6.1.2 COMPLEMENTARY TECHNOLOGIES
- 6.1.2.1 Dose-error reduction systems
- 6.1.2.2 Telemedicine & remote infusion monitoring
- 6.1.3 ADJACENT TECHNOLOGIES
- 6.1.3.1 Embedded software
- 6.1.1 KEY EMERGING TECHNOLOGIES
- 6.2 TECHNOLOGY/PRODUCT ROADMAP
- 6.2.1 NEAR TERM (2025-2027)
- 6.2.2 MID TERM (2028-2030)
- 6.2.3 LONG TERM (2030+)
- 6.3 PATENT ANALYSIS
- 6.3.1 JURISDICTION & TOP APPLICANT ANALYSIS
- 6.4 FUTURE APPLICATIONS
- 6.4.1 REMOTE & HOME-BASED INFUSION THERAPY
- 6.4.2 CLOSED-LOOP IV THERAPY & AUTOMATED DOSING
- 6.4.3 AI-DRIVEN INFUSION SAFETY & PREDICTIVE QUALITY MANAGEMENT
- 6.5 IMPACT OF AI/GEN AI ON IV EQUIPMENT MARKET
- 6.5.1 INTRODUCTION
- 6.5.2 MARKET POTENTIAL OF AI IN IV EQUIPMENT
- 6.5.3 AI USE CASES
- 6.5.4 KEY COMPANIES IMPLEMENTING AI
7 SUSTAINABILITY & REGULATORY LANDSCAPE
- 7.1 REGULATORY LANDSCAPE
- 7.1.1 REGULATORY FRAMEWORK
- 7.1.1.1 North America
- 7.1.1.2 Europe
- 7.1.1.3 Asia Pacific
- 7.1.1.4 Latin America
- 7.1.1.5 Middle East & Africa
- 7.1.2 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.3 INDUSTRY STANDARDS
- 7.1.1 REGULATORY FRAMEWORK
- 7.2 SUSTAINABILITY INITIATIVES
- 7.2.1 RECYCLED AND ECO-FRIENDLY MATERIALS FOR IV EQUIPMENT
- 7.2.2 SUSTAINABILITY IMPACT & REGULATORY POLICY INITIATIVES
- 7.2.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 KEY STAKEHOLDERS & BUYING CRITERIA
- 8.1.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.1.2 BUYING CRITERIA
- 8.2 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.2.1 DECISION-MAKING PROCESS
- 8.2.2 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.2.3 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.2.4 MARKET PROFITABILITY
9 IV EQUIPMENT MARKET, BY TYPE
- 9.1 INTRODUCTION
- 9.2 IV CATHETERS
- 9.2.1 GROWING HOSPITAL ADMISSIONS AND VASCULAR ACCESS DEMAND TO DRIVE USE OF IV CATHETERS
- 9.3 INFUSION PUMPS
- 9.3.1 SHIFT TOWARD PRECISION MEDICINE AND DIGITAL SAFETY TO ACCELERATE SMART PUMP ADOPTION
- 9.4 IV ADMINISTRATION SETS & TUBING
- 9.4.1 HIGH-VOLUME THERAPY DEMAND AND SUPPLY CHAIN RESILIENCE TO DRIVE IV SET CONSUMPTION
- 9.5 SECUREMENT & STABILIZATION DEVICES
- 9.5.1 GROWING CLINICAL EMPHASIS ON LINE STABILITY AND INFECTION PREVENTION TO ELEVATE SECUREMENT DEMAND
- 9.6 NEEDLE-FREE CONNECTORS & EXTENSION SETS
- 9.6.1 NEEDLESTICK PREVENTION AND INFECTION CONTROL POLICIES TO EXPAND CONNECTOR ADOPTION
- 9.7 IV DRIP CHAMBERS
- 9.7.1 ESSENTIAL FLOW-CONTROL COMPONENTS TO SUPPORT HIGH-VOLUME GRAVITY INFUSIONS
- 9.8 OTHER IV EQUIPMENT
10 IV EQUIPMENT MARKET, BY APPLICATION
- 10.1 INTRODUCTION
- 10.2 MEDICATION ADMINISTRATION
- 10.2.1 ONCOLOGY (IV CHEMOTHERAPY)
- 10.2.1.1 Rising cancer burden to drive market growth
- 10.2.2 ANTIBIOTIC/ANTIMICROBIAL IV THERAPY
- 10.2.2.1 Prolonged antibiotic therapy to boost market growth
- 10.2.3 CRITICAL CARE & VASOACTIVE DRUG INFUSION
- 10.2.3.1 Demand for advanced IV pumps and compatible disposables to aid market growth
- 10.2.4 ANALGESIA/PAIN MANAGEMENT
- 10.2.4.1 Surge in chronic pain conditions to propel market growth
- 10.2.5 SEDATION & ANESTHESIA INFUSION
- 10.2.5.1 High procedural sedation demand to propel market growth
- 10.2.6 IMMUNOTHERAPY
- 10.2.6.1 Increasing use in immune-mediated conditions to support market growth
- 10.2.1 ONCOLOGY (IV CHEMOTHERAPY)
- 10.3 PARENTERAL NUTRITION (PN)
- 10.3.1 GASTROINTESTINAL DISORDERS
- 10.3.1.1 Rising GI disorders to support market growth
- 10.3.2 CRITICAL CARE NUTRITION
- 10.3.2.1 Increasing demand in ICU units to support market growth
- 10.3.3 NEONATAL & PEDIATRIC PARENTERAL NUTRITION
- 10.3.3.1 Increasing demand for neonatal lipid formulations to propel market growth
- 10.3.4 OTHER PARENTERAL NUTRITION APPLICATIONS
- 10.3.1 GASTROINTESTINAL DISORDERS
- 10.4 FLUID & ELECTROLYTE REPLACEMENT
- 10.4.1 GROWING DEMAND FOR FLUID AND REPLACEMENT THERAPY TO DRIVE MARKET GROWTH
- 10.5 BLOOD & BLOOD PRODUCTS TRANSFUSION
- 10.5.1 RED BLOOD CELL TRANSFUSION
- 10.5.1.1 Growing use in symptomatic anemia, acute blood loss, and perioperative blood replacement to support market growth
- 10.5.2 PLATELET TRANSFUSION
- 10.5.2.1 Growing use of standard blood administration sets to fuel growth
- 10.5.3 PLASMA TRANSFUSION
- 10.5.3.1 Wide usage in critical therapy for coagulation disorders to expand market
- 10.5.4 OTHER BLOOD & BLOOD PRODUCTS TRANSFUSION APPLICATIONS
- 10.5.1 RED BLOOD CELL TRANSFUSION
- 10.6 DIAGNOSTIC TESTING
- 10.6.1 INCREASING DEMAND FOR DIAGNOSTIC IMAGING PROCEDURES TO AID MARKET GROWTH
- 10.7 OTHER APPLICATIONS
11 IV EQUIPMENT MARKET, BY END USER
- 11.1 INTRODUCTION
- 11.2 HOSPITALS
- 11.2.1 HIGH DEMAND AND COMPLEX INFUSION WORKFLOWS TO SUSTAIN HOSPITAL DOMINANCE IN IV EQUIPMENT USE
- 11.3 AMBULATORY SURGICAL CENTERS
- 11.3.1 GROWING NUMBER OF SURGICAL PROCEDURES IN ASCS TO PROPEL MARKET GROWTH
- 11.4 OUTPATIENT INFUSION & SPECIALTY CLINICS
- 11.4.1 HIGH DEMAND FOR IV CONSUMABLES AND INFUSION PUMPS TO DRIVE MARKET GROWTH
- 11.5 HOME CARE SETTINGS
- 11.5.1 EXPANSION OF HOME INFUSION THERAPY TO CREATE NEW DEMAND FOR PORTABLE IV DEVICES AND CONSUMABLES
- 11.6 OTHER END USERS
12 IV EQUIPMENT MARKET, BY REGION
- 12.1 INTRODUCTION
- 12.2 NORTH AMERICA
- 12.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 12.2.2 US
- 12.2.2.1 US to dominate North American IV equipment market
- 12.2.3 CANADA
- 12.2.3.1 Rising geriatric patient pool to drive market growth
- 12.3 EUROPE
- 12.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 12.3.2 GERMANY
- 12.3.2.1 Increasing healthcare expenditure to drive market growth
- 12.3.3 UK
- 12.3.3.1 Rising adoption of advanced healthcare systems to support market growth
- 12.3.4 FRANCE
- 12.3.4.1 Rising digital expansion to propel market growth
- 12.3.5 ITALY
- 12.3.5.1 Rising elderly patient pool to drive market growth
- 12.3.6 SPAIN
- 12.3.6.1 Steady modernization of healthcare system in Spain to support market growth
- 12.3.7 NETHERLANDS
- 12.3.7.1 Strong healthcare infrastructure and aging population to drive market
- 12.3.8 DENMARK
- 12.3.8.1 Well-established healthcare infrastructure to boost market
- 12.3.9 SWEDEN
- 12.3.9.1 Rising healthcare expenditure to aid market growth
- 12.3.10 REST OF EUROPE
- 12.4 ASIA PACIFIC
- 12.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 12.4.2 CHINA
- 12.4.2.1 Government support and rising disease burden to drive market growth
- 12.4.3 JAPAN
- 12.4.3.1 High healthcare expenditure, favorable reimbursement, and insurance coverage to support market growth
- 12.4.4 INDIA
- 12.4.4.1 Government initiatives to propel market growth
- 12.4.5 SOUTH KOREA
- 12.4.5.1 Rising aging population to bolster market growth
- 12.4.6 AUSTRALIA
- 12.4.6.1 Rising hospitalization rates to aid market growth
- 12.4.7 PHILIPPINES
- 12.4.7.1 Government investment and public hospital modernization to fuel demand
- 12.4.8 MALAYSIA
- 12.4.8.1 Rising healthcare investment and technological advancements to drive market
- 12.4.9 VIETNAM
- 12.4.9.1 Growing aging population and healthcare infrastructure development to fuel market
- 12.4.10 SINGAPORE
- 12.4.10.1 Advanced healthcare infrastructure and technological integration to boost market
- 12.4.11 INDONESIA
- 12.4.11.1 Rising prevalence of chronic diseases to drive market
- 12.4.12 THAILAND
- 12.4.12.1 Rising geriatric population and chronic diseases to drive market
- 12.4.13 REST OF ASIA PACIFIC
- 12.5 LATIN AMERICA
- 12.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 12.5.2 BRAZIL
- 12.5.2.1 Favorable government initiatives to propel market growth
- 12.5.3 MEXICO
- 12.5.3.1 Rising disease burden to aid market growth
- 12.5.4 ARGENTINA
- 12.5.4.1 Rapid aging population and subsequent rise in chronic diseases to boost market
- 12.5.5 COLOMBIA
- 12.5.5.1 Integration of technological advancements in hospitals & infusion therapy settings to propel market
- 12.5.6 CHILE
- 12.5.6.1 Growing focus on local infusion-therapy product manufacturers to fuel uptake
- 12.5.7 ECUADOR
- 12.5.7.1 Digital health initiatives to support market growth
- 12.5.8 PERU
- 12.5.8.1 Increasing focus on oncology therapeutics to boost market
- 12.5.9 REST OF LATIN AMERICA
- 12.6 MIDDLE EAST & AFRICA
- 12.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 12.6.2 GCC COUNTRIES
- 12.6.2.1 Kingdom of Saudi Arabia (KSA)
- 12.6.2.1.1 Favorable government initiatives to boost growth
- 12.6.2.2 United Arab Emirates (UAE)
- 12.6.2.2.1 Government strategies to drive market in UAE
- 12.6.2.3 Rest of GCC countries
- 12.6.2.1 Kingdom of Saudi Arabia (KSA)
- 12.6.3 REST OF MIDDLE EAST & AFRICA
13 COMPETITIVE LANDSCAPE
- 13.1 OVERVIEW
- 13.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 13.2.1 OVERVIEW OF STRATEGIES ADOPTED BY PLAYERS
- 13.3 REVENUE ANALYSIS, 2023-2025
- 13.4 GLOBAL MARKET SHARE ANALYSIS, 2025
- 13.4.1 US MARKET SHARE ANALYSIS, 2025
- 13.4.2 EUROPE MARKET SHARE ANALYSIS, 2025
- 13.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 13.5.1 STARS
- 13.5.2 EMERGING LEADERS
- 13.5.3 PERVASIVE PLAYERS
- 13.5.4 PARTICIPANTS
- 13.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 13.5.5.1 Company footprint
- 13.5.5.2 Region footprint
- 13.5.5.3 Type footprint
- 13.5.5.4 Application footprint
- 13.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 13.6.1 PROGRESSIVE COMPANIES
- 13.6.2 RESPONSIVE COMPANIES
- 13.6.3 DYNAMIC COMPANIES
- 13.6.4 STARTING BLOCKS
- 13.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2025
- 13.6.5.1 Detailed list of key startups/SMEs
- 13.6.5.2 Competitive benchmarking of key startups/SME players
- 13.7 COMPANY VALUATION & FINANCIAL METRICS
- 13.7.1 FINANCIAL METRICS
- 13.7.2 COMPANY VALUATION
- 13.8 BRAND/PRODUCT COMPARISON
- 13.9 COMPETITIVE SCENARIO
- 13.9.1 PRODUCT LAUNCHES & APPROVALS
- 13.9.2 DEALS
- 13.9.3 EXPANSIONS
- 13.9.4 OTHER DEVELOPMENTS
14 COMPANY PROFILES
- 14.1 KEY PLAYERS
- 14.1.1 BECTON, DICKINSON AND COMPANY
- 14.1.1.1 Business overview
- 14.1.1.2 Products offered
- 14.1.1.3 Recent developments
- 14.1.1.3.1 Product approvals
- 14.1.1.3.2 Deals
- 14.1.1.3.3 Expansions
- 14.1.1.3.4 Other developments
- 14.1.1.4 MnM view
- 14.1.1.4.1 Right to win
- 14.1.1.4.2 Strategic choices
- 14.1.1.4.3 Weaknesses & competitive threats
- 14.1.2 B. BRAUN SE
- 14.1.2.1 Business overview
- 14.1.2.2 Products offered
- 14.1.2.3 Recent developments
- 14.1.2.3.1 Product launches & approvals
- 14.1.2.3.2 Deals
- 14.1.2.4 MnM view
- 14.1.2.4.1 Right to win
- 14.1.2.4.2 Strategic choices
- 14.1.2.4.3 Weaknesses & competitive threats
- 14.1.3 BAXTER
- 14.1.3.1 Business overview
- 14.1.3.2 Products offered
- 14.1.3.3 Recent developments
- 14.1.3.3.1 Product approvals
- 14.1.3.3.2 Deals
- 14.1.3.4 MnM view
- 14.1.3.4.1 Right to win
- 14.1.3.4.2 Strategic choices
- 14.1.3.4.3 Weaknesses & competitive threats
- 14.1.4 FRESENIUS SE & CO. KGAA
- 14.1.4.1 Business overview
- 14.1.4.2 Products offered
- 14.1.4.3 Recent developments
- 14.1.4.3.1 Product approvals
- 14.1.4.3.2 Deals
- 14.1.4.4 MnM view
- 14.1.4.4.1 Right to win
- 14.1.4.4.2 Strategic choices
- 14.1.4.4.3 Weaknesses & competitive threats
- 14.1.5 ICU MEDICAL, INC.
- 14.1.5.1 Business overview
- 14.1.5.2 Products offered
- 14.1.5.3 Recent developments
- 14.1.5.3.1 Product approvals
- 14.1.5.3.2 Deals
- 14.1.5.4 MnM view
- 14.1.5.4.1 Right to win
- 14.1.5.4.2 Strategic choices
- 14.1.5.4.3 Weaknesses & competitive threats
- 14.1.6 TERUMO CORPORATION
- 14.1.6.1 Business overview
- 14.1.6.2 Products offered
- 14.1.6.3 Recent developments
- 14.1.6.3.1 Deals
- 14.1.6.3.2 Expansions
- 14.1.7 MOOG, INC.
- 14.1.7.1 Business overview
- 14.1.7.2 Products offered
- 14.1.7.2.1 Product approvals
- 14.1.8 TELEFLEX INCORPORATED
- 14.1.8.1 Business overview
- 14.1.8.2 Products offered
- 14.1.9 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
- 14.1.9.1 Business overview
- 14.1.9.2 Products offered
- 14.1.9.3 Recent developments
- 14.1.9.3.1 Product launches
- 14.1.9.3.2 Other developments
- 14.1.10 CARDINAL HEALTH
- 14.1.10.1 Business overview
- 14.1.10.2 Products offered
- 14.1.10.3 Recent developments
- 14.1.10.3.1 Expansions
- 14.1.11 AVANOS MEDICAL, INC.
- 14.1.11.1 Business overview
- 14.1.11.2 Products offered
- 14.1.12 NIPRO
- 14.1.12.1 Business overview
- 14.1.12.2 Products offered
- 14.1.12.3 Recent developments
- 14.1.12.3.1 Expansions
- 14.1.13 JMS CO., LTD.
- 14.1.13.1 Business overview
- 14.1.13.2 Products offered
- 14.1.14 VYGON
- 14.1.14.1 Business overview
- 14.1.14.2 Products offered
- 14.1.14.3 Recent developments
- 14.1.14.3.1 Deals
- 14.1.15 MICREL MEDICAL DEVICES SA
- 14.1.15.1 Business overview
- 14.1.15.2 Products offered
- 14.1.15.3 Recent developments
- 14.1.15.3.1 Deals
- 14.1.1 BECTON, DICKINSON AND COMPANY
- 14.2 OTHER PLAYERS
- 14.2.1 ARCOMED
- 14.2.2 HINDUSTAN SYRINGES & MEDICAL DEVICES LTD.
- 14.2.3 EITAN MEDICAL
- 14.2.4 ANGIPLAST PRIVATE LIMITED
- 14.2.5 PRIMEGUARD MEDICAL
- 14.2.6 IRADIMED CORPORATION
- 14.2.7 EPIC MEDICAL
- 14.2.8 ROMSONS
- 14.2.9 BEIJING KELLYMED CO., LTD.
- 14.2.10 INTUVIE HOLDINGS LLC
- 14.2.11 PROMED TECHNOLOGY CO., LIMITED
- 14.2.12 MEDCAPTAIN MEDICAL TECHNOLOGY CO., LTD.
15 RESEARCH METHODOLOGY
- 15.1 RESEARCH APPROACH
- 15.2 RESEARCH METHODOLOGY DESIGN
- 15.2.1 SECONDARY RESEARCH
- 15.2.1.1 Key data from secondary sources
- 15.2.2 PRIMARY DATA
- 15.2.2.1 Key industry insights
- 15.2.1 SECONDARY RESEARCH
- 15.3 MARKET SIZE ESTIMATION
- 15.3.1 BOTTOM-UP APPROACH
- 15.4 MARKET BREAKDOWN & DATA TRIANGULATION
- 15.5 MARKET SHARE ESTIMATION
- 15.6 ASSUMPTIONS
- 15.6.1 GROWTH RATE ASSUMPTIONS
- 15.7 RISK ASSESSMENT
- 15.8 LIMITATIONS
- 15.8.1 METHODOLOGY-RELATED LIMITATIONS
- 15.8.2 SCOPE-RELATED LIMITATIONS
16 APPENDIX
- 16.1 DISCUSSION GUIDE
- 16.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 16.3 CUSTOMIZATION OPTIONS
- 16.3.1 COMPANY INFORMATION
- 16.3.2 GEOGRAPHIC ANALYSIS
- 16.3.3 REGIONAL/COUNTRY-LEVEL MARKET SHARE ANALYSIS
- 16.3.4 COUNTRY-LEVEL VOLUME ANALYSIS
- 16.3.5 ANY CONSULTS/CUSTOM REQUIREMENTS AS PER CLIENT REQUEST
- 16.4 RELATED REPORTS
- 16.5 AUTHOR DETAILS
List of Tables
- TABLE 1 IV EQUIPMENT MARKET: INCLUSIONS & EXCLUSIONS
- TABLE 2 EXCHANGE RATES UTILIZED FOR CONVERSION TO USD
- TABLE 3 IV EQUIPMENT MARKET: PORTER'S FIVE FORCES ANALYSIS
- TABLE 4 GDP CHANGE, BY KEY COUNTRY, 2021-2030 (USD BILLION)
- TABLE 5 IV EQUIPMENT MARKET: ROLE IN ECOSYSTEM
- TABLE 6 AVERAGE SELLING PRICE TREND OF IV EQUIPMENT, BY KEY PLAYER, 2023-2025 (USD)
- TABLE 7 AVERAGE SELLING PRICE TREND OF INFUSION PUMPS, BY REGION, 2023-2025 (USD)
- TABLE 8 IMPORT DATA FOR INSTRUMENTS AND APPLIANCES USED IN MEDICAL, SURGICAL, OR VETERINARY SCIENCES (HS CODE 901890), BY COUNTRY, 2020-2024 (USD THOUSAND)
- TABLE 9 EXPORT DATA FOR INSTRUMENTS AND APPLIANCES USED IN MEDICAL, SURGICAL, OR VETERINARY SCIENCES (HS CODE 901890), BY COUNTRY, 2020-2024 (USD THOUSAND)
- TABLE 10 IV EQUIPMENT MARKET: MAJOR CONFERENCES & EVENTS IN 2026-2027
- TABLE 11 CASE STUDY 1: NEXT-GENERATION INTRAVENOUS (IV) INFUSION PUMPS
- TABLE 12 CASE STUDY 2: PREVENTING IV CATHETER DISLODGMENT IN PEDIATRIC NEUROSURGERY
- TABLE 13 CASE STUDY 3: INFUSION PUMP INTEROPERABILITY TRANSFORMS MEDICATION SAFETY
- TABLE 14 US-ADJUSTED RECIPROCAL TARIFF RATES, 2024
- TABLE 15 KEY PRODUCT-RELATED TARIFFS EFFECTIVE
- TABLE 16 NORTH AMERICA: IMPACT OF US TARIFFS ON CANADA
- TABLE 17 ASIA PACIFIC: IMPACT OF US TARIFFS ON CHINA, JAPAN, AND INDIA
- TABLE 18 EUROPE: IMPACT OF US TARIFFS ON GERMANY AND UK
- TABLE 19 LATIN AMERICA: IMPACT OF US TARIFFS ON MEXICO
- TABLE 20 IV EQUIPMENT MARKET: KEY PATENTS, 2023-2025
- TABLE 21 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 22 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 23 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 24 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 25 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 26 INFLUENCE OF STAKEHOLDERS IN BUYING PROCESS OF IV EQUIPMENT
- TABLE 27 KEY BUYING CRITERIA FOR IV EQUIPMENT, BY TYPE
- TABLE 28 IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 29 COMMERCIAL IV CATHETERS AVAILABLE IN MARKET
- TABLE 30 IV CATHETERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 31 COMMERCIAL INFUSION PUMPS AVAILABLE IN MARKET
- TABLE 32 INFUSION PUMPS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 33 COMMERCIAL IV ADMINISTRATION SETS & TUBING AVAILABLE IN MARKET
- TABLE 34 IV ADMINISTRATION SETS & TUBING MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 35 COMMERCIAL SECUREMENT & STABILIZATION DEVICES AVAILABLE IN MARKET
- TABLE 36 SECUREMENT & STABILIZATION DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 37 COMMERCIAL NEEDLE-FREE CONNECTORS & EXTENSION SETS AVAILABLE IN MARKET
- TABLE 38 NEEDLE-FREE CONNECTORS & EXTENSION SETS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 39 COMMERCIAL IV DRIP CHAMBERS AVAILABLE IN MARKET
- TABLE 40 IV DRIP CHAMBERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 41 COMMERCIAL OTHER IV EQUIPMENT AVAILABLE IN MARKET
- TABLE 42 OTHER IV EQUIPMENT MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 43 IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 44 IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 45 IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 46 IV EQUIPMENT MARKET FOR ONCOLOGY (IV CHEMOTHERAPY), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 47 IV EQUIPMENT MARKET FOR ANTIBIOTIC/ANTIMICROBIAL IV THERAPY, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 48 IV EQUIPMENT MARKET FOR CRITICAL CARE & VASOACTIVE DRUG INFUSION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 49 IV EQUIPMENT MARKET FOR ANALGESIA/PAIN MANAGEMENT, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 50 IV EQUIPMENT MARKET FOR SEDATION & ANESTHESIA INFUSION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 51 IV EQUIPMENT MARKET FOR IMMUNOTHERAPY, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 52 IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION (PN), BY TYPE, 2024-2031 (USD MILLION)
- TABLE 53 IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION (PN), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 54 IV EQUIPMENT MARKET FOR GASTROINTESTINAL DISORDERS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 55 IV EQUIPMENT MARKET FOR CRITICAL CARE NUTRITION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 56 IV EQUIPMENT MARKET FOR NEONATAL & PEDIATRIC PARENTERAL NUTRITION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 57 IV EQUIPMENT MARKET FOR OTHER PARENTERAL NUTRITION APPLICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 58 IV EQUIPMENT MARKET FOR FLUID & ELECTROLYTE REPLACEMENT, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 59 IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 60 IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 61 IV EQUIPMENT MARKET FOR RED BLOOD CELL TRANSFUSION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 62 IV EQUIPMENT MARKET FOR PLATELET TRANSFUSION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 63 IV EQUIPMENT MARKET FOR PLASMA TRANSFUSION, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 64 IV EQUIPMENT MARKET FOR OTHER BLOOD & BLOOD PRODUCTS TRANSFUSION APPLICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 65 IV EQUIPMENT MARKET FOR DIAGNOSTIC TESTING, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 66 IV EQUIPMENT MARKET FOR OTHER APPLICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 67 IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 68 IV EQUIPMENT MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 69 IV EQUIPMENT MARKET FOR AMBULATORY SURGICAL CENTERS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 70 IV EQUIPMENT MARKET FOR OUTPATIENT INFUSION & SPECIALTY CLINICS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 71 IV EQUIPMENT MARKET FOR HOME CARE SETTINGS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 72 IV EQUIPMENT MARKET FOR OTHER END USERS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 73 IV EQUIPMENT MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 74 IV CATHETERS MARKET, BY REGION, 2024-2031 (MILLION UNITS)
- TABLE 75 INFUSION PUMPS MARKET, BY REGION, 2024-2031 (MILLION UNITS)
- TABLE 76 IV ADMINISTRATION SETS & TUBING MARKET, BY REGION, 2024-2031 (MILLION UNITS)
- TABLE 77 NORTH AMERICA: IV EQUIPMENT MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 78 NORTH AMERICA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 79 NORTH AMERICA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 80 NORTH AMERICA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 81 NORTH AMERICA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 82 NORTH AMERICA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 83 NORTH AMERICA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 84 US: KEY MACROECONOMIC INDICATORS
- TABLE 85 US: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 86 US: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 87 US: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 88 US: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 89 US: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 90 US: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 91 CANADA: KEY MACROECONOMIC INDICATORS
- TABLE 92 CANADA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 93 CANADA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 94 CANADA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 95 CANADA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 96 CANADA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 97 CANADA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 98 EUROPE: IV EQUIPMENT MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 99 EUROPE: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 100 EUROPE: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 101 EUROPE: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 102 EUROPE: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 103 EUROPE: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 104 EUROPE: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 105 GERMANY: KEY MACROECONOMIC INDICATORS
- TABLE 106 GERMANY: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 107 GERMANY: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 108 GERMANY: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 109 GERMANY: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 110 GERMANY: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 111 GERMANY: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 112 UK: KEY MACROECONOMIC INDICATORS
- TABLE 113 UK: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 114 UK: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 115 UK: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 116 UK: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 117 UK: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 118 UK: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 119 FRANCE: KEY MACROECONOMIC INDICATORS
- TABLE 120 FRANCE: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 121 FRANCE : IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 122 FRANCE : IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 123 FRANCE: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 124 FRANCE: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 125 FRANCE: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 126 ITALY: KEY MACROECONOMIC INDICATORS
- TABLE 127 ITALY: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 128 ITALY: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 129 ITALY: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 130 ITALY: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 131 ITALY: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 132 ITALY: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 133 SPAIN: KEY MACROECONOMIC INDICATORS
- TABLE 134 SPAIN: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 135 SPAIN: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 136 SPAIN: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 137 SPAIN: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 138 SPAIN: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 139 SPAIN: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 140 NETHERLANDS: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 141 NETHERLANDS: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 142 NETHERLANDS: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 143 NETHERLANDS: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 144 NETHERLANDS: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 145 NETHERLANDS: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 146 DENMARK: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 147 DENMARK: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 148 DENMARK: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 149 DENMARK: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 150 DENMARK: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 151 DENMARK: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 152 SWEDEN: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 153 SWEDEN: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 154 SWEDEN: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 155 SWEDEN: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 156 SWEDEN: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 157 SWEDEN: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 158 REST OF EUROPE: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 159 REST OF EUROPE: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 160 REST OF EUROPE: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 161 REST OF EUROPE: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 162 REST OF EUROPE: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 163 REST OF EUROPE: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 164 ASIA PACIFIC: IV EQUIPMENT MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 165 ASIA PACIFIC: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 166 ASIA PACIFIC: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 167 ASIA PACIFIC: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 168 ASIA PACIFIC: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 169 ASIA PACIFIC: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 170 ASIA PACIFIC: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 171 CHINA: KEY MACROECONOMIC INDICATORS
- TABLE 172 CHINA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 173 CHINA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 174 CHINA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 175 CHINA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 176 CHINA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 177 CHINA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 178 JAPAN: KEY MACROECONOMIC INDICATORS
- TABLE 179 JAPAN: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 180 JAPAN: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 181 JAPAN: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 182 JAPAN: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 183 JAPAN: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 184 JAPAN: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 185 INDIA: KEY MACROECONOMIC INDICATORS
- TABLE 186 INDIA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 187 INDIA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 188 INDIA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 189 INDIA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 190 INDIA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 191 INDIA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 192 SOUTH KOREA: KEY MACROECONOMIC INDICATORS
- TABLE 193 SOUTH KOREA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 194 SOUTH KOREA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 195 SOUTH KOREA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 196 SOUTH KOREA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 197 SOUTH KOREA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 198 SOUTH KOREA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 199 AUSTRALIA: KEY MACROECONOMIC INDICATORS
- TABLE 200 AUSTRALIA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 201 AUSTRALIA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 202 AUSTRALIA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 203 AUSTRALIA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 204 AUSTRALIA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 205 AUSTRALIA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 206 PHILIPPINES: KEY MACROECONOMIC INDICATORS
- TABLE 207 PHILIPPINES: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 208 PHILIPPINES: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 209 PHILIPPINES: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 210 PHILIPPINES: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 211 PHILIPPINES: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 212 PHILIPPINES: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 213 MALAYSIA: KEY MACROECONOMIC INDICATORS
- TABLE 214 MALAYSIA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 215 MALAYSIA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 216 MALAYSIA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 217 MALAYSIA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 218 MALAYSIA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 219 MALAYSIA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 220 VIETNAM: KEY MACROECONOMIC INDICATORS
- TABLE 221 VIETNAM: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 222 VIETNAM: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 223 VIETNAM: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 224 VIETNAM: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 225 VIETNAM: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 226 VIETNAM: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 227 SINGAPORE: KEY MACROECONOMIC INDICATORS
- TABLE 228 SINGAPORE: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 229 SINGAPORE: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 230 SINGAPORE: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 231 SINGAPORE: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 232 SINGAPORE: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 233 SINGAPORE: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 234 INDONESIA: KEY MACROECONOMIC INDICATORS
- TABLE 235 INDONESIA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 236 INDONESIA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 237 INDONESIA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 238 INDONESIA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 239 INDONESIA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 240 INDONESIA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 241 THAILAND: KEY MACROECONOMIC INDICATORS
- TABLE 242 THAILAND: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 243 THAILAND: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 244 THAILAND: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 245 THAILAND: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 246 THAILAND: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 247 THAILAND: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 248 REST OF ASIA PACIFIC: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 249 REST OF ASIA PACIFIC: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 250 REST OF ASIA PACIFIC: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 251 REST OF ASIA PACIFIC: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 252 REST OF ASIA PACIFIC: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 253 REST OF ASIA PACIFIC: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 254 LATIN AMERICA: IV EQUIPMENT MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 255 LATIN AMERICA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 256 LATIN AMERICA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 257 LATIN AMERICA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 258 LATIN AMERICA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 259 LATIN AMERICA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 260 LATIN AMERICA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 261 BRAZIL: KEY MACROECONOMIC INDICATORS
- TABLE 262 BRAZIL: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 263 BRAZIL: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 264 BRAZIL: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 265 BRAZIL: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 266 BRAZIL: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 267 BRAZIL: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 268 MEXICO: KEY MACROECONOMIC INDICATORS
- TABLE 269 MEXICO: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 270 MEXICO: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 271 MEXICO: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 272 MEXICO: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 273 MEXICO: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 274 MEXICO: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 275 ARGENTINA: KEY MACROECONOMIC INDICATORS
- TABLE 276 ARGENTINA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 277 ARGENTINA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 278 ARGENTINA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 279 ARGENTINA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 280 ARGENTINA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 281 ARGENTINA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 282 COLOMBIA: KEY MACROECONOMIC INDICATORS
- TABLE 283 COLOMBIA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 284 COLOMBIA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 285 COLOMBIA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 286 COLOMBIA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 287 COLOMBIA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 288 COLOMBIA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 289 CHILE: KEY MACROECONOMIC INDICATORS
- TABLE 290 CHILE: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 291 CHILE: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 292 CHILE: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 293 CHILE: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 294 CHILE: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 295 CHILE: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 296 ECUADOR: KEY MACROECONOMIC INDICATORS
- TABLE 297 ECUADOR: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 298 ECUADOR: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 299 ECUADOR: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 300 ECUADOR: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 301 ECUADOR: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 302 ECUADOR: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 303 PERU: KEY MACROECONOMIC INDICATORS
- TABLE 304 PERU: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 305 PERU: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 306 PERU: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 307 PERU: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 308 PERU: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 309 PERU: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 310 REST OF LATIN AMERICA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 311 REST OF LATIN AMERICA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 312 REST OF LATIN AMERICA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 313 REST OF LATIN AMERICA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 314 REST OF LATIN AMERICA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 315 REST OF LATIN AMERICA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 316 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 317 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 318 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 319 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 320 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 321 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 322 MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 323 GCC COUNTRIES: IV EQUIPMENT MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 324 GCC COUNTRIES: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 325 GCC COUNTRIES: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 326 GCC COUNTRIES: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 327 GCC COUNTRIES: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 328 GCC COUNTRIES: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 329 GCC COUNTRIES: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 330 KINGDOM OF SAUDI ARABIA: KEY MACROECONOMIC INDICATORS
- TABLE 331 KINGDOM OF SAUDI ARABIA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 332 KINGDOM OF SAUDI ARABIA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 333 KINGDOM OF SAUDI ARABIA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 334 KINGDOM OF SAUDI ARABIA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 335 KINGDOM OF SAUDI ARABIA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 336 KINGDOM OF SAUDI ARABIA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 337 UAE: KEY MACROECONOMIC INDICATORS
- TABLE 338 UAE: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 339 UAE: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 340 UAE: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 341 UAE: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 342 UAE: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 343 UAE: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 344 REST OF GCC COUNTRIES: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 345 REST OF GCC COUNTRIES: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 346 REST OF GCC COUNTRIES: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 347 REST OF GCC COUNTRIES: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 348 REST OF GCC COUNTRIES: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 349 REST OF GCC COUNTRIES: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 350 REST OF MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 351 REST OF MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 352 REST OF MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET FOR MEDICATION ADMINISTRATION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 353 REST OF MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET FOR PARENTERAL NUTRITION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 354 REST OF MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET FOR BLOOD & BLOOD PRODUCTS TRANSFUSION, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 355 REST OF MIDDLE EAST & AFRICA: IV EQUIPMENT MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 356 OVERVIEW OF STRATEGIES ADOPTED BY KEY IV EQUIPMENT MANUFACTURING COMPANIES
- TABLE 357 GLOBAL IV EQUIPMENT MARKET: DEGREE OF COMPETITION
- TABLE 358 IV EQUIPMENT MARKET: REGION FOOTPRINT
- TABLE 359 IV EQUIPMENT MARKET: TYPE FOOTPRINT
- TABLE 360 IV EQUIPMENT MARKET: APPLICATION FOOTPRINT
- TABLE 361 IV EQUIPMENT MARKET: DETAILED LIST OF KEY STARTUP/SME PLAYERS
- TABLE 362 IV EQUIPMENT MARKET: COMPETITIVE BENCHMARKING OF KEY STARTUPS/SME PLAYERS
- TABLE 363 IV EQUIPMENT MARKET: PRODUCT LAUNCHES & APPROVALS, JANUARY 2022-DECEMBER 2025
- TABLE 364 IV EQUIPMENT MARKET: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 365 IV EQUIPMENT MARKET: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 366 IV EQUIPMENT MARKET: OTHER DEVELOPMENTS, JANUARY 2022-DECEMBER 2025
- TABLE 367 BECTON, DICKINSON AND COMPANY: COMPANY OVERVIEW
- TABLE 368 BECTON, DICKINSON AND COMPANY: PRODUCTS OFFERED
- TABLE 369 BECTON, DICKINSON AND COMPANY: PRODUCT APPROVALS, JANUARY 2022-DECEMBER 2025
- TABLE 370 BECTON, DICKINSON AND COMPANY: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 371 BECTON, DICKINSON AND COMPANY: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 372 BECTON, DICKINSON AND COMPANY: OTHER DEVELOPMENTS, JANUARY 2022-DECEMBER 2025
- TABLE 373 B. BRAUN SE: COMPANY OVERVIEW
- TABLE 374 B. BRAUN SE: PRODUCTS OFFERED
- TABLE 375 B. BRAUN SE: PRODUCT LAUNCHES & APPROVALS, JANUARY 2022-NOVEMBER 2025
- TABLE 376 B. BRAUN SE: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 377 BAXTER: COMPANY OVERVIEW
- TABLE 378 BAXTER: PRODUCTS OFFERED
- TABLE 379 BAXTER: PRODUCT APPROVALS, JANUARY 2022-NOVEMBER 2025
- TABLE 380 BAXTER: DEALS, JANUARY 2022-NOVEMBER 2025
- TABLE 381 FRESENIUS SE & CO. KGAA: COMPANY OVERVIEW
- TABLE 382 FRESENIUS SE & CO. KGAA: PRODUCTS OFFERED
- TABLE 383 FRESENIUS SE & CO. KGAA: PRODUCT APPROVALS, JANUARY 2022-DECEMBER 2025
- TABLE 384 FRESENIUS SE & CO. KGAA: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 385 ICU MEDICAL, INC.: COMPANY OVERVIEW
- TABLE 386 ICU MEDICAL, INC.: PRODUCTS OFFERED
- TABLE 387 ICU MEDICAL, INC.: PRODUCT APPROVALS, JANUARY 2022-DECEMBER 2025
- TABLE 388 ICU MEDICAL, INC.: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 389 TERUMO CORPORATION: COMPANY OVERVIEW
- TABLE 390 TERUMO CORPORATION: PRODUCTS OFFERED
- TABLE 391 TERUMO CORPORATION: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 392 TERUMO CORPORATION: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 393 MOOG INC.: COMPANY OVERVIEW
- TABLE 394 MOOG INC.: PRODUCTS OFFERED
- TABLE 395 MOOG INC.: PRODUCT APPROVALS, JANUARY 2022-DECEMBER 2025
- TABLE 396 TELEFLEX INCORPORATED: COMPANY OVERVIEW
- TABLE 397 TELEFLEX INCORPORATED: PRODUCTS OFFERED
- TABLE 398 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.: COMPANY OVERVIEW
- TABLE 399 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.: PRODUCTS OFFERED
- TABLE 400 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.: PRODUCT LAUNCHES, JANUARY 2022-DECEMBER 2025
- TABLE 401 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.: OTHER DEVELOPMENTS, JANUARY 2022-DECEMBER 2025
- TABLE 402 CARDINAL HEALTH: COMPANY OVERVIEW
- TABLE 403 CARDINAL HEALTH: PRODUCTS OFFERED
- TABLE 404 CARDINAL HEALTH: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 405 AVANOS MEDICAL, INC.: COMPANY OVERVIEW
- TABLE 406 AVANOS MEDICAL, INC.: PRODUCTS OFFERED
- TABLE 407 NIPRO: COMPANY OVERVIEW
- TABLE 408 NIPRO: PRODUCTS OFFERED
- TABLE 409 NIPRO: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 410 JMS CO., LTD.: COMPANY OVERVIEW
- TABLE 411 JMS CO., LTD.: PRODUCTS OFFERED
- TABLE 412 VYGON: COMPANY OVERVIEW
- TABLE 413 VYGON: PRODUCTS OFFERED
- TABLE 414 VYGON: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 415 MICREL MEDICAL DEVICES SA: COMPANY OVERVIEW
- TABLE 416 MICREL MEDICAL DEVICES SA: PRODUCTS OFFERED
- TABLE 417 MICREL MEDICAL DEVICES SA: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 418 ARCOMED: COMPANY OVERVIEW
- TABLE 419 HINDUSTAN SYRINGES & MEDICAL DEVICES LTD.: COMPANY OVERVIEW
- TABLE 420 EITAN MEDICAL: COMPANY OVERVIEW
- TABLE 421 ANGIPLAST PRIVATE LIMITED: COMPANY OVERVIEW
- TABLE 422 PRIMEGUARD MEDICAL: COMPANY OVERVIEW
- TABLE 423 IRADIMED CORPORATION: COMPANY OVERVIEW
- TABLE 424 EPIC MEDICAL: COMPANY OVERVIEW
- TABLE 425 ROMSONS: COMPANY OVERVIEW
- TABLE 426 BEIJING KELLYMED CO., LTD.: COMPANY OVERVIEW
- TABLE 427 INTUVIE HOLDINGS LLC: COMPANY OVERVIEW
- TABLE 428 PROMED TECHNOLOGY CO., LIMITED: COMPANY OVERVIEW
- TABLE 429 MEDCAPTAIN MEDICAL TECHNOLOGY CO., LTD.: COMPANY OVERVIEW
- TABLE 430 RISK ASSESSMENT: IV EQUIPMENT MARKET
List of Figures
- FIGURE 1 IV EQUIPMENT MARKET: SEGMENTS CONSIDERED & GEOGRAPHICAL SCOPE
- FIGURE 2 IV EQUIPMENT MARKET: YEARS CONSIDERED
- FIGURE 3 MARKET SCENARIO
- FIGURE 4 GLOBAL IV EQUIPMENT MARKET, 2024-2031
- FIGURE 5 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN IV EQUIPMENT MARKET, 2022-2025
- FIGURE 6 DISRUPTIONS INFLUENCING GROWTH OF IV EQUIPMENT MARKET
- FIGURE 7 HIGH-GROWTH SEGMENTS IN IV EQUIPMENT MARKET, 2026-2031
- FIGURE 8 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN IV EQUIPMENT MARKET, IN TERMS OF VALUE, DURING FORECAST PERIOD
- FIGURE 9 GROWING INCIDENCE OF CHRONIC DISEASES TO DRIVE MARKET GROWTH
- FIGURE 10 HOSPITALS SEGMENT IN ASIA PACIFIC REGION ACCOUNTED FOR LARGEST SHARE IN 2025
- FIGURE 11 CHINA TO WITNESS HIGHEST CAGR DURING FORECAST PERIOD
- FIGURE 12 IV EQUIPMENT MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 13 IV EQUIPMENT MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 14 IV EQUIPMENT MARKET: SUPPLY CHAIN ANALYSIS
- FIGURE 15 IV EQUIPMENT MARKET: VALUE CHAIN ANALYSIS
- FIGURE 16 IV EQUIPMENT MARKET ECOSYSTEM
- FIGURE 17 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- FIGURE 18 INVESTMENT & FUNDING SCENARIO FOR IV EQUIPMENT MARKET PLAYERS
- FIGURE 19 TOP APPLICANTS/OWNERS (COMPANIES/INSTITUTIONS) FOR INFUSION PUMP PATENTS (JANUARY 2014-DECEMBER 2025)
- FIGURE 20 IV EQUIPMENT MARKET: AI USE CASES
- FIGURE 21 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS OF IV EQUIPMENT
- FIGURE 22 KEY BUYING CRITERIA FOR IV EQUIPMENT, BY TYPE
- FIGURE 23 NORTH AMERICA: IV EQUIPMENT MARKET SNAPSHOT
- FIGURE 24 ASIA PACIFIC: IV EQUIPMENT MARKET SNAPSHOT
- FIGURE 25 REVENUE ANALYSIS OF KEY PLAYERS IN IV EQUIPMENT MARKET, 2023-2025
- FIGURE 26 GLOBAL MARKET SHARE ANALYSIS OF KEY PLAYERS IN IV EQUIPMENT MARKET, 2025
- FIGURE 27 US MARKET SHARE ANALYSIS OF KEY PLAYERS IN IV EQUIPMENT MARKET, 2025
- FIGURE 28 EUROPE MARKET SHARE ANALYSIS OF KEY PLAYERS IN IV EQUIPMENT MARKET, 2025
- FIGURE 29 IV EQUIPMENT MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
- FIGURE 30 IV EQUIPMENT MARKET: COMPANY FOOTPRINT
- FIGURE 31 IV EQUIPMENT MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 32 EV/EBITDA OF KEY VENDORS
- FIGURE 33 YEAR-TO-DATE PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS
- FIGURE 34 IV EQUIPMENT MARKET: BRAND/PRODUCT COMPARISON
- FIGURE 35 BECTON, DICKINSON AND COMPANY: COMPANY SNAPSHOT (2025)
- FIGURE 36 B. BRAUN SE: COMPANY SNAPSHOT (2024)
- FIGURE 37 BAXTER: COMPANY SNAPSHOT (2024)
- FIGURE 38 FRESENIUS SE & CO. KGAA: COMPANY SNAPSHOT (2024)
- FIGURE 39 ICU MEDICAL, INC.: COMPANY SNAPSHOT (2024)
- FIGURE 40 TERUMO CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 41 MOOG INC.: COMPANY SNAPSHOT (2024)
- FIGURE 42 TELEFLEX INCORPORATED: COMPANY SNAPSHOT (2024)
- FIGURE 43 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.: COMPANY SNAPSHOT (2024)
- FIGURE 44 CARDINAL HEALTH: COMPANY SNAPSHOT (2025)
- FIGURE 45 AVANOS MEDICAL, INC.: COMPANY SNAPSHOT (2024)
- FIGURE 46 NIPRO: COMPANY SNAPSHOT (2024)
- FIGURE 47 JMS CO., LTD.: COMPANY SNAPSHOT (2024)
- FIGURE 48 IV EQUIPMENT MARKET: RESEARCH DESIGN
- FIGURE 49 PRIMARY SOURCES
- FIGURE 50 BREAKDOWN OF PRIMARY INTERVIEWS: BY COMPANY TYPE, DESIGNATION, AND REGION (SUPPLY SIDE)
- FIGURE 51 BREAKDOWN OF PRIMARY INTERVIEWS: BY COMPANY TYPE, DESIGNATION, AND REGION (DEMAND SIDE)
- FIGURE 52 BREAKDOWN OF PRIMARY INTERVIEWS: SUPPLY-SIDE AND DEMAND-SIDE PARTICIPANTS
- FIGURE 53 SUPPLY-SIDE MARKET SIZE ESTIMATION: REVENUE SHARE ANALYSIS
- FIGURE 54 REVENUE SHARE ANALYSIS ILLUSTRATION: BECTON, DICKINSON AND COMPANY
- FIGURE 55 CAGR PROJECTIONS FROM ANALYSIS OF DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF IV EQUIPMENT MARKET (2025-2030)
- FIGURE 56 CAGR PROJECTIONS: SUPPLY-SIDE ANALYSIS
- FIGURE 57 TOP-DOWN APPROACH
- FIGURE 58 BOTTOM-UP APPROACH
- FIGURE 59 MARKET DATA TRIANGULATION METHODOLOGY









