 |
市场调查报告书
商品编码
1958585
生物製剂安全检测市场:依产品类型、检测类型、检测方式、技术类型及地区划分 - 趋势与预测(至2035年)Biologics Safety Testing Market by Type of Offering, Type of Testing, Type of Modality, Type of Technology and Geographical Regions - Trends and Forecast, Till 2035 |
||||||
生物製剂安全检测市场:概要
全球生物製剂安全检测市场预计将从目前的53亿美元成长到2035年的94亿美元,预测期内(至2035年)的年复合成长率(CAGR)为6.6%。
生物製剂安全检测市场:成长与趋势
生物製剂是由各种生物分子(例如蛋白质、碳水化合物、核酸或这些成分的混合物)组成的复杂治疗药物。生物製剂通常由活体生物生产,并且对光照、温度、pH值和氧气浓度等环境因素高度敏感。
由于生物来源和结构复杂性,生物製剂可能存在多种安全风险,包括免疫原性(不良免疫反应)、污染和批间差异。为解决这些问题,安全性测试非常重要,以确保治疗分子不含任何有害杂质,疗效可靠,且对人体应用安全。为应对这项挑战,生物製剂安全性测试是一项严格的程序,目的是验证生物製剂在临床应用或上市前的安全性、纯度、效力和一致性。受慢性病盛行率上升和个人化医疗普及推动,对先进疗法的需求快速成长,预计生物製剂安全性测试市场在预测期内将保持强劲成长。
成长因素 - 市场扩张的策略驱动因素
推动生物製剂安全性测试市场发展的关键因素是对生物製剂和先进疗法的需求不断成长。这使得对这些产品进行全面的安全性测试变得特别必要。此外,诸如新一代定序等先进技术能够快速检测和识别生物製品中的杂质、污染物和安全风险。同时,监管机构也日益认识到先进的安全检测方案在全面评估生物製品方面的重要性。因此,业界制定了全面的指导方针,要求对生物製品的污染物、免疫原性、病毒安全性和生产一致性进行广泛的检测。
市场挑战 - 阻碍发展的重大障碍
生物製品安全检测市场持续成长,但仍存在一些挑战阻碍其广泛应用。复杂的设备、试剂和内部检测能力带来的高昂成本对中小企业造成了沉重负担,限制了它们的市场渗透率,并增加了它们对外包的依赖。此外,复杂且具地域性的法规会导致合规延迟、审批流程冗长,并因标准差异而增加研发成本。生物製剂日益复杂,推动了检测需求的成长,但永续性问题在未能充分解决成本问题的情况下,也带来了营运方面的挑战。
生物製剂安全检测市场 - 关键洞察
本报告深入分析了生物製剂安全检测市场的现状,并指出了该行业的潜在成长机会。主要发现包括:
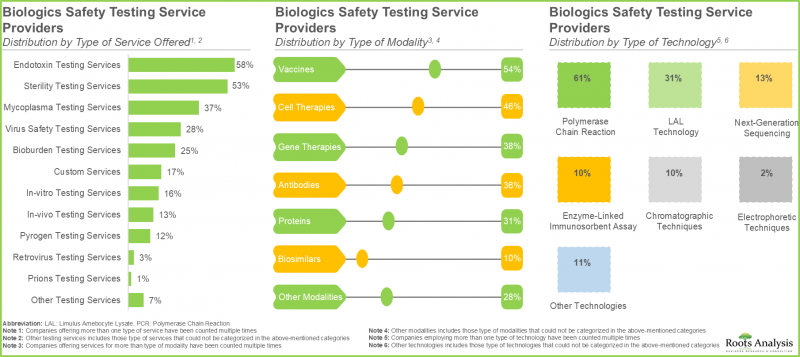
- 60%的生物製剂安全检测服务提供者提供内毒素检测服务。值得注意的是,约55%的服务提供者采用各种技术来评估疫苗的安全性。
- 为了追求竞争优势并满足不断变化的行业需求,利害关係人改进其服务组合併积极提升现有能力。
- 全球约有 40 家公司提供生物製品安全检测解决方案。值得注意的是,超过 80%的生物製品安全检测提供者提供研究用途的解决方案。
- 目前,超过 75%的生物製品安全检测解决方案提供者使用聚合酶炼式反应(PCR)技术,50%的解决方案提供者致力于提供细胞疗法安全检测解决方案。
- 为了确保病人安全并满足严格的监管要求,行业利益相关者积极创新和开发可供各种终端用户使用的生物製品安全检测解决方案。
- 该领域完成的大部分交易都是收购,这表明其战略重点在于扩大市场覆盖范围,并确保在不同地区都能获得专业的生物安全检测解决方案。
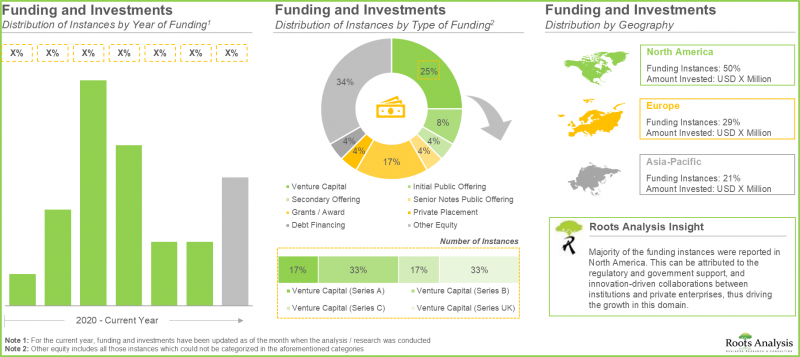
- 过去几年,生物製品安全检测解决方案提供商透过多轮融资筹集了超过 52亿美元的资金,其中超过 90%的资金来自北美公司。
- 在过去两年中,与生物製品安全检测相关的专利约有 40%获得授权,其中绝大多数专利(89%)由产业参与者提交。
- 预计到2035年,生物製剂安全检测市场将保持稳定成长,其中耗材预计将在不久的将来占据大部分市场占有率(超过60%)。
- 目前,抗体细分市场占据了大部分市场占有率。这主要得益于单株抗体的广泛应用,单株抗体在癌症、自体免疫疾病和传染病的标靶治疗中发挥重要作用。
- 美国生物製剂安全检测市场的主要驱动因素是生物製剂日益复杂化以及需要进行生物製剂安全检测的慢性疾病盛行率不断上升。
- 预计到2035年,生物製剂安全检测市场将以约7.1%的年复合成长率成长。其中,生物製剂安全检测耗材预计将占据大部分市场占有率(约60%)。
- 复杂生物製品的快速发展,加上技术的进步,使生物製品安全检测产业在未来具有良好的成长前景。
生物製品安全检测市场
市场规模和机会分析基于以下参数进行细分:
依产品类型
- 耗材
- 设备
- 服务
依检测类型
- 病毒安全检测
- 支原体检测
- 无菌检测
- 内毒素检测
- 生物负荷检测
- 其他检测
依安全性类型
- 抗体
- 疫苗
- 细胞疗法
- 基因疗法
- 生物相似药
- 传统病毒疗法
- 蛋白质
- 其他疗法
依技术类型
- 聚合酶炼式反应(PCR)
- 传统技术
- 色谱与电泳技术
- 次世代定序
- 其他技术
依最终使用者
- 製药和生技公司
- 合约研究组织/合约开发与生产组织
- 学术和研究机构
依地区
- 北美
- 美国
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 义大利
- 西班牙
- 欧洲其他地区
- 亚太地区
- 中国
- 日本
- 印度
- 韩国
- 亚太其他地区
- 中东和北非非洲
- 沙乌地阿拉伯
- 埃及
- 其他中东和北非国家
- 拉丁美洲
- 巴西
- 阿根廷
- 其他拉丁美洲国家
生物製品安全检测市场:主要细分市场
耗材占据生物製品安全侦测市场最大占有率
根据生物製品安全检测市场分析,耗材细分市场占据主导地位(60%),这主要得益于生物製品安全检测所需耗材需求的不断成长。此外,仪器的年复合成长率预计更高,在预测期内展现出显着的成长潜力。这主要归功于能够提高工作流程效率并减少人为错误的仪器的引进。
未来病毒安全性检测将占据最大占有率
生物製剂安全性检测市场预测显示,病毒安全性检测区隔市场将占最大占有率(约30%)。这主要受慢性疾病发生率上升和新型生物疗法接受度提高的推动。预计支原体检测在预测期内将以较高的年复合成长率成长,展现出巨大的成长潜力。这一成长主要得益于严格的监管要求,即在生物製剂生产过程中必须对支原体污染进行彻底检测。 今年,抗体细分市场将占据生物製剂安全性检测市场的最大占有率。 市场分析显示,抗体细分市场约占生物製剂安全性检测市场的35%。其领先地位主要归功于其卓越的特异性、适应性和经证实的有效性。此外,疫苗具有广泛的治疗应用,包括肿瘤、自体免疫疾病和传染病等领域,催生了对生物製剂安全性检测的需求。预计在预测期内,疫苗的年复合成长率将更高,展现出巨大的成长潜力。这一成长与疫苗获批数量的增加以及针对各种靶向适应症的持续研究密切相关。
今年,聚合酶炼式反应(PCR)将占据生物製剂安全性检测市场最大的占有率。
该市场依技术类型细分,包括聚合酶炼式反应(PCR)、传统方法、色谱和电泳、下一代定序以及其他技术。今年,聚合酶炼式反应(PCR)子领域占据了生物製剂安全性检测市场约 35%的显着占有率。这项优势得益于聚合酶炼式反应技术的最新进展。该技术具有更高的准确性、绝对定量能力以及同时检测多个标靶的能力,提高了检测效率和可靠性。此外,PCR技术易于实现高通量筛选的自动化,可以同时检测多种污染物。这种自动化非常重要。提高生物製品安全检测实验室的营运效率并降低检测成本。
北美市场占主导地位,占有率最大
生物製品安全检测市场分析预测,由于先进分子检测技术的日益普及,製药和生物技术公司今年将占据生物製品安全检测市场的大部分占有率(约 50%)。这得归功于它们强大的研发能力、大量的资本投资以及目的是应对日益严峻的全球健康挑战的创新生物製品研发管线。预计製药和生物技术公司在预测期内将实现 7.8%的高年复合成长率,表明其具有强劲的成长潜力。
生物製品安全检测市场主要参与者
- BioMerieux
- Charles River Laboratories
- Clean Cells
- Eurofins
- Genezen
- SGS
- Solvias
- Texcell
- Vimta Labs
- Wickham Micro
生物製品安全检测市场:研究范围
- 市场规模和机会分析:本报告对生物製品安全检测市场进行了详细分析,重点关注以下关键市场细分:[A] 交付形式,[B] 检测类型,[C] 模式类型,[D] 技术类型,以及 [E] 地理区域。
- 生物製品安全检测服务提供者市场概览:除了对整个生物製品安全检测服务提供商市场进行全面的市场概览外,本报告还提供了有关相关参数的详细资讯,例如:[A] 成立年份,[B] 公司规模,[C] 总部所在地,[D] 认证/认可,[E] 服务类型,[F] 检测方法
- 生物製品安全侦测服务提供者竞争分析:基于以下三个面向对生物製品安全侦测服务提供者进行深入分析:[A] 供应商优势,[B] 服务优势,以及 [C] 产品组合多样性。
- 生物製品安全检测解决方案提供者市场概览:除了对生物製品安全检测服务提供者市场的整体评估外,本报告还包含以下资讯:[A] 成立年份,[B] 公司规模,[C] 总部所在地,[D] 服务类型,[E] 检测类型,[F] 实施方法类型,[G] 分析的样本,[H] 技术类型。
- 生物製品安全检测解决方案提供者竞争分析:基于 [A] 供应能力,[B] 服务能力,以及 [C] 产品组合多样性,对生物製品安全检测解决方案提供者进行深入分析。
- 公司简介:基于 [A] 成立年份,[B] 总部所在地,[C] 产品组合,[D] 近期发展,以及 [E] 未来展望,对北美、欧洲和亚太地区从事生物製品安全检测领域的主要公司进行详细介绍。
- 合作伙伴关係与合作:基于多个相关参数(例如[A] 合作年份、[B] 合作类型、[C] 合作伙伴类型和[F] 最活跃参与者)分析利害关係人之间达成的合作伙伴关係与合作。
- 资金与投资分析:基于相关参数(例如[A] 资金年份、[B] 投资金额、[C] 资金类型、[D] 地域分布和[E] 最活跃参与者)详细分析参与生物製品安全检测市场的利害关係人的资金和投资状况。
- 专利分析:基于多个相关参数(例如[A] 公开年份、[B] 专利类型、[C] 申请年份、[D] 专利管辖区和[E] 主要持有人)详细分析与生物製品安全检测相关的各项专利。
- 市场影响分析:详细分析可能影响生物製品安全检测市场成长的因素。它还包括以下方面的识别和市场分析:[A]关键驱动因素,[B]潜在限制因素,[C]新兴机会,以及[D]现有挑战。
目录
第1章 报告概述
第2章 研究方法
第3章 市场动态
第4章 宏观经济指标
第5章 执行摘要
第6章 引言
- 生物安全检测概述
- 主要生物安全检测
- 生物安全领域面临的挑战
- 生物安全检测领域的最新趋势
- 未来展望
第7章 市场格局:生物安全侦测服务供应商
- 研究方法及关键指标参数
- 生物安全检测服务提供者:市场概况
第8章 竞争分析:生物製品安全侦测服务提供者
- 研究方法与关键参数
- 评分标准
- 同侪比较
- 生物製品安全侦测服务提供者:竞争分析
第9章 市场概况:生物製品安全检测解决方案提供者
- 研究方法与关键参数
- 生物製品安全检测解决方案提供者:市场概况
第10章 竞争分析:生物製品安全侦测解决方案提供者
- 研究方法与关键参数
- 评分标准
- 同侪比较
- 生物製品安全检测解决方案供应商:竞争分析
第11章 公司简介:生物製品安全侦测供应商
- 章节概述
- BioMerieux
- Charles River Laboratories
- Clean Cells
- Eurofins
- Genezen
- SGS
- Solvias
- Texcell
- Vimta Labs
- Wickham Micro
第12章 合作关係
- 合作模式
- 生物安全检测:合作关係
第13章 资金与投资
第14章 专利分析
第15章 市场影响分析
- 市场驱动因素
- 市场限制
- 市场机会
- 市场挑战
第16章 全球生物製品安全侦测市场
- 关键假设与研究方法
- 全球生物製品安全检测市场:历史趋势(2021年及以后)与预测(至2035年)
- 情境分析
- 主要市场区隔
第17章 生物製品安全检测市场(依服务类型划分)
第18章 生物製品安全检测市场(依研究类型划分)
第19章 生物製品安全检测市场(依检测方式划分)
第20章 生物製品安全检测市场(依技术划分)
第21章 生物製品安全检测市场(依最终用户划分)
第22章 生物製品安全检测市场(依地区划分)
第23章 市场机会分析:北美
第24章 市场机会分析:欧洲
第25章 市场机会分析:亚太地区
第26章 市场机会分析:中东与北非
第27章 市场机会分析:拉丁美洲
第28章 波特五力分析
第29章 结论
第30章 高层洞察
第31章 附录1:表格资料
第32章 公司与组织清单
Biologics Safety Testing Market: Overview
As per Roots Analysis, the global biologics safety testing market is estimated to grow from USD 5.3 billion in the current year to USD 9.4 billion by 2035, at a CAGR of 6.6% during the forecast period, till 2035.
Biologics Safety Testing Market: Growth and Trends
Biologics are intricate therapeutic agents made up of different biomolecules, including proteins, carbohydrates, nucleic acids, or a mix of these components. Biologics are typically produced by living organisms and are extremely responsive to environmental influences, such as light, temperature, pH, and oxygen concentrations.
Biologics can present various safety risks due to their biological origin and structural complexity, including immunogenicity (undesired immune reactions), contamination, and variability between batches. To tackle these issues, safety testing is essential for confirming that these therapeutic molecules are devoid of hazardous impurities, reliably effective, and safe for human application. To tackle this, biologics safety testing is a rigorous procedure aimed at verifying biologic safety, purity, potency, and consistency prior to clinical application or market launch. Driven by surging demand for advanced therapies amid rising chronic disease prevalence and personalized medicine adoption, the biologics safety testing market is poised for robust growth during the forecast period.
Growth Drivers: Strategic Enablers of Market Expansion
Key factors driving the biologics safety testing market include the increasing demand for biologics and advanced therapies, which creates a need for thorough safety testing of these products. Moreover, cutting-edge technologies like next generation sequencing allow for quicker detection and identification of impurities, contaminants, and safety risks in biologics. Furthermore, regulatory agencies are becoming more aware of the importance of advanced safety testing protocols for the thorough assessment of biologic products. As a result, they are establishing comprehensive guidelines that necessitate extensive testing for contaminants, immunogenicity, viral safety, and consistency in the manufacturing of biologics.
Market Challenges: Critical Barriers Impeding Progress
Challenges continue to exist in the biologics safety testing market, even with its ongoing growth, which obstructs quicker adoption. The high costs associated with advanced equipment, reagents, and internal capabilities strain small to mid-sized companies, limiting their market reach and increasing their dependence on outsourcing. Additionally, intricate and region-specific regulations lead to compliance delays, longer approval processes, and increased development costs due to varying standards. The increasing complexity of biologics raises the demands for testing, while sustainability concerns introduce operational challenges without adequately addressing the cost issues.
Biologics Safety Testing Market: Key Insights
The report delves into the current state of the biologics safety testing market and identifies potential growth opportunities within industry. Some key findings from the report include:
- 60% of the biologics safety testing service providers offer endotoxin testing services; interestingly, close to 55% of the service providers are employing various technologies to evaluate safety profile of vaccines.

- In pursuit of gaining a competitive edge and to meet the evolving industry requirements, stakeholders are actively enhancing their existing capabilities by improving their respective service portfolios.
- Close to 40 companies are engaged in offering biologics safety testing solutions across the globe; notably, over 80% of the biologics safety testing providers offer solutions for research purpose.
- Presently, more than 75% of the biologics safety testing solution providers use polymerase chain reaction technique; 50% of the solution providers claim to offer safety testing solutions for cell therapies.
- Industry stakeholders are actively innovating and developing biologics safety testing solutions that can be utilized by various end-users to ensure patient safety and meet stringent regulatory requirements.
- Majority of the deals inked in this domain were acquisitions, indicating a strategic focus on expanding market reach and ensuring availability of specialized biosafety testing solutions across diverse geographic regions.
- In the past few years, over USD 5.2 billion has been raised by biologics safety testing solution providers across various funding rounds; of these, >90% of the total amount was raised by players based in North America.

- Close to 40% of the patents have been granted related to biologics safety testing in the last two years; further, most of the patents have been filed by industry players (89%).
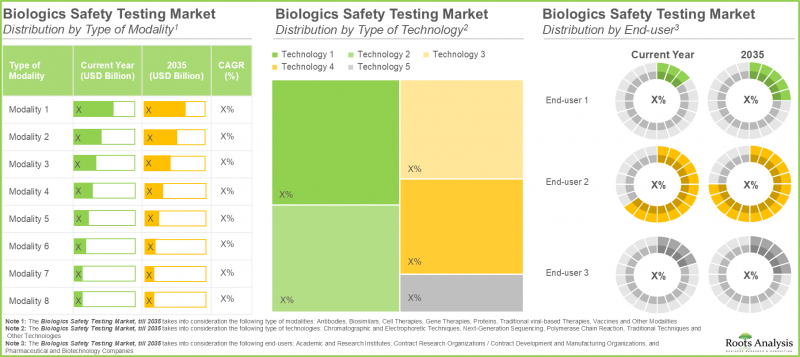
- The biologics safety testing market is anticipated to grow at a steady rate, till 2035; consumables are expected to capture the majority share (more than 60%) of the market in foreseeable future.
- Currently, majority of the market share is captured by antibodies subsegment owing to the widespread use of monoclonal antibodies, which play a vital role in targeted therapies for cancer, autoimmune, and infectious diseases.
- The US biologics safety testing market is primarily driven by the rising number of complex biologics and prevalence of chronic disorders necessitating the need for biologics safety testing.
- The biologics safety testing market is likely to grow at an annualized rate (CAGR) of ~7.1%, till 2035; majority of the market share (~60%) is expected to be captured by biologics safety testing consumables.

- Given the surge in development of complex biologics, combined with technological advancements, the biologics safety testing industry is well-positioned for future growth.
Biologics Safety Testing Market
The market sizing and opportunity analysis has been segmented across the following parameters:
By Type of Offering
- Consumables
- Instruments
- Services
By Type of Testing
- Virus Safety Testing
- Mycoplasma Testing
- Sterility Testing
- Endotoxin Testing
- Bioburden Testing
- Other Testing
By Type of Modality
- Antibodies
- Vaccines
- Cell Therapies
- Gene Therapies
- Biosimilars
- Traditional viral-based Therapies
- Proteins
- Other Modalities
By Type of Technology
- Polymerase Chain Reaction
- Traditional Techniques
- Chromatographic and Electrophoretic Techniques
- Next-Generation Sequencing
- Other Technologies
By End-user
- Pharmaceutical and Biotechnology Companies
- Contract Research Organizations / Contract Development and Manufacturing Organizations
- Academic and Research Institutes
By Geographical Regions
- North America
- US
- Canada
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Rest of Asia-Pacific
- Middle East and North Africa
- Saudi Arabia
- Egypt
- Rest of Middle East and North Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Biologics Safety Testing Market: Key Segments
Consumables Account for the Largest Biologics Safety Testing Market Share
According to the biologics safety testing market analysis, the consumables sub-segment dominates the market (60%), driven by the increased demand for consumables, essential for biologics safety testing. Further, instruments are likely to grow at a higher CAGR, showcasing substantial growth potential during the forecast period. This is because the adoption of instruments can streamline the overall workflow and reduce human error.
Virus Safety Testing Hold the Highest Share in the Future
The forecast for the biologics safety testing market indicates that the virus safety testing sub-segment holds the largest share (approximately 30%), fueled by the increasing incidence of chronic illnesses and the growing acceptance of novel biologic treatments. Mycoplasma testing is expected to experience a higher compound annual growth rate (CAGR), demonstrating significant growth opportunities during the forecast period. This growth is largely attributed to stringent regulatory requirements that necessitate thorough testing for mycoplasma contamination in the production of biologics.
Antibodies Segment Holds the Highest Biologics Safety Testing Market Share in the Current Year
As per market analysis, the antibodies sub-segment represents approximately 35% of the biologics safety testing market share. This leading position is largely attributed to their remarkable specificity, adaptability, and proven efficiency. Furthermore, they possess a wide array of therapeutic uses, including in the fields of oncology, autoimmune disorders, and infectious diseases, which creates a demand for biological safety testing. Vaccines are expected to experience a higher compound annual growth rate (CAGR), indicating significant growth potential throughout the forecast period. This increase can be linked to the rising number of vaccine approvals and their ongoing investigation for various target indications.
Polymerase Chain Reaction Holds the Highest Biologics Safesty Testing Market Share in the Current Year
The overall market is segmented into various technology types, including polymerase chain reaction, traditional methods, chromatographic and electrophoretic methods, next-generation sequencing, and other technologies. The polymerase chain reaction sub-segment holds a significant share of the biologics safety testing market (~35%) this year. This dominance is attributed to recent advancements in polymerase chain reaction technology that provide greater accuracy, absolute quantification, and the capability to detect multiple targets simultaneously, thus improving testing efficiency and reliability. Additionally, the ease of automating PCR technologies for high-throughput screening allows for the simultaneous detection of several contaminants. Such automation enhances operational efficiency and lowers testing costs for biological safety testing laboratories.
North America Dominates the Market by Securing Highest Share
As per the analysis of the biologics safety testing market, the growing implementation of advanced molecular testing technologies is expected to enable pharmaceutical and biotechnology firms to capture a significant portion of the biologics safety testing market (approximately 50% share) in the current year. This is attributed to their strong capabilities in research and development (R&D), considerable financial investments, and robust pipelines of innovative biologics designed to meet the increasing global health challenges. Pharmaceutical and biotechnology firms are predicted to experience a higher compound annual growth rate (CAGR) of 7.8%, indicating considerable growth potential throughout the forecast period.
Primary Research Overview
Discussions with multiple stakeholders in this domain influenced the opinions and insights presented in this study. The market report includes transcripts of the following other third-party discussions:
- Business Development Officer, Mid-sized Organization, Austria
- Business Development Director, Mid-sized Organization, Poland
In addition, the market report includes transcripts of the following other third-party discussions:
- R&D Scientist and Product Manager, Very Large Organization, Germany
- Scientific Expert, Very Large Organization, United States
Example Players in Biologics Safety Testing Market
- BioMerieux
- Charles River Laboratories
- Clean Cells
- Eurofins
- Genezen
- SGS
- Solvias
- Texcell
- Vimta Labs
- Wickham Micro
Biologics Safety Testing Market: Research Coverage
- Market Sizing and Opportunity Analysis: The report features an in-depth analysis of the biologics safety testing market, focusing on key market segments, including [A] type of offering [B] type of testing, [C] type of modality, [D] type of technology, and [E] geographical regions.
- Biologics Safety Testing Service Providers Market Landscape: A detailed assessment of the overall biologics safety testing service providers market landscape, along with information on several relevant parameters, such as [A] year of establishment, [B] company size, [C] location of headquarters, [D] certification / accreditation, [E] type of service offered [F] type of modality, [G] type of sample supported, [H type of technology, and [I] end user.
- Competitiveness Analysis of Biologics Safety Testing Service Providers: An insightful analysis of biologics safety testing service providers based on the [A] supplier strength and [B] service strength and [C] portfolio diversity.
- Biologics Safety Testing Solution Providers Market Landscape: A detailed assessment of the overall biologics safety testing service providers market landscape, along with information on several relevant parameters, such as [A] year of establishment, [B] company size, [C] location of headquarters, [D] type of offering, [E] type of testing [F] type of modality, [G] type of sample analyzed, [H type of technology, [I] application area and [J] end user.
- Competitiveness Analysis of Biologics Safety Testing Solution Providers: An insightful analysis of biologics safety testing solution providers based on the [A] supplier strength and [B] service strength and [C] portfolio diversity.
- Company Profiles: In-depth profiles of leading players engaged in biologics safety testing domain in North America, Europe and Asia-Pacific based on [A] year of establishment, [B] location of headquarters, [C] product portfolio, [D] recent developments and [E] an informed future outlook.
- Partnerships and Collaborations: An analysis of partnerships and collaborations inked between stakeholders, based on several relevant parameters, such as [A] year of partnership, [B] type of partnership, [C] type of partner, and [F] most active players.
- Funding and Investment Analysis: A detailed analysis of the funding and investments raised by stakeholders engaged in the biologics safety testing market, based on several relevant parameters, such as [A] year of funding, [B] amount invested, [C] type of funding, [D] regional distribution, [E] and most active players.
- Patent Analysis: an in-depth analysis of various patents that have been filed / granted for biologics safety testing based on several relevant parameters, such as [A] publication year, [B] type of patent, [C] application year, [D] patent jurisdiction, [E] and leading individual assignees.
- Market Impact Analysis: An in-depth analysis of the factors that can impact the growth of the biologics safety testing market. It also features identification and market analysis of [A] key drivers, [B] potential restraints, [C] emerging opportunities, and [D] existing challenges.
Key Questions Answered in this Report
- Which are the leading companies in biologics safety testing market?
- Which region dominates the biologics safety testing market?
- What are the key trends observed in the biologics safety testing market?
- What factors are likely to influence the evolution of this market?
- What are the primary challenges faced by biologics safety testing service providers?
- What is the current and future market size?
- What is the CAGR of this market?
- How is the current and future market opportunity likely to be distributed across key market segments?
Reasons to Buy this Report
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
- The report can aid businesses in identifying future opportunities in any sector. It also helps in understanding if those opportunities are worth pursuing.
- The report helps in identifying customer demand by understanding the needs, preferences, and behavior of the target audience in order to tailor products or services effectively.
- The report equips new entrants with requisite information regarding a particular market to help them build successful business strategies.
- The report allows for more effective communication with the audience and in building strong business relations.
Additional Benefits
- Complementary PPT Insights Pack
- Complimentary Excel Data Packs for all Analytical Modules in the Report
- 15% Free Content Customization
- Detailed Report Walkthrough Session with Research Team
- Free Updated report if the report is 6-12 months old or older
TABLE OF CONTENTS
1 REPORT OVERVIEW
- 1.1. Context
- 1.2. Project Objectives
2 RESEARCH METHODOLOGY
- 2.1. Chapter Overview
- 2.2. Research Assumptions
- 2.2.1. Market Landscape and Market Trends
- 2.2.2. Market Forecast and Opportunity Analysis
- 2.2.3. Comparative Analysis
- 2.3. Database Building
- 2.3.1. Data Collection
- 2.3.2. Data Validation
- 2.3.3. Data Analysis
- 2.4. Project Methodology
- 2.4.1. Secondary Research
- 2.4.1.1. Annual Reports
- 2.4.1.2. Academic Research Papers
- 2.4.1.3. Company Websites
- 2.4.1.4. Investor Presentations
- 2.4.1.5. Regulatory Filings
- 2.4.1.6. White Papers
- 2.4.1.7. Industry Publications
- 2.4.1.8. Conferences and Seminars
- 2.4.1.9. Government Portals
- 2.4.1.10. Media and Press Releases
- 2.4.1.11. Newsletters
- 2.4.1.12. Industry Databases
- 2.4.1.13. Roots Proprietary Databases
- 2.4.1.14. Paid Databases and Sources
- 2.4.1.15. Social Media Portals
- 2.4.1.16. Other Secondary Sources
- 2.4.2. Primary Research
- 2.4.2.1. Types of Primary Research
- 2.4.2.1.1. Qualitative Research
- 2.4.2.1.2. Quantitative Research
- 2.4.2.1.3. Hybrid Approach
- 2.4.2.2. Advantages of Primary Research
- 2.4.2.3. Techniques for Primary Research
- 2.4.2.3.1. Interviews
- 2.4.2.3.2. Surveys
- 2.4.2.3.3. Focus Groups
- 2.4.2.3.4. Observational Research
- 2.4.2.3.5. Social Media Interactions
- 2.4.2.4. Key Opinion Leaders Considered in Primary Research
- 2.4.2.4.1. Company Executives (CXOs)
- 2.4.2.4.2. Board of Directors
- 2.4.2.4.3. Company Presidents and Vice Presidents
- 2.4.2.4.4. Research and Development Heads
- 2.4.2.4.5. Technical Experts
- 2.4.2.4.6. Subject Matter Experts
- 2.4.2.4.7. Scientists
- 2.4.2.4.8. Doctors and Other Healthcare Providers
- 2.4.2.5. Ethics and Integrity
- 2.4.2.5.1. Research Ethics
- 2.4.2.5.2. Data Integrity
- 2.4.2.1. Types of Primary Research
- 2.4.3. Analytical Tools and Databases
- 2.4.1. Secondary Research
- 2.5. Robust Quality Control
3 MARKET DYNAMICS
- 3.1. Chapter Overview
- 3.2. Forecast Methodology
- 3.2.1. Top-down Approach
- 3.2.2. Bottom-up Approach
- 3.2.3. Hybrid Approach
- 3.3. Market Assessment Framework
- 3.3.1. Total Addressable Market (TAM)
- 3.3.2. Serviceable Addressable Market (SAM)
- 3.3.3. Serviceable Obtainable Market (SOM)
- 3.3.4. Currently Acquired Market (CAM)
- 3.4. Forecasting Tools and Techniques
- 3.4.1. Qualitative Forecasting
- 3.4.2. Correlation
- 3.4.3. Regression
- 3.4.4. Extrapolation
- 3.4.5. Convergence
- 3.4.6. Sensitivity Analysis
- 3.4.7. Scenario Planning
- 3.4.8. Data Visualization
- 3.4.9. Time Series Analysis
- 3.4.10. Forecast Error Analysis
- 3.5. Key Considerations
- 3.5.1. Demographics
- 3.5.2. Government Regulations
- 3.5.3. Reimbursement Scenarios
- 3.5.4. Market Access
- 3.5.5. Supply Chain
- 3.5.6. Industry Consolidation
- 3.5.7. Pandemic / Unforeseen Disruptions Impact
- 3.6. Limitations
4 MACRO-ECONOMIC INDICATORS
- 4.1. Chapter Overview
- 4.2. Market Dynamics
- 4.2.1. Time Period
- 4.2.1.1. Historical Trends
- 4.2.1.2. Current and Forecasted Estimates
- 4.2.2. Currency Coverage
- 4.2.2.1. Major Currencies Affecting the Market
- 4.2.2.2. Factors Affecting Currency Fluctuations
- 4.2.2.3. Impact of Currency Fluctuations on the Industry
- 4.2.3. Foreign Currency Exchange Rate
- 4.2.3.1. Impact of Foreign Exchange Rate Volatility on the Market
- 4.2.3.2. Strategies for Mitigating Foreign Exchange Risk
- 4.2.4. Recession
- 4.2.4.1. Assessment of Current Economic Conditions and Potential Impact on the Market
- 4.2.4.2. Historical Analysis of Past Recessions and Lessons Learnt
- 4.2.5. Inflation
- 4.2.5.1. Measurement and Analysis of Inflationary Pressures in the Economy
- 4.2.5.2. Potential Impact of Inflation on the Market Evolution
- 4.2.6. Interest Rates
- 4.2.6.1. Interest Rates and Their Impact on the Market
- 4.2.6.2. Strategies for Managing Interest Rate Risk
- 4.2.7. Commodity Flow Analysis
- 4.2.7.1. Type of Commodity
- 4.2.7.2. Origins and Destinations
- 4.2.7.3. Values and Weights
- 4.2.7.4. Modes of Transportation
- 4.2.8. Global Trade Dynamics
- 4.2.8.1. Import Scenario
- 4.2.8.2. Export Scenario
- 4.2.8.3. Trade Policies
- 4.2.8.4. Strategies for Mitigating the Risks Associated with Trade Barriers
- 4.2.8.5. Impact of Trade Barriers on the Market
- 4.2.9. War Impact Analysis
- 4.2.9.1. Russian-Ukraine War
- 4.2.9.2. Israel-Hamas War
- 4.2.10. COVID Impact / Related Factors
- 4.2.10.1. Global Economic Impact
- 4.2.10.2. Industry-specific Impact
- 4.2.10.3. Government Response and Stimulus Measures
- 4.2.10.4. Future Outlook and Adaptation Strategies
- 4.2.11. Other Indicators
- 4.2.11.1. Fiscal Policy
- 4.2.11.2. Consumer Spending
- 4.2.11.3. Gross Domestic Product (GDP)
- 4.2.11.4. Employment
- 4.2.11.5. Taxes
- 4.2.11.6. Stock Market Performance
- 4.2.11.7. Cross-Border Dynamics
- 4.2.1. Time Period
- 4.3. Conclusion
5 EXECUTIVE SUMMARY
- 5.1. Executive Summary: Market Landscape
- 5.2. Executive Summary: Market Trends
- 5.3. Executive Summary: Market Forecast and Opportunity Analysis
6 INTRODUCTION
- 6.1. Overview of Biologics Safety Testing
- 6.2. Key Biosafety Tests
- 6.3. Challenges in the Biologics Safety Domain
- 6.4. Recent Developments in the Biologics Safety Testing Domain
- 6.5. Future Perspectives
7 MARKET LANDSCAPE: BIOLOGICS SAFETY TESTING SERVICE PROVIDERS
- 7.1. Methodology and Key Parameters
- 7.2. Biologics Safety Testing Service Providers: Overall Market Landscape
- 7.2.1. Analysis by Year of Establishment
- 7.2.2. Analysis by Company Size
- 7.2.3. Analysis by Location of Headquarters
- 7.2.4. Analysis by Certification / Accreditation
- 7.2.5. Analysis by Type of Service Offered
- 7.2.6. Analysis by Type of Modality
- 7.2.7. Analysis by Type of Sample Supported
- 7.2.8. Analysis by Type of Technology
- 7.2.9. Analysis by End-user
8 COMPANY COMPETITIVENESS ANALYSIS: BIOLOGICS SAFETY TESTING SERVICE PROVIDERS
- 8.1. Methodology and Key Parameters
- 8.2. Scoring Criteria
- 8.3. Peer Groups
- 8.4. Biologics Safety Testing Service Providers: Company Competitiveness Analysis
- 8.4.1. Biologics Safety Testing Service Providers Offering Services for Vaccines
- 8.4.2. Biologics Safety Testing Service Providers Offering Services for Cell Therapies
- 8.4.3. Biologics Safety Testing Service Providers Offering Services for Gene Therapies
- 8.4.4. Biologics Safety Testing Service Providers Offering Services for Antibodies
- 8.4.5. Biologics Safety Testing Service Providers Offering Services for Proteins
- 8.4.6. Biologics Safety Testing Service Providers Offering Services for Other Modalities
9 MARKET LANDSCAPE: BIOLOGICS SAFETY TESTING SOLUTION PROVIDERS
- 9.1. Methodology and Key Parameters
- 9.2. Biologics Safety Testing Solution Providers: Overall Market Landscape
- 9.2.1. Analysis by Year of Establishment
- 9.2.2. Analysis by Company Size
- 9.2.3. Analysis by Location of Headquarters
- 9.2.4. Analysis by Type of Offering
- 9.2.5. Analysis by Type of Testing
- 9.2.6. Analysis by Type of Modality
- 9.2.7. Analysis by Type of Sample Analyzed
- 9.2.8. Analysis by Type of Technology
- 9.2.9. Analysis by Application Area
- 9.2.10. Analysis by End-user
10 COMPANY COMPETITIVENESS ANALYSIS: BIOLOGICS SAFETY TESTING SOLUTION PROVIDERS
- 10.1. Methodology and Key Parameters
- 10.2. Scoring Criteria
- 10.3. Peer Groups
- 10.3.1. Overview of Peer Groups
- 10.4. Biologics Safety Testing Solution Providers: Company Competitiveness Analysis
- 10.4.1. Biologics Safety Testing Solution Providers Headquartered in North America
- 10.4.2. Biologics Safety Testing Solution Providers Headquartered in Europe
- 10.4.3. Biologics Safety Testing Solution Providers Headquartered in Asia-Pacific and Rest of the world
11 COMPANY PROFILES: BIOLOGICS SAFETY TESTING PROVIDERS
- 11.1. Chapter Overview
- 11.2. BioMerieux
- 11.2.1. Company Overview
- 11.2.2. Service Portfolio
- 11.2.3. Financial Information
- 11.2.4. Recent Developments and Future Outlook
- 11.3. Charles River Laboratories
- 11.4. Clean Cells
- 11.5. Eurofins
- 11.6. Genezen
- 11.7. SGS
- 11.8. Solvias
- 11.9. Texcell
- 11.10. Vimta Labs
- 11.11. Wickham Micro
12 PARTNERSHIPS AND COLLABORATIONS
- 12.1. Partnership Models
- 12.2. Biologics Safety Testing: Partnerships and Collaborations
- 12.2.1. Analysis by Year of Partnership
- 12.2.2. Analysis by Type of Partnership
- 12.2.3. Analysis by Year and Type of Partnership
- 12.2.4. Analysis by Type of Biologic
- 12.2.5. Analysis by Type of Testing
- 12.2.6. Analysis by Type of Partner
- 12.2.7. Most Active Players: Analysis by Number of Partnerships
- 12.2.8. Analysis by Geography
- 12.2.8.1. Local and International Deals
- 12.2.8.2. Intracontinental and Intercontinental Deals
13 FUNDING AND INVESTMENTS
- 13.1. Funding Models
- 13.2. Funding Lifecycle Analysis
- 13.3. Investment Case: Risk and Return
- 13.4. Biologics Safety Testing: Funding and Investments
- 13.4.1. Analysis of Instances by Year of Funding
- 13.4.2. Analysis of Instances by Type of Funding
- 13.4.3. Analysis of Instances by Year and Type of Funding
- 13.4.4. Analysis of Amount by Year of Funding
- 13.4.5. Analysis of Amount by Type of Funding
- 13.4.6. Analysis by Geography
- 13.4.7. Key Industry Players: Analysis by Number of Funding Instances
- 13.4.8. Most Active Players: Analysis by Amount Invested
- 13.4.9. Most Active Investors: Analysis by Number of Funding Instances
- 13.4.10. Summary of Investments
14 PATENT ANALYSIS
- 14.1. Methodology and Key Parameters
- 14.2. Biologics Safety Testing: Patent Analysis
- 14.2.1. Analysis by Publication Year
- 14.2.2. Analysis by Type of Patent
- 14.2.3. Analysis by Patent Application Year
- 14.2.4. Analysis by Patent Age
- 14.2.5. Analysis by Type of Applicant
- 14.2.6. Analysis by Patent Jurisdiction
- 14.2.7. Analysis by CPC Symbols
- 14.2.8. Leading Industry Players: Analysis by Number of Patents
- 14.2.9. Leading Non-Industry Players: Analysis by Number of Patents
- 14.2.10. Leading Individual Assignees: Analysis by Number of Patents
- 14.3. Biologics Safety Testing: Patent Benchmarking
- 14.4. Biologics Safety Testing: Patent Valuation
15 MARKET IMPACT ANALYSIS
- 15.1. Market Drivers
- 15.2. Market Restraints
- 15.3. Market Opportunities
- 15.4. Market Challenges
16 GLOBAL BIOLOGICS SAFETY TESTING MARKET
- 16.1. Key Assumptions and Methodology
- 16.2. Global Biologics Safety Testing Market, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 16.2.1. Scenario Analysis
- 16.2.1.1. Conservative Scenario
- 16.2.1.2. Optimistic Scenario
- 16.2.1. Scenario Analysis
- 16.3. Key Market Segments
17 BIOLOGICS SAFETY TESTING MARKET, BY TYPE OF OFFERING
- 17.1. Key Assumptions and Methodology
- 17.2. Biologics Safety Testing Market: Distribution by Type of Offering
- 17.2.1. Biologics Safety Testing Market for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 17.2.2. Biologics Safety Testing Market for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 17.2.3. Biologics Safety Testing Market for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
18 BIOLOGICS SAFETY TESTING MARKET, BY TYPE OF TESTING
- 18.1. Key Assumptions and Methodology
- 18.2. Biologics Safety Testing Market: Distribution by Type of Testing
- 18.2.1. Biologics Safety Testing Market for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 18.2.2. Biologics Safety Testing Market for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 18.2.3. Biologics Safety Testing Market for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 18.2.4. Biologics Safety Testing Market for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 18.2.5. Biologics Safety Testing Market for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 18.2.6. Biologics Safety Testing Market for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
19 BIOLOGICS SAFETY TESTING MARKET, BY TYPE OF MODALITY
- 19.1. Key Assumptions and Methodology
- 19.2. Biologics Safety Testing Market: Distribution by Type of Modality
- 19.2.1. Biologics Safety Testing Market for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.2. Biologics Safety Testing Market for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.3. Biologics Safety Testing Market for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.4. Biologics Safety Testing Market for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.5. Biologics Safety Testing Market for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.6. Biologics Safety Testing Market for Traditional viral-based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.7. Biologics Safety Testing Market for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 19.2.8. Biologics Safety Testing Market for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
20 BIOLOGICS SAFETY TESTING MARKET, BY TYPE OF TECHNOLOGY
- 20.1. Key Assumptions and Methodology
- 20.2. Biologics Safety Testing Market: Distribution by Type of Technology
- 20.2.1. Biologics Safety Testing Market for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 20.2.2. Biologics Safety Testing Market for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 20.2.3. Biologics Safety Testing Market for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 20.2.4. Biologics Safety Testing Market for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 20.2.5. Biologics Safety Testing Market for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
21 BIOLOGICS SAFETY TESTING MARKET, BY END-USER
- 21.1. Key Assumptions and Methodology
- 21.2. Biologics Safety Testing Market: Distribution by End-user
- 21.2.1. Biologics Safety Testing Market for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 21.2.2. Biologics Safety Testing Market for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 21.2.3. Biologics Safety Testing Market for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
22 BIOLOGICS SAFETY TESTING MARKET, BY GEOGRAPHICAL REGIONS
- 22.1. Key Assumptions and Methodology
- 22.2. Biologics Safety Testing Market: Distribution by Geographical Regions
- 22.2.1. Biologics Safety Testing Market in North America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.1.1. Biologics Safety Testing Market in the US, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.1.2. Biologics Safety Testing Market in Canada, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2. Biologics Safety Testing Market in Europe, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2.1. Biologics Safety Testing Market in Germany, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2.2. Biologics Safety Testing Market in the UK, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2.3. Biologics Safety Testing Market in France, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2.4. Biologics Safety Testing Market in Italy, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2.5. Biologics Safety Testing Market in Spain, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.2.6. Biologics Safety Testing Market in Rest of Europe, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.3. Biologics Safety Testing Market in Asia-Pacific, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.3.1. Biologics Safety Testing Market in China, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.3.2. Biologics Safety Testing Market in Japan, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.3.3. Biologics Safety Testing Market in India, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.3.4. Biologics Safety Testing Market in South Korea, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.3.5. Biologics Safety Testing Market in Rest of Asia-Pacific, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.4. Biologics Safety Testing Market in Middle East and North Africa, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.4.1. Biologics Safety Testing Market in Saudi Arabia, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.4.2. Biologics Safety Testing Market in Egypt, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.4.3. Biologics Safety Testing Market in Rest of Middle East and North Africa, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.5. Biologics Safety Testing Market in Latin America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.5.1. Biologics Safety Testing Market in Brazil, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.5.2. Biologics Safety Testing Market in Argentina, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.5.3. Biologics Safety Testing Market in Rest of Latin America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 22.2.1. Biologics Safety Testing Market in North America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
23 MARKET OPPORTUNITY ANALYSIS: NORTH AMERICA
- 23.1. Biologics Safety Testing Market in North America: Distribution by Type of Offering
- 23.1.1. Biologics Safety Testing Market in North America for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.1.2. Biologics Safety Testing Market in North America for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.1.3. Biologics Safety Testing Market in North America for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.2. Biologics Safety Testing Market in North America: Distribution by Type of Testing
- 23.2.1. Biologics Safety Testing Market in North America for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.2.2. Biologics Safety Testing Market in North America for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.2.3. Biologics Safety Testing Market in North America for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.2.4. Biologics Safety Testing Market in North America for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.2.5. Biologics Safety Testing Market in North America for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.2.6. Biologics Safety Testing Market in North America for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3. Biologics Safety Testing Market in North America: Distribution by Type of Modality
- 23.3.1. Biologics Safety Testing Market in North America for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.2. Biologics Safety Testing Market in North America for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.3. Biologics Safety Testing Market in North America for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.4. Biologics Safety Testing Market in North America for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.5. Biologics Safety Testing Market in North America for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.6. Biologics Safety Testing Market in North America for Traditional-viral based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.7. Biologics Safety Testing Market in North America for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.3.8. Biologics Safety Testing Market in North America for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.4. Biologics Safety Testing Market in North America: Distribution by Type of Technology
- 23.4.1. Biologics Safety Testing Market in North America for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.4.2. Biologics Safety Testing Market in North America for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.4.3. Biologics Safety Testing Market in North America for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.4.4. Biologics Safety Testing Market in North America for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.4.5. Biologics Safety Testing Market in North America for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.5. Biologics Safety Testing Market in North America: Distribution by End-user
- 23.5.1. Biologics Safety Testing Market in North America for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.5.2. Biologics Safety Testing Market in North America for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 23.5.3. Biologics Safety Testing Market in North America for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
24 MARKET OPPORTUNITY ANALYSIS: EUROPE
- 24.1. Biologics Safety Testing Market in Europe: Distribution by Type of Offering
- 24.1.1. Biologics Safety Testing Market in Europe for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.1.2. Biologics Safety Testing Market in Europe for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.1.3. Biologics Safety Testing Market in Europe for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.2. Biologics Safety Testing Market in Europe: Distribution by Type of Testing
- 24.2.1. Biologics Safety Testing Market in Europe for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.2.2. Biologics Safety Testing Market in Europe for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.2.3. Biologics Safety Testing Market in Europe for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.2.4. Biologics Safety Testing Market in Europe for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.2.5. Biologics Safety Testing Market in Europe for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.2.6. Biologics Safety Testing Market in Europe for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3. Biologics Safety Testing Market in Europe: Distribution by Type of Modality
- 24.3.1. Biologics Safety Testing Market in Europe for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.2. Biologics Safety Testing Market in Europe for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.3. Biologics Safety Testing Market in Europe for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.4. Biologics Safety Testing Market in Europe for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.5. Biologics Safety Testing Market in Europe for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.6. Biologics Safety Testing Market in Europe for Traditional-viral based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.7. Biologics Safety Testing Market in Europe for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.3.8. Biologics Safety Testing Market in Europe for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.4. Biologics Safety Testing Market in Europe: Distribution by Type of Technology
- 24.4.1. Biologics Safety Testing Market in Europe for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.4.2. Biologics Safety Testing Market in Europe for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.4.3. Biologics Safety Testing Market in Europe for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.4.4. Biologics Safety Testing Market in Europe for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.4.5. Biologics Safety Testing Market in Europe for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.5. Biologics Safety Testing Market in Europe: Distribution by End-user
- 24.5.1. Biologics Safety Testing Market in Europe for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.5.2. Biologics Safety Testing Market in Europe for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 24.5.3. Biologics Safety Testing Market in Europe for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
25 MARKET OPPORTUNITY ANALYSIS: ASIA-PACIFIC
- 25.1. Biologics Safety Testing Market in Asia-Pacific: Distribution by Type of Offering
- 25.1.1. Biologics Safety Testing Market in Asia-Pacific for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.1.2. Biologics Safety Testing Market in Asia-Pacific for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.1.3. Biologics Safety Testing Market in Asia-Pacific for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.2. Biologics Safety Testing Market in Asia-Pacific: Distribution by Type of Testing
- 25.2.1. Biologics Safety Testing Market in Asia-Pacific for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.2.2. Biologics Safety Testing Market in Asia-Pacific for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.2.3. Biologics Safety Testing Market in Asia-Pacific for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.2.4. Biologics Safety Testing Market in Asia-Pacific for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.2.5. Biologics Safety Testing Market in Asia-Pacific for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.2.6. Biologics Safety Testing Market in Asia-Pacific for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3. Biologics Safety Testing Market in Asia-Pacific: Distribution by Type of Modality
- 25.3.1. Biologics Safety Testing Market in Asia-Pacific for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.2. Biologics Safety Testing Market in Asia-Pacific for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.3. Biologics Safety Testing Market in Asia-Pacific for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.4. Biologics Safety Testing Market in Asia-Pacific for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.5. Biologics Safety Testing Market in Asia-Pacific for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.6. Biologics Safety Testing Market in Asia-Pacific for Traditional-viral based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.7. Biologics Safety Testing Market in Asia-Pacific for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.3.8. Biologics Safety Testing Market in Asia-Pacific for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.4. Biologics Safety Testing Market in Asia-Pacific: Distribution by Type of Technology
- 25.4.1. Biologics Safety Testing Market in Asia-Pacific for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.4.2. Biologics Safety Testing Market in Asia-Pacific for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.4.3. Biologics Safety Testing Market in Asia-Pacific for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.4.4. Biologics Safety Testing Market in Asia-Pacific for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.4.5. Biologics Safety Testing Market in Asia-Pacific for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.5. Biologics Safety Testing Market in Asia-Pacific: Distribution by End-user
- 25.5.1. Biologics Safety Testing Market in Asia-Pacific for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.5.2. Biologics Safety Testing Market in Asia-Pacific for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 25.5.3. Biologics Safety Testing Market in Asia-Pacific for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
26 MARKET OPPORTUNITY ANALYSIS: MIDDLE EAST AND NORTH AFRICA
- 26.1. Biologics Safety Testing Market in Middle East and North Africa: Distribution by Type of Offering
- 26.1.1. Biologics Safety Testing Market in Middle East and North Africa for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.1.2. Biologics Safety Testing Market in Middle East and North Africa for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.1.3. Biologics Safety Testing Market in Middle East and North Africa for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.2. Biologics Safety Testing Market in Middle East and North Africa: Distribution by Type of Testing
- 26.2.1. Biologics Safety Testing Market in Middle East and North Africa for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.2.2. Biologics Safety Testing Market in Middle East and North Africa for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.2.3. Biologics Safety Testing Market in Middle East and North Africa for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.2.4. Biologics Safety Testing Market in Middle East and North Africa for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.2.5. Biologics Safety Testing Market in Middle East and North Africa for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.2.6. Biologics Safety Testing Market in Middle East and North Africa for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3. Biologics Safety Testing Market in Middle East and North Africa: Distribution by Type of Modality
- 26.3.1. Biologics Safety Testing Market in Middle East and North Africa for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.2. Biologics Safety Testing Market in Middle East and North Africa for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.3. Biologics Safety Testing Market in Middle East and North Africa for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.4. Biologics Safety Testing Market in Middle East and North Africa for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.5. Biologics Safety Testing Market in Middle East and North Africa for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.6. Biologics Safety Testing Market in Middle East and North Africa for Traditional-viral based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.7. Biologics Safety Testing Market in Middle East and North Africa for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.3.8. Biologics Safety Testing Market in Middle East and North Africa for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.4. Biologics Safety Testing Market in Middle East and North Africa: Distribution by Type of Technology
- 26.4.1. Biologics Safety Testing Market in Middle East and North Africa for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.4.2. Biologics Safety Testing Market in Middle East and North Africa for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.4.3. Biologics Safety Testing Market in Middle East and North Africa for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.4.4. Biologics Safety Testing Market in Middle East and North Africa for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.4.5. Biologics Safety Testing Market in Middle East and North Africa for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.5. Biologics Safety Testing Market in Middle East and North Africa: Distribution by End-user
- 26.5.1. Biologics Safety Testing Market in Middle East and North Africa for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.5.2. Biologics Safety Testing Market in Middle East and North Africa for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 26.5.3. Biologics Safety Testing Market in Middle East and North Africa for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
27 MARKET OPPORTUNITY ANALYSIS: LATIN AMERICA
- 27.1. Biologics Safety Testing Market in Latin America: Distribution by Type of Offering
- 27.1.1. Biologics Safety Testing Market in Latin America for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.1.2. Biologics Safety Testing Market in Latin America for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.1.3. Biologics Safety Testing Market in Latin America for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.2. Biologics Safety Testing Market in Latin America: Distribution by Type of Testing
- 27.2.1. Biologics Safety Testing Market in Latin America for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.2.2. Biologics Safety Testing Market in Latin America for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.2.3. Biologics Safety Testing Market in Latin America for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.2.4. Biologics Safety Testing Market in Latin America for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.2.5. Biologics Safety Testing Market in Latin America for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.2.6. Biologics Safety Testing Market in Latin America for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3. Biologics Safety Testing Market in Latin America: Distribution by Type of Modality
- 27.3.1. Biologics Safety Testing Market in Latin America for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.2. Biologics Safety Testing Market in Latin America for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.3. Biologics Safety Testing Market in Latin America for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.4. Biologics Safety Testing Market in Latin America for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.5. Biologics Safety Testing Market in Latin America for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.6. Biologics Safety Testing Market in Latin America for Traditional-viral based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.7. Biologics Safety Testing Market in Latin America for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.3.8. Biologics Safety Testing Market in Latin America for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.4. Biologics Safety Testing Market in Latin America: Distribution by Type of Technology
- 27.4.1. Biologics Safety Testing Market in Latin America for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.4.2. Biologics Safety Testing Market in Latin America for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.4.3. Biologics Safety Testing Market in Latin America for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.4.4. Biologics Safety Testing Market in Latin America for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.4.5. Biologics Safety Testing Market in Latin America for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.5. Biologics Safety Testing Market in Latin America: Distribution by End-user
- 27.5.1. Biologics Safety Testing Market in Latin America for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.5.2. Biologics Safety Testing Market in Latin America for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- 27.5.3. Biologics Safety Testing Market in Latin America for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- *Detailed information on Chapter 23 to 27 is available in the Excel Data Packs shared along with the report**
28 PORTER'S FIVE FORCES ANALYSIS
- 28.1. Chapter Overview
- 28.1.1. Significance of Porter's Five Forces Analysis
- 28.1.2. Porter's Five Forces Analysis
- 28.2. Methodology
- 28.3. Key Parameters
- 28.3.1. Threats of New Entrants
- 28.3.2. Bargaining Power of Buyers
- 28.3.3. Bargaining Power of Solution Providers
- 28.3.4. Threat of Substitute Solutions
- 28.3.5. Rivalry Among Existing Competitors
- 28.4. Porter's Five Force Analysis: Harvey Ball Analysis
29 CONCLUDING INSIGHTS
30 EXECUTIVE INSIGHTS
31 APPENDIX I: TABULATED DATA
32 APPENDIX II: LIST OF COMPANIES AND ORGANIZATION
List of Tables
- Table 7.1 List of Biologics Safety Testing Service Providers
- Table 7.2 List of Biologics Safety Testing Solution Providers
- Table 11.1 BioMerieux: Service Portfolio
- Table 11.2 BioMerieux: Recent Developments and Future Outlook
- Table 11.3 Charles River Laboratories: Service Portfolio
- Table 11.4 Charles River Laboratories: Recent Developments and Future Outlook
- Table 11.5 Clean Cells: Service Portfolio
- Table 11.6 Clean Cells: Recent Developments and Future Outlook
- Table 11.7 Eurofins: Service Portfolio
- Table 11.8 Eurofins: Developments and Future Outlook
- Table 11.9 Genezen: Service Portfolio
- Table 11.10 SGS: Service Portfolio
- Table 11.11 SGS: Developments and Future Outlook
- Table 11.12 Solvias: Service Portfolio
- Table 11.13 Texcell: Service Portfolio
- Table 11.14 Vimta Labs: Service Portfolio
- Table 11.15 Wickham Micro: Service Portfolio
- Table 12.1 List of Partnerships and Collaborations, Since 2020
- Table 13.1 List of Funding and Investments, Since 2019
- Table 14.1 List of Published Patents, Since 2020
- Table 26.1 Biologics Safety Testing Service Providers: Distribution by Year of Establishment
- Table 26.2 Biologics Safety Testing Service Providers: Distribution by Company Size
- Table 26.3 Biologics Safety Testing Service Providers: Distribution by Location of Headquarters
- Table 26.4 Biologics Safety Testing Service Providers: Distribution by Certification / Accreditation
- Table 26.5 Biologics Safety Testing Service Providers: Distribution by Type of Service Offered
- Table 26.6 Biologics Safety Testing Service Providers: Distribution by Type of Modality
- Table 26.7 Biologics Safety Testing Service Providers: Distribution by Type of Sample Supported
- Table 26.8 Biologics Safety Testing Service Providers: Distribution by Type of Technology
- Table 26.9 Biologics Safety Testing Service Providers: Distribution by End-user
- Table 26.10 Biologics Safety Testing Solution Providers: Distribution by Year of Establishment
- Table 26.11 Biologics Safety Testing Solution Providers: Distribution by Company Size
- Table 26.12 Biologics Safety Testing Solution Providers: Distribution by Location of Headquarters
- Table 26.13 Biologics Safety Testing Solution Providers: Distribution by Type of Offering
- Table 26.14 Biologics Safety Testing Solution Providers: Distribution by Type of Testing
- Table 26.15 Biologics Safety Testing Solution Providers: Distribution by Type of Modality
- Table 26.16 Biologics Safety Testing Solution Providers: Distribution by Type of Sample Analyzed
- Table 26.17 Biologics Safety Testing Solution Providers: Distribution by Type of Technology
- Table 26.18 Biologics Safety Testing Solution Providers: Distribution by Application Area
- Table 26.19 Biologics Safety Testing Solution Providers: Distribution by End-user
- Table 26.20 BioMerieux: Financial Information, Since FY 2022 (EUR Million)
- Table 26.21 Charles River Laboratories: Financial Information, Since FY 2022 (USD Million)
- Table 26.22 Eurofins: Annual Revenues, Financial Information, Since FY 2022 (EUR Million)
- Table 26.23 SGS: Financial Information, Since FY 2022 (CHF Billion)
- Table 26.24 Vimta Labs: Financial Information, Since FY 2022 (INR Million)
- Table 26.25 Partnerships and Collaborations: Cumulative Year-wise Trend, Since Pre-2020
- Table 26.26 Partnerships and Collaborations: Distribution by Type of Partnership
- Table 26.27 Partnerships and Collaborations: Distribution by Year and Type of Partnership
- Table 26.28 Partnerships and Collaborations: Distribution by Type of Biologic
- Table 26.29 Partnerships and Collaborations: Distribution by Type of Testing
- Table 26.30 Partnerships and Collaborations: Distribution by Type of Partner
- Table 26.31 Most Active Players: Distribution by Number of Partnerships
- Table 26.32 Partnerships and Collaborations: Local and International Agreements
- Table 26.33 Partnerships and Collaborations: Intracontinental and Intercontinental Agreements
- Table 26.34 Funding and Investments Cumulative Year-wise Trend, Since 2019
- Table 26.35 Funding and Investments: Distribution of Instances by Type of Funding
- Table 26.36 Funding and Investments: Distribution of Instances by Year and Type of Funding
- Table 26.37 Funding and Investments: Distribution of Amount by Year of Funding
- Table 26.38 Funding and Investments: Distribution of Amount by Type of Funding
- Table 26.39 Funding and Investments: Distribution by Geography
- Table 26.40 Key Industry Players: Analysis by Number of Funding Instances
- Table 26.41 Most Active Players: Analysis by Amount Invested
- Table 26.42 Most Active Investors: Analysis by Number of Funding Instances
- Table 26.43 Patent Analysis: Distribution by Publication Year
- Table 26.44 Patent Analysis: Distribution by Type of Patent
- Table 26.45 Patent Analysis: Distribution by Patent Application Year
- Table 26.46 Patent Analysis: Distribution by Patent Age
- Table 26.47 Patent Analysis: Distribution by Type of Applicant
- Table 26.48 Patent Analysis: Distribution by Patent Jurisdiction
- Table 26.49 Leading Industry Players: Analysis by Number of Patents
- Table 26.50 Leading Non-Industry Players: Analysis by Number of Patents
- Table 26.51 Leading Individual Assignees: Analysis by Number of Patents
- Table 26.52 Biologics Safety Testing Market, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035): Base Scenario (USD Million)
- Table 26.53 Biologics Safety Testing Market, Forecasted Estimates (Till 2035): Conservative Scenario (USD Million)
- Table 26.54 Biologics Safety Testing Market, Forecasted Estimates (Till 2035): Optimistic Scenario (USD Million)
- Table 26.55 Biologics Safety Testing Market: Distribution by Type of Offering, Current Year, 2030 and 2035
- Table 26.56 Biologics Safety Testing Market for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.57 Biologics Safety Testing Market for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.58 Biologics Safety Testing Market for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.59 Biologics Safety Testing Market: Distribution by Type of Testing, Current Year, 2030 and 2035
- Table 26.60 Biologics Safety Testing Market for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.61 Biologics Safety Testing Market for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.62 Biologics Safety Testing Market for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.63 Biologics Safety Testing Market for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.64 Biologics Safety Testing Market for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.65 Biologics Safety Testing Market for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.66 Biologics Safety Testing Market: Distribution by Type of Modality, Current Year, 2030 and 2035
- Table 26.67 Biologics Safety Testing Market for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.68 Biologics Safety Testing Market for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.69 Biologics Safety Testing Market for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.70 Biologics Safety Testing Market for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.71 Biologics Safety Testing Market for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.72 Biologics Safety Testing Market for Traditional viral-based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.73 Biologics Safety Testing Market for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.74 Biologics Safety Testing Market for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.75 Biologics Safety Testing Market: Distribution by Type of Technology, Current Year, 2030 and 2035
- Table 26.76 Biologics Safety Testing Market for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.77 Biologics Safety Testing Market for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.78 Biologics Safety Testing Market for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.79 Biologics Safety Testing Market for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.80 Biologics Safety Testing Market for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.81 Biologics Safety Testing Market: Distribution by End-user, Current Year, 2030 and 2035
- Table 26.82 Biologics Safety Testing Market for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.83 Biologics Safety Testing Market for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.84 Biologics Safety Testing Market for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.85 Biologics Safety Testing Market: Distribution by Geographical Regions, Current Year, 2030 and 2035
- Table 26.86 Biologics Safety Testing Market in North America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.87 Biologics Safety Testing Market in the US, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.88 Biologics Safety Testing Market in Canada, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.89 Biologics Safety Testing Market in Europe, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.90 Biologics Safety Testing Market in Germany, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.91 Biologics Safety Testing Market in the UK, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.92 Biologics Safety Testing Market in France, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.93 Biologics Safety Testing Market in Italy, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.94 Biologics Safety Testing Market in Spain, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.95 Biologics Safety Testing Market in Rest of Europe, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.96 Biologics Safety Testing Market in Asia-Pacific, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.97 Biologics Safety Testing Market in China, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.98 Biologics Safety Testing Market in Japan, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.99 Biologics Safety Testing Market in India, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.100 Biologics Safety Testing Market in South Korea, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.101 Biologics Safety Testing Market in Rest of Asia-Pacific, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.102 Biologics Safety Testing Market in Middle East and North Africa, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.103 Biologics Safety Testing Market in Saudi Arabia, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.104 Biologics Safety Testing Market in Egypt, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.105 Biologics Safety Testing Market in Rest of Middle East and North Africa, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.106 Biologics Safety Testing Market in Latin America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.107 Biologics Safety Testing Market in Brazil, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.108 Biologics Safety Testing Market in Argentina, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Table 26.109 Biologics Safety Testing Market in Rest of Latin America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
List of Figures
- Figure 2.1 Research Methodology: Project Methodology
- Figure 2.2 Research Methodology: Data Sources for Secondary Research
- Figure 2.3 Research Methodology: Robust Quality Control
- Figure 3.1 Market Dynamics: Forecast Methodology
- Figure 3.2 Market Dynamics: Market Assessment Framework
- Figure 4.1 Lessons Learnt from Past Recessions
- Figure 5.1 Executive Summary: Market Landscape
- Figure 5.2 Executive Summary: Market Trends
- Figure 5.3 Executive Summary: Market Sizing and Opportunity Analysis
- Figure 7.1 Biologics Safety Testing Service Providers: Distribution by Year of Establishment
- Figure 7.2 Biologics Safety Testing Service Providers: Distribution by Company Size
- Figure 7.3 Biologics Safety Testing Service Providers: Distribution by Location of Headquarters
- Figure 7.4 Biologics Safety Testing Service Providers: Distribution by Certification / Accreditation
- Figure 7.5 Biologics Safety Testing Service Providers: Distribution by Type of Service Offered
- Figure 7.6 Biologics Safety Testing Service Providers: Distribution by Type of Modality
- Figure 7.7 Biologics Safety Testing Service Providers: Distribution by Type of Sample Supported
- Figure 7.8 Biologics Safety Testing Service Providers: Distribution by Type of Technology
- Figure 7.9 Biologics Safety Testing Service Providers: Distribution by End-user
- Figure 8.1 Company Competitiveness Analysis: Biologics Safety Testing Service Providers Offering Services for Vaccines
- Figure 8.2 Company Competitiveness Analysis: Biologics Safety Testing Service Providers Offering Services for Cell Therapies
- Figure 8.3 Company Competitiveness Analysis: Biologics Safety Testing Service Providers Offering Services for Gene Therapies
- Figure 8.4 Company Competitiveness Analysis: Biologics Safety Testing Service Providers Offering Services for Antibodies
- Figure 8.5 Company Competitiveness Analysis: Biologics Safety Testing Service Providers Offering Services for Proteins
- Figure 8.6 Company Competitiveness Analysis: Biologics Safety Testing Service Providers Offering Services for Other Modalities
- Figure 9.1 Biologics Safety Testing Solution Providers: Distribution by Year of Establishment
- Figure 9.2 Biologics Safety Testing Solution Providers: Distribution by Company Size
- Figure 9.3 Biologics Safety Testing Solution Providers: Distribution by Location of Headquarters
- Figure 9.4 Biologics Safety Testing Solution Providers: Distribution by Type of Offering
- Figure 9.5 Biologics Safety Testing Solution Providers: Distribution by Type of Testing
- Figure 9.6 Biologics Safety Testing Solution Providers: Distribution by Type of Modality
- Figure 9.7 Biologics Safety Testing Solution Providers: Distribution by Type of Sample Analyzed
- Figure 9.8 Biologics Safety Testing Solution Providers: Distribution by Type of Technology
- Figure 9.9 Biologics Safety Testing Solution Providers: Distribution by Application Area
- Figure 9.10 Biologics Safety Testing Solution Providers: Distribution by End-user
- Figure 10.1 Company Competitiveness Analysis: Biologics Safety Testing Solution Providers Headquartered in North America
- Figure 10.2 Company Competitiveness Analysis: Biologics Safety Testing Solution Providers Headquartered in Europe
- Figure 10.3 Company Competitiveness Analysis: Biologics Safety Testing Solution Providers Headquartered in Asia-Pacific and Rest of the world
- Figure 11.1 BioMerieux: Annual Revenues, Since FY 2022 (EUR Million)
- Figure 11.2 Charles River Laboratories: Annual Revenues, Since FY 2022 (USD Million)
- Figure 11.3 Eurofins: Annual Revenues, Since FY 2022 (EUR Million)
- Figure 11.4 SGS: Annual Revenues, Since FY 2022 (CHF Billion)
- Figure 11.5 Vimta Labs: Annual Revenues, Since FY 2022 (INR Million)
- Figure 12.1 Partnerships and Collaborations: Cumulative Year-wise Trend, Since Pre-2020
- Figure 12.2 Partnerships and Collaborations: Distribution by Type of Partnership
- Figure 12.3 Partnerships and Collaborations: Distribution by Year and Type of Partnership
- Figure 12.4 Partnerships and Collaborations: Distribution by Type of Biologic
- Figure 12.5 Partnerships and Collaborations: Distribution by Type of Testing
- Figure 12.6 Partnerships and Collaborations: Distribution by Type of Partner
- Figure 12.7 Most Active Players: Distribution by Number of Partnerships
- Figure 12.8 Partnerships and Collaborations: Local and International Agreements
- Figure 12.9 Partnerships and Collaborations: Intracontinental and Intercontinental Agreements
- Figure 13.1 Funding and Investments: Cumulative Year-wise Trend, Since 2019
- Figure 13.2 Funding and Investments: Distribution of Instances by Type of Funding
- Figure 13.3 Funding and Investments: Distribution of Instances by Year and Type of Funding
- Figure 13.4 Funding and Investments: Distribution of Amount by Year of Funding
- Figure 13.5 Funding and Investments: Distribution of Amount by Type of Funding
- Figure 13.6 Funding and Investments: Distribution by Geography
- Figure 13.7 Key Industry Players: Analysis by Number of Funding Instances
- Figure 13.8 Most Active Players: Analysis by Amount Invested
- Figure 13.9 Most Active Investors: Analysis by Number of Funding Instances
- Figure 13.10 Summary of Investments
- Figure 14.1 Patent Analysis: Distribution by Publication Year
- Figure 14.2 Patent Analysis: Distribution by Type of Patent
- Figure 14.3 Patent Analysis: Distribution by Patent Application Year
- Figure 14.4 Patent Analysis: Distribution by Patent Age
- Figure 14.5 Patent Analysis: Distribution by Type of Applicant
- Figure 14.6 Patent Analysis: Distribution by Patent Jurisdiction
- Figure 14.7 Patent Analysis: Distribution by CPC Symbols
- Figure 14.8 Leading Industry Players: Analysis by Number of Patents
- Figure 14.9 Leading Non-Industry Players: Analysis by Number of Patents
- Figure 14.10 Leading Individual Assignees: Analysis by Number of Patents
- Figure 16.1 Biologics Safety Testing Market, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035): Base Scenario (USD Million)
- Figure 16.2 Biologics Safety Testing Market, Forecasted Estimates (Till 2035): Conservative Scenario (USD Million)
- Figure 16.3 Biologics Safety Testing Market, Forecasted Estimates (Till 2035): Optimistic Scenario (USD Million)
- Figure 17.1 Biologics Safety Testing Market: Distribution by Type of Offering, Current Year, 2030 and 2035
- Figure 17.2 Biologics Safety Testing Market for Consumables, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 17.3 Biologics Safety Testing Market for Instruments, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 17.4 Biologics Safety Testing Market for Services, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 18.1 Biologics Safety Testing Market: Distribution by Type of Testing, Current Year, 2030 and 2035
- Figure 18.2 Biologics Safety Testing Market for Virus Safety Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 18.3 Biologics Safety Testing Market for Mycoplasma Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 18.4 Biologics Safety Testing Market for Sterility Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 18.5 Biologics Safety Testing Market for Endotoxin Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 18.6 Biologics Safety Testing Market for Bioburden Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 18.7 Biologics Safety Testing Market for Other Testing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.1 Biologics Safety Testing Market: Distribution by Type of Modality, Current Year, 2030 and 2035
- Figure 19.2 Biologics Safety Testing Market for Antibodies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.3 Biologics Safety Testing Market for Vaccines, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.4 Biologics Safety Testing Market for Cell Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.5 Biologics Safety Testing Market for Gene Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.6 Biologics Safety Testing Market for Biosimilars, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.7 Biologics Safety Testing Market for Traditional viral-based Therapies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.8 Biologics Safety Testing Market for Proteins, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 19.9 Biologics Safety Testing Market for Other Modalities, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 20.1 Biologics Safety Testing Market: Distribution by Type of Technology, Current Year, 2030 and 2035
- Figure 20.2 Biologics Safety Testing Market for Polymerase Chain Reaction, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 20.3 Biologics Safety Testing Market for Traditional Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 20.4 Biologics Safety Testing Market for Chromatographic and Electrophoretic Techniques, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 20.5 Biologics Safety Testing Market for Next-Generation Sequencing, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 20.6 Biologics Safety Testing Market for Other Technologies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 21.1 Biologics Safety Testing Market: Distribution by End-user, Current Year, 2030 and 2035
- Figure 21.2 Biologics Safety Testing Market for Pharmaceutical and Biotechnology Companies, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 21.3 Biologics Safety Testing Market for Contract Research Organizations / Contract Development and Manufacturing Organizations, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 21.4 Biologics Safety Testing Market for Academic and Research Institutes, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.1 Biologics Safety Testing Market: Distribution by Geographical Regions, Current Year, 2030 and 2035
- Figure 22.2 Biologics Safety Testing Market in North America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.3 Biologics Safety Testing Market in the US, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.4 Biologics Safety Testing Market in Canada, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.5 Biologics Safety Testing Market in Europe, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.6 Biologics Safety Testing Market in Germany, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.7 Biologics Safety Testing Market in the UK, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.8 Biologics Safety Testing Market in France, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.9 Biologics Safety Testing Market in Italy, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.10 Biologics Safety Testing Market in Spain, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.11 Biologics Safety Testing Market in Rest of Europe, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.12 Biologics Safety Testing Market in Asia-Pacific, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.13 Biologics Safety Testing Market in China, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.14 Biologics Safety Testing Market in Japan, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.15 Biologics Safety Testing Market in India, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.16 Biologics Safety Testing Market in South Korea, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.17 Biologics Safety Testing Market in Rest of Asia-Pacific, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.18 Biologics Safety Testing Market in Middle East and North Africa, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.19 Biologics Safety Testing Market in Saudi Arabia, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.20 Biologics Safety Testing Market in Egypt, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.21 Biologics Safety Testing Market in Rest of Middle East and North Africa, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.22 Biologics Safety Testing Market in Latin America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.23 Biologics Safety Testing Market in Brazil, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.24 Biologics Safety Testing Market in Argentina, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)
- Figure 22.25 Biologics Safety Testing Market in Rest of Latin America, Historical Trends (Since 2021) and Forecasted Estimates (Till 2035)











