 |
市场调查报告书
商品编码
1959570
免疫查核点抑制剂市场机会、成长要素、产业趋势分析及2026年至2035年预测。Immune Checkpoint Inhibitors Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035 |
||||||
2025 年全球免疫查核点抑制剂市场价值为 662 亿美元,预计到 2035 年将达到 3,039 亿美元,年复合成长率为 16.6%。
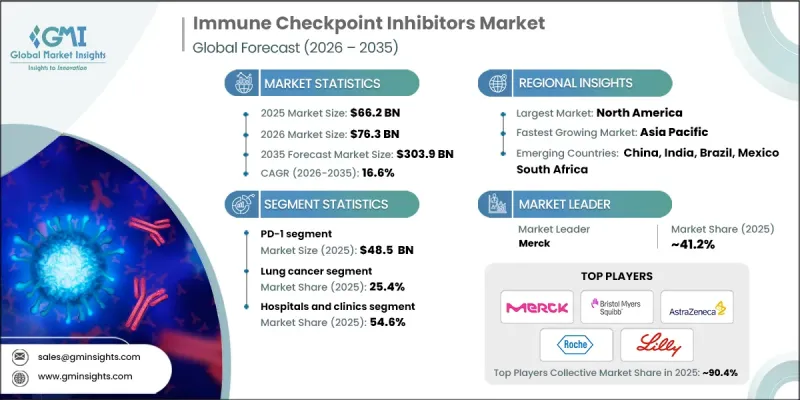
市场扩张的驱动力在于这些治疗方法在多种癌症类型中已证实的临床疗效,以及它们在肿瘤治疗中日益普及并成为标准疗法。随着全球癌症发生率的持续上升,对强效标靶治疗方法(例如免疫查核点抑制剂)的需求也日益增长。这些治疗方法透过抑制PD-1、PD-L1和CTLA-4等免疫查核点蛋白发挥作用,进而活化人类免疫系统对抗癌细胞。癌细胞利用这些蛋白质来逃避免疫系统的检测。抑制这些蛋白质可以恢復T细胞功能,使免疫系统能够有效地靶向并清除恶性细胞。随着晚期难治性癌症患者数量的增加,免疫疗法的适用人群也在不断扩大,而开发更有效、更精准、更安全的抑制剂的持续创新,仍然是推动市场成长的关键因素。
| 市场范围 | |
|---|---|
| 开始年份 | 2025 |
| 预测年份 | 2026-2035 |
| 起始值 | 662亿美元 |
| 预测金额 | 3039亿美元 |
| 复合年增长率 | 16.6% |
预计到2025年,PD-1抑制剂市场规模将达到485亿美元,反映了其在癌症免疫疗法中的关键作用。 PD-1抑制剂透过抑制PD-1路径来增强免疫系统攻击癌细胞的能力,而癌细胞通常利用PD-1路径来逃避免疫系统的辨识。这些治疗方法的成功,得益于显着的临床疗效、不断增加的核准适应症以及在肿瘤临床实践中的广泛应用,已使PD-1抑制剂成为现代癌症治疗的基石。它们在多种癌症类型中的疗效正在巩固其市场主导地位,并强化其在临床肿瘤学中的重要性。
肺癌领域占25.4%的市场份额,预计2035年将以16%的复合年增长率成长。这一主导地位源自于肺癌的高发生率以及免疫查核点抑制剂在提高存活率和改善患者预后方面所展现的临床效益。免疫疗法作为一线治疗方法的日益普及,以及正在进行的探索其在不同肺癌阶段疗效的临床试验,进一步巩固了其市场地位。沉重的疾病负担和未被满足的医疗需求推动了对这些治疗方法的需求,从而维持了该领域的强劲成长。
预计到2025年,北美免疫查核点抑制剂市占率将达48.3%。这一主导地位得益于许多大型製药公司积极参与此类治疗方法的研发和商业化。该地区拥有良好的法规环境,政府和非政府组织对癌症治疗倡议的大力支持,以及癌症发病率上升带来的日益增长的需求。加之先进的医疗基础设施、对免疫疗法的广泛认知以及获得创新治疗方案的便利,北美正在巩固其在全球市场的主导地位。
目录
第一章:调查方法和范围
第二章执行摘要
第三章业界考察
- 生态系分析
- 影响产业的因素
- 促进因素
- 全球癌症发生率不断上升
- 扩大核准,用于多种癌症适应症
- 免疫疗法作为标准治疗方案的转变正在加速。
- 扩大投资和合作关係
- 产业潜在风险与挑战
- 免疫相关不利事件风险增加
- 高昂的医疗费用
- 市场机会
- 联合免疫疗法方案的增长
- 下一代免疫查核点靶点的开发
- 促进因素
- 成长潜力分析
- 监理情势
- 救赎方案
- 管道分析
- 价格分析
- 未来市场趋势
- 波特五力分析
- PESTEL 分析
第四章 竞争情势
- 介绍
- 企业市占率分析
- 世界
- 北美洲
- 欧洲
- 亚太地区
- 公司矩阵分析
- 主要市场公司的竞争分析
- 竞争定位矩阵
- 主要进展
- 併购
- 伙伴关係与合作
- 新产品发布
- 扩张计划
第五章 市场估计与预测:依类型划分,2022-2035年
- PD-1
- PD-L1
- CTLA-4
- 其他类型
第六章 市场估计与预测:依应用领域划分,2022-2035年
- 肺癌
- 乳癌
- 膀胱癌
- 恶性黑色素瘤
- 子宫颈癌
- 何杰金氏淋巴瘤
- 结肠癌
- 其他用途
第七章 市场估计与预测:依最终用途划分,2022-2035年
- 医院和诊所
- 癌症中心
- 学术研究机构
第八章 市场估计与预测:依地区划分,2022-2035年
- 北美洲
- 我们
- 加拿大
- 欧洲
- 德国
- 英国
- 法国
- 西班牙
- 义大利
- 荷兰
- 亚太地区
- 中国
- 印度
- 日本
- 澳洲
- 韩国
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中东和非洲
- 南非
- 沙乌地阿拉伯
- 阿拉伯聯合大公国
第九章:公司简介
- AstraZeneca
- BeiGene
- Bristol-Myers Squibb Company
- Eli Lilly and Company
- F. Hoffmann-La Roche
- GlaxoSmithKline
- Incyte Corporation
- Immutep Limited
- Merck
- Regeneron Pharmaceuticals
- Sanofi
- Shanghai Junshi Biosciences
- Sun Pharmaceuticals
- Zydus Lifesciences
The Global Immune Checkpoint Inhibitors Market was valued at USD 66.2 billion in 2025 and is estimated to grow at a CAGR of 16.6% to reach USD 303.9 billion by 2035.

The market expansion is driven by the proven clinical effectiveness of these therapies across a broad spectrum of cancers and their increasing adoption as a standard of care in oncology. As cancer incidence continues to rise worldwide, the demand for potent and targeted treatments such as immune checkpoint inhibitors is intensifying. These therapies work by enhancing the body's immune system to combat tumor cells, specifically by blocking immune checkpoint proteins like PD-1, PD-L1, and CTLA-4, which are exploited by cancer cells to evade immune detection. By inhibiting these proteins, T-cell function is restored, enabling the immune system to effectively target and eliminate malignant cells. The rising prevalence of hard-to-treat and late-stage cancers is increasing the patient population for immunotherapy, while ongoing innovation in the development of more effective, precise, and safe inhibitors continues to propel market growth.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $66.2 Billion |
| Forecast Value | $303.9 Billion |
| CAGR | 16.6% |
The PD-1 segment reached USD 48.5 billion in 2025, reflecting its critical role in cancer immunotherapy. PD-1 inhibitors enhance the immune system's ability to attack tumor cells by blocking the PD-1 pathway, which is commonly used by cancer cells to escape immune recognition. The success of these therapies is supported by strong clinical outcomes, a growing list of approved indications, and widespread adoption in oncology practices, positioning PD-1 inhibitors as a cornerstone in modern cancer treatment. Their effectiveness across multiple cancer types reinforces their dominant share and solidifies their importance in clinical oncology.
The lung cancer segment held 25.4% share and is expected to grow at a CAGR of 16% through 2035. This dominance is attributed to the high prevalence of lung cancer and the demonstrated clinical benefits of immune checkpoint inhibitors in improving survival rates and patient outcomes. The increasing adoption of immunotherapy as a primary treatment, combined with ongoing clinical trials exploring its efficacy in different stages of lung cancer, continues to strengthen market presence. The high disease burden and unmet medical needs further drive demand for these therapies, sustaining robust growth in this segment.
North America Immune Checkpoint Inhibitors Market held 48.3% share in 2025. This leadership is supported by the presence of major pharmaceutical companies actively engaged in research, development, and commercialization of these therapies. The region benefits from a favorable regulatory environment, strong governmental and non-governmental support for cancer treatment initiatives, and rising demand driven by increasing cancer prevalence. Advanced healthcare infrastructure, widespread awareness of immunotherapy options, and access to innovative treatment solutions collectively strengthen North America's dominant position in the global market.
Key players in the Global Immune Checkpoint Inhibitors Market include Merck, Bristol-Myers Squibb Company, AstraZeneca, BeiGene, GlaxoSmithKline, Eli Lilly and Company, Sanofi, Immutep Limited, Incyte Corporation, Shanghai Junshi Biosciences, Zydus Lifesciences, Regeneron Pharmaceuticals, F. Hoffmann-La Roche, and Sun Pharmaceuticals. Companies in the immune checkpoint inhibitors market are leveraging multiple strategies to expand their footprint and reinforce their market presence. They are investing heavily in research and development to create next-generation inhibitors with improved safety profiles, broader efficacy, and expanded indications across cancer types. Strategic partnerships with biotechnology firms, hospitals, and academic institutions accelerate clinical trials and innovation. Geographic expansion into emerging markets allows firms to tap into growing patient populations. Additionally, mergers and acquisitions, licensing agreements, and collaborations with regulatory agencies streamline market entry and product approvals.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research approach
- 1.3 Quality commitments
- 1.3.1 GMI AI policy and data integrity commitment
- 1.3.1.1 Source consistency protocol
- 1.3.1 GMI AI policy and data integrity commitment
- 1.4 Research trail and confidence scoring
- 1.4.1 Research trail components
- 1.4.2 Scoring components
- 1.5 Data collection
- 1.5.1 Partial list of primary sources
- 1.6 Data mining sources
- 1.6.1 Paid sources
- 1.6.1.1 Sources, by region
- 1.6.1 Paid sources
- 1.7 Base estimates and calculations
- 1.7.1 Revenue share analysis
- 1.7.2 Base year calculation
- 1.8 Forecast model
- 1.9 Research transparency addendum
- 1.9.1 Source attribution framework
- 1.9.2 Quality assurance metrics
- 1.9.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Type trends
- 2.2.3 Application trends
- 2.2.4 End use trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising prevalence of cancer worldwide
- 3.2.1.2 Expanding approvals across multiple cancer indications
- 3.2.1.3 Growing shift toward immunotherapy as standard of care
- 3.2.1.4 Increasing investments and partnerships
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Growing risk of immune-related adverse events
- 3.2.2.2 High treatment costs
- 3.2.3 Market opportunities
- 3.2.3.1 Growth of combination immunotherapy regimens
- 3.2.3.2 Development of next-generation immune checkpoint targets
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.4 Latin America
- 3.4.5 Middle East and Africa
- 3.5 Reimbursement scenario
- 3.6 Pipeline analysis
- 3.7 Pricing analysis
- 3.8 Future market trends
- 3.9 Porter's analysis
- 3.10 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 Global
- 4.2.2 North America
- 4.2.3 Europe
- 4.2.4 Asia Pacific
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 PD-1
- 5.3 PD-L1
- 5.4 CTLA-4
- 5.5 Other types
Chapter 6 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Lung cancer
- 6.3 Breast cancer
- 6.4 Bladder cancer
- 6.5 Melanoma
- 6.6 Cervical cancer
- 6.7 Hodgkin lymphoma
- 6.8 Colorectal cancer
- 6.9 Other applications
Chapter 7 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Hospitals and clinics
- 7.3 Cancer centers
- 7.4 Academic and research institutes
Chapter 8 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 India
- 8.4.3 Japan
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 Middle East and Africa
- 8.6.1 South Africa
- 8.6.2 Saudi Arabia
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 AstraZeneca
- 9.2 BeiGene
- 9.3 Bristol-Myers Squibb Company
- 9.4 Eli Lilly and Company
- 9.5 F. Hoffmann-La Roche
- 9.6 GlaxoSmithKline
- 9.7 Incyte Corporation
- 9.8 Immutep Limited
- 9.9 Merck
- 9.10 Regeneron Pharmaceuticals
- 9.11 Sanofi
- 9.12 Shanghai Junshi Biosciences
- 9.13 Sun Pharmaceuticals
- 9.14 Zydus Lifesciences










